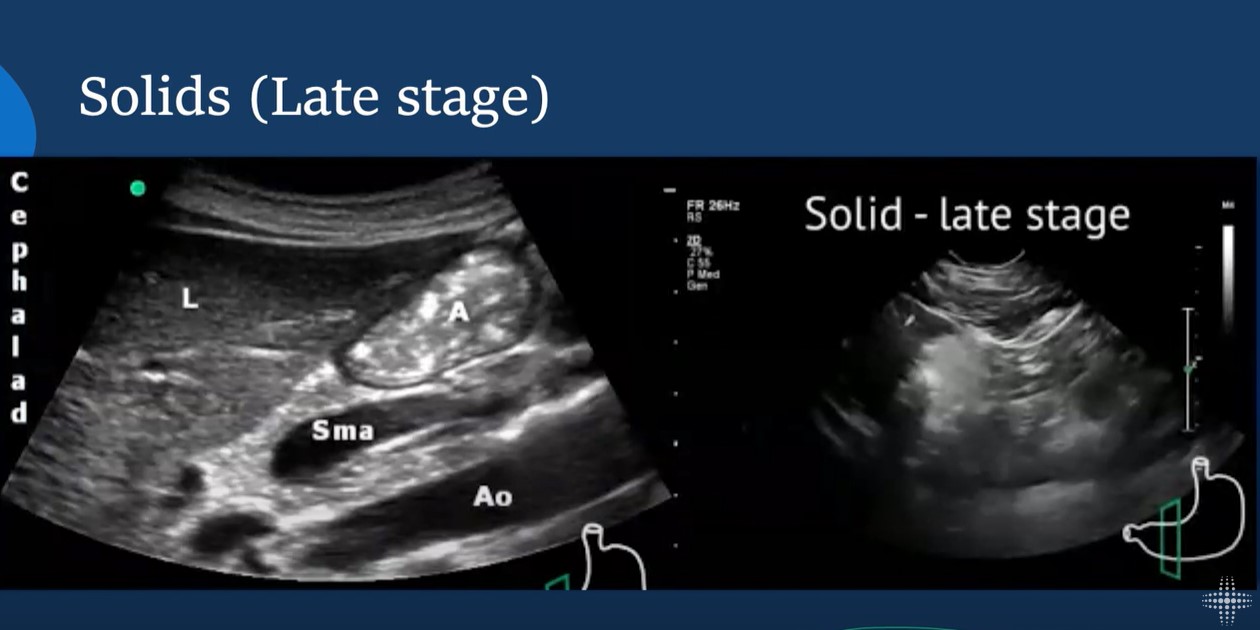
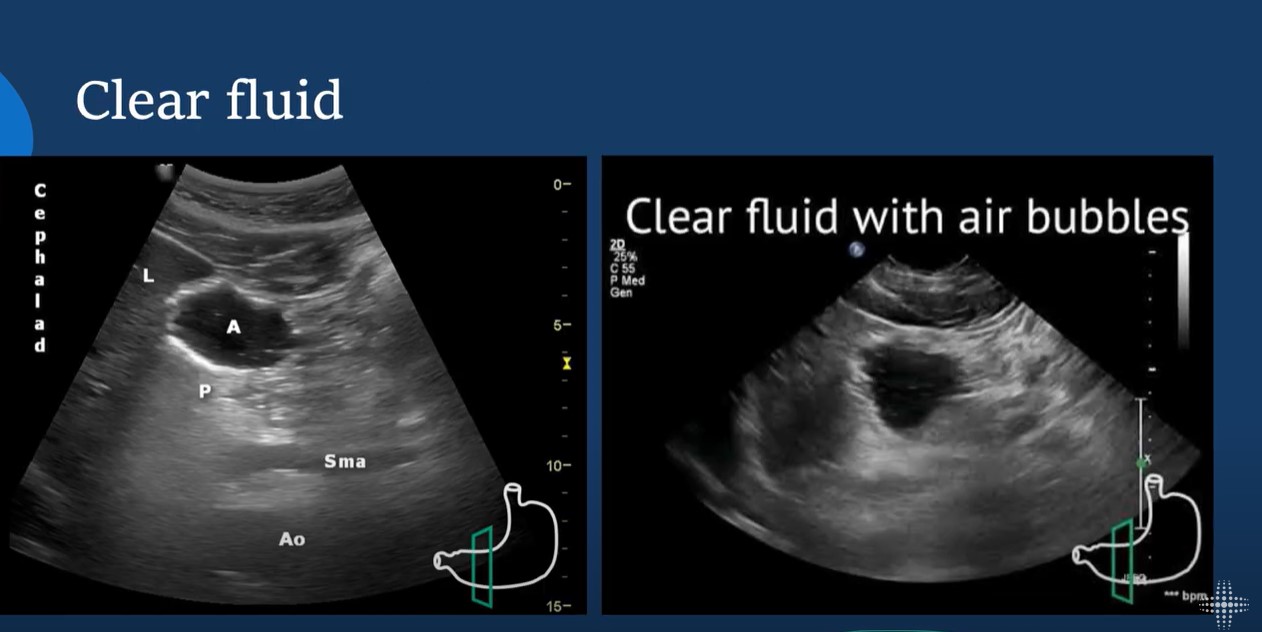
Today, I review, link to, and embed Gastric POCUS From MetroHealth Emergency Ultrasound.
All that follows is from the above resource.
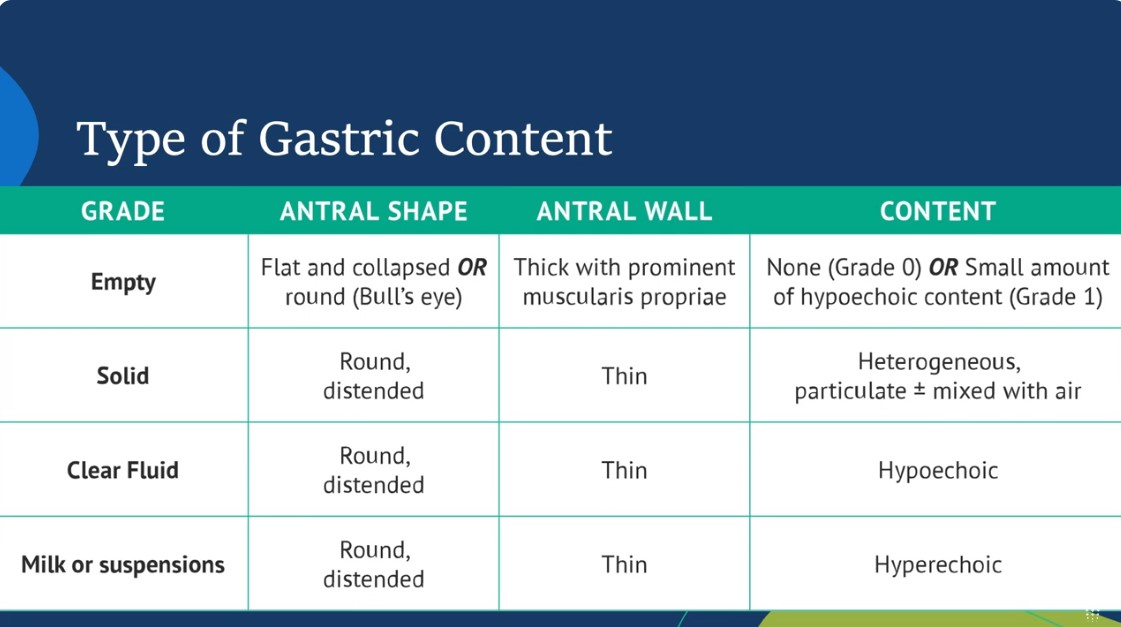
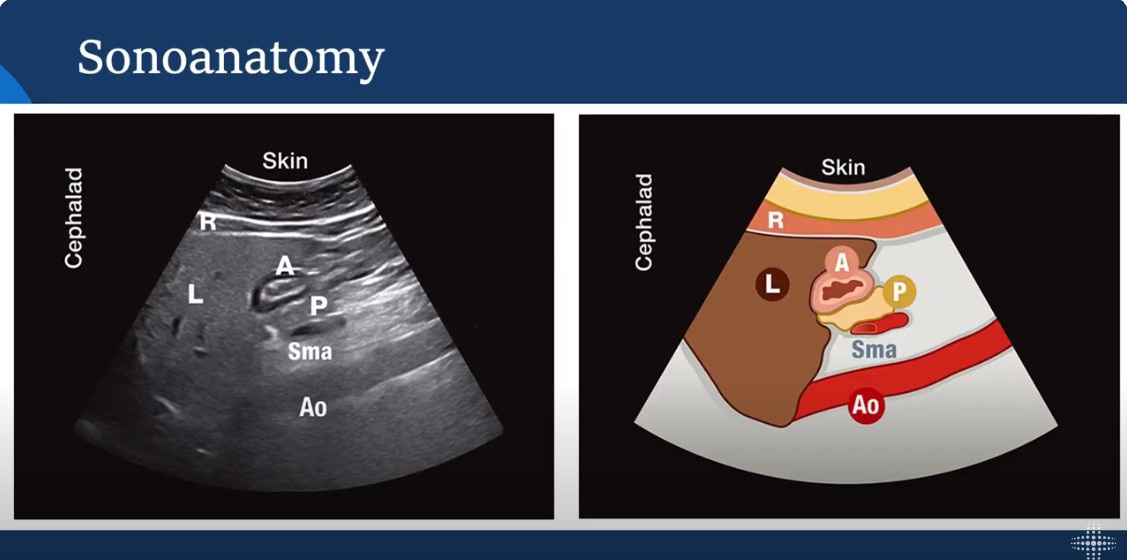
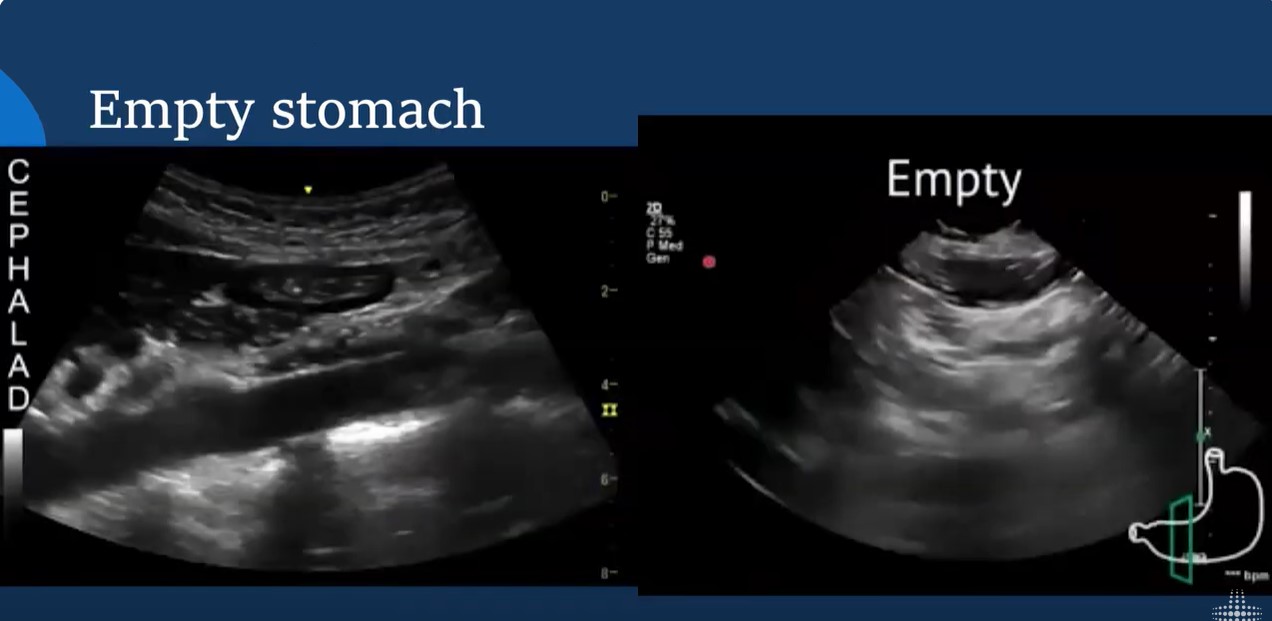
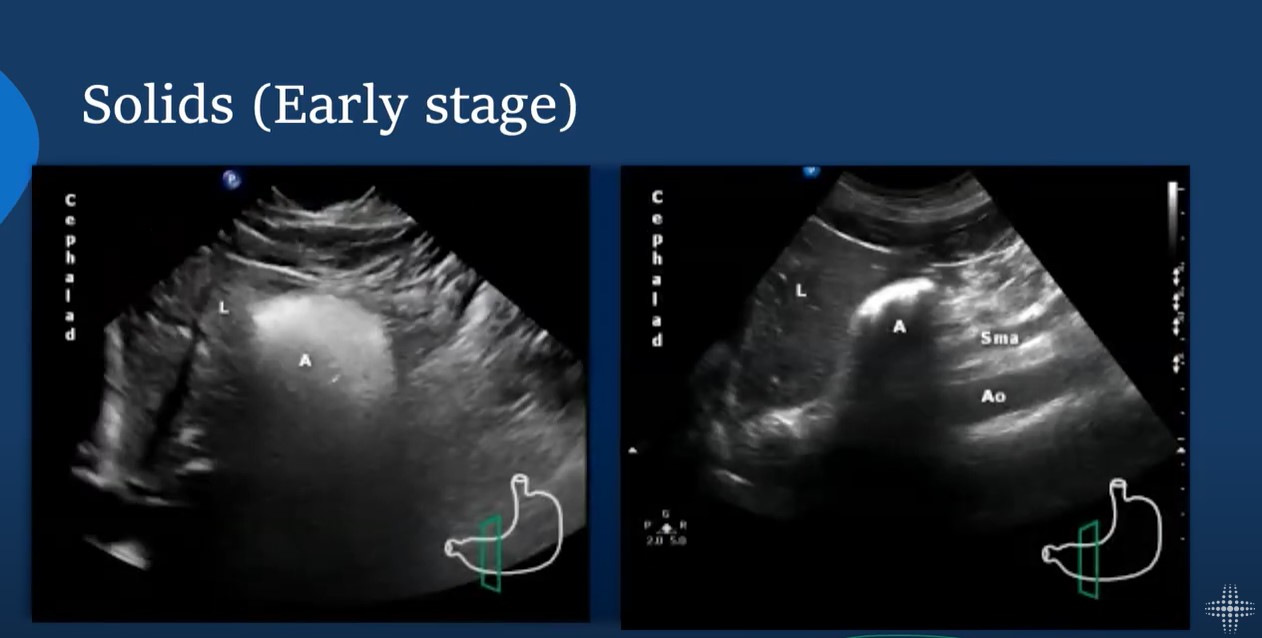
Apr 15, 2024In this lecture from our Ultrasound Grand Rounds, Dr. Dan Schwartz MD and Dr. Michael Leeds MD from anesthesiology talks gastric ultrasound and how obtaining views of the stomach can assist in minimizing aspiration risk during airway management.
Follow us on Online and on Social Media:
Website: https://www.metrohealth.org/pocus
Twitter:
/ mh_emultrasound
Instagram:
/ mh_emultrasound
1:15
2:35
3:19
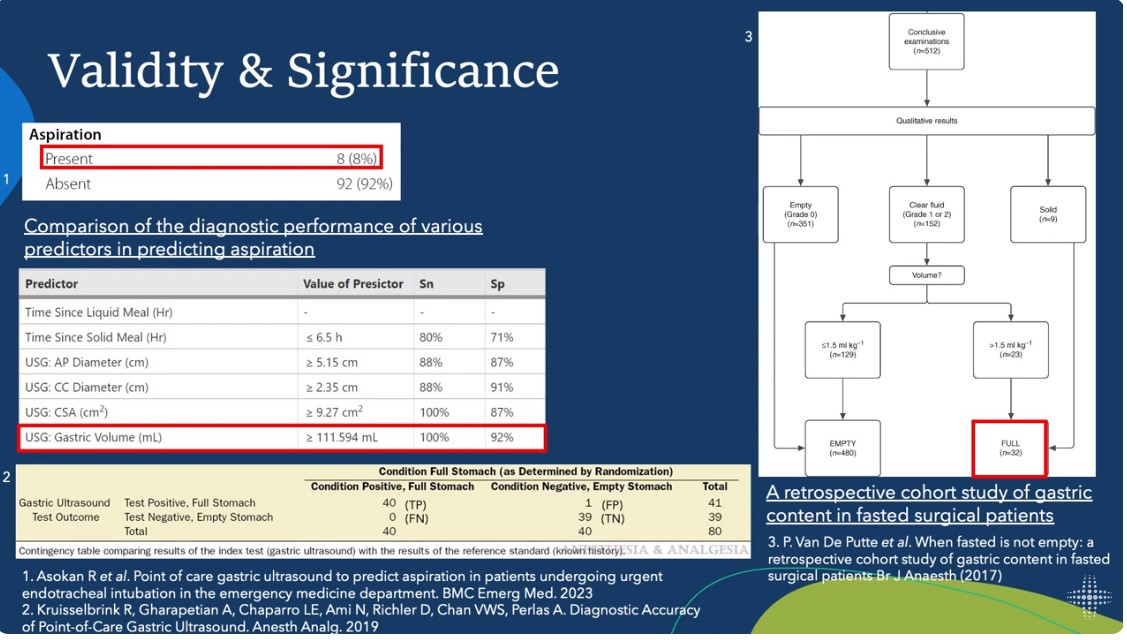
5:13 Aspiration is more common in theED than in the OR [because of need for emergency intubation]
8:32
9:35
11:31
14:10
14:30
15:05
16:50
18:18
19:05