In addition to today’s article, please review Transient Elastography and Fibroscan: Stethoscope of a Hepatologist in Today’s World [PubMed Abstract] [Full-Text HTML] [Full-Text PDF]. Curr Hepatol Rep. 2025 Dec;24(1):10.1007/s11901-025-00713-7. doi: 10.1007/s11901-025-00713-7. Epub 2025 Nov 27.
Abstract
Purpose of review: Vibration-controlled transient elastography (VCTE) has become a cornerstone in the noninvasive assessment of liver disease across etiologies. This review highlights its role in risk stratification and longitudinal monitoring, emphasizing its value in both clinical practice and research settings, including community and academic hepatology.
Recent findings: VCTE reliably assesses liver fibrosis severity and clinically significant portal hypertension and enables ongoing monitoring of therapeutic response. It also predicts liver-related events and mortality. Recognizing its limitations and potential confounders is essential to ensure diagnostic accuracy, and its performance can be further enhanced by integrating clinical and biochemical variables.
Summary: Despite inherent limitations, improvements in probe design, standardized protocols, and composite algorithms have expanded its clinical utility. The 2022 Baveno consensus established clear guidance for using VCTE to identify clinically significant portal hypertension, ultimately aiming to prevent first decompensation and improve patient outcomes.
Keywords: Clinically significant portal hypertension (CSPH); Compensated advanced chronic liver disease (cACLD); Liver fibrosis measurement (LSM); Non-invasive test (NIT); Vibration controlled transient elastography (VCTE).
Today, I review, link to, and excerpt from Clinical Care Pathway for the Risk Stratification and Management of Patients With Nonalcoholic Fatty Liver Disease. [PubMed Abstract] [Full-Text HTML] [Full-Text PDF]. Gastroenterology. 2021 Nov;161(5):1657-1669. doi: 10.1053/j.gastro.2021.07.049. Epub 2021 Sep 20.
There are 106 similar articles in PubMed.
The above article has been cited by 241 articles in PubMed.
All that follows is from the above resource.
- Abstract
- Pathway Development
- Screening for Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis
- Management of Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis
- Management of Nonalcoholic Fatty Liver Disease in Patients at Low Risk of Advanced Fibrosis
- Management of Nonalcoholic Fatty Liver Disease in Patients at High Risk of Advanced Fibrosis
- Management of Nonalcoholic Fatty Liver Disease in Patients at Indeterminate Risk of Advanced Fibrosis
- Summary
- Acknowledgments
- Abbreviations used in this paper:
- References
Abstract
Find AGA’s NASH Clinical Care Pathway App for iOS and Android mobile devices at nash.gastro.org. Scan this QR code to be taken directly to the website.Nonalcoholic fatty liver disease (NAFLD) is becoming increasingly common, currently affecting approximately 37% of US adults. NAFLD is most often managed in primary care or endocrine clinics, where clinicians must determine which patients might benefit from secondary care to address hepatic manifestations, comorbid metabolic traits, and cardiovascular risks of the disease. Because NAFLD is largely asymptomatic, and because optimal timing of treatment depends on accurate staging of fibrosis risk, screening at the primary care level is critical, together with consistent, timely, evidence-based, widely accessible, and testable management processes. To achieve these goals, the American Gastroenterological Association assembled a multidisciplinary panel of experts to develop a Clinical Care Pathway providing explicit guidance on the screening, diagnosis, and treatment of NAFLD. This article describes the NAFLD Clinical Care Pathway they developed and provides a rationale supporting proposed steps to assist clinicians in diagnosing and managing NAFLD with clinically significant fibrosis (stage F2-F4) based on the best available evidence. This Pathway is intended to be applicable in any setting where care for patients with NAFLD is provided, including primary care, endocrine, obesity medicine, and gastroenterology practices.
Keywords: Clinical Care Pathway; Liver Disease; NAFLD; NASH; Nonalcoholic Fatty Liver Disease; Nonalcoholic Steatohepatitis.
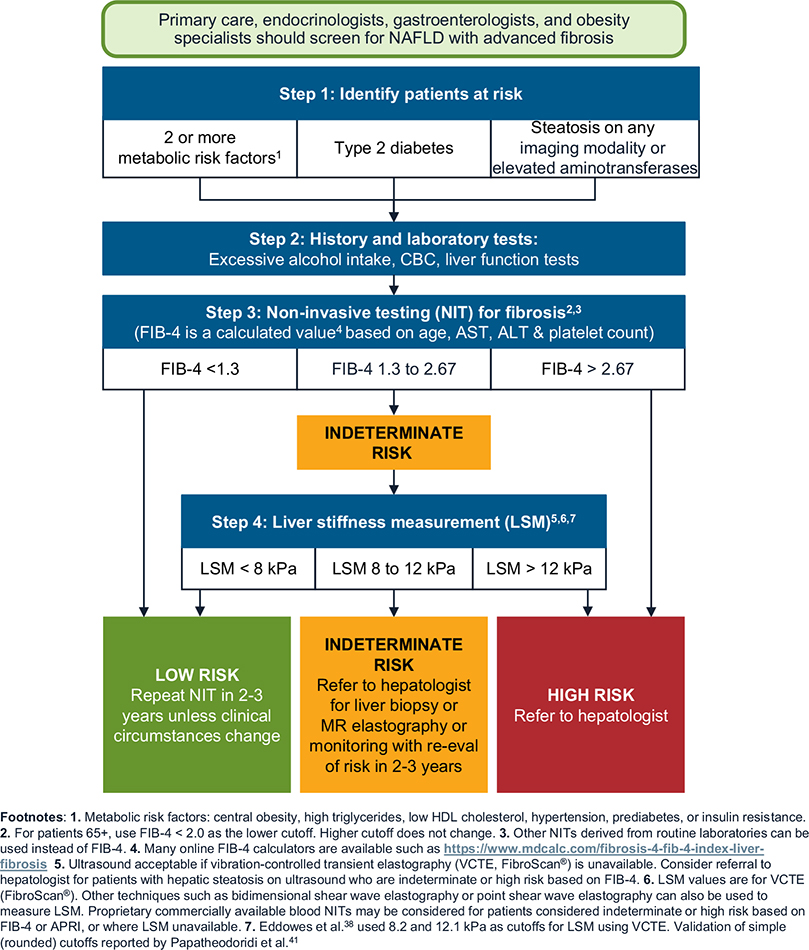
Screening for advanced fibrosis related to NAFLD/NASH.
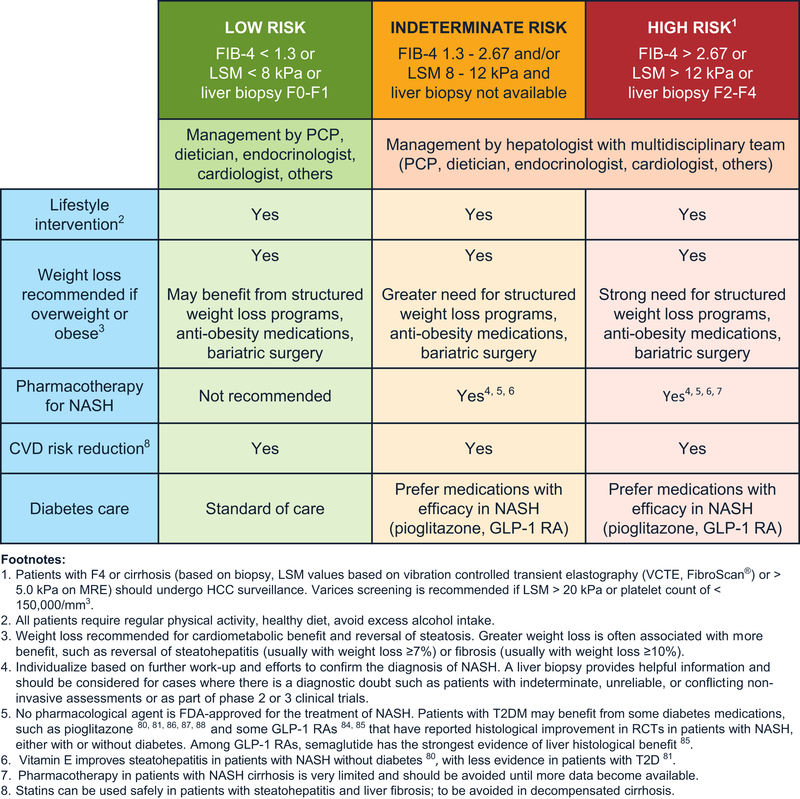
Management of NAFLD/NASH.
Screening for Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis
Step 1: Identify Patients at Risk for Clinically Significant FIbrosis
Patients with T2D: Many studies report a high prevalence of clinically significant fibrosis in patients with T2D, with as many as 20% of these patients affected in recent studies.1,17 We recommend clinicians screen all patients with T2D. This approach has been demonstrated to be cost-effective.16
Patients with 2 or more metabolic risk factors: In a large retrospective study of 271,906 patients with NAFLD, patients with only 1 or no metabolic trait (eg, hypertension, dyslipidemia, or obesity) had a low risk of progression to cirrhosis or HCC.18 There was a stepwise increase in risk of progression to cirrhosis or HCC with each additional metabolic trait. Compared with patients with no metabolic trait, patients with both hypertension and dyslipidemia had a 1.8-fold higher risk of progression to cirrhosis or HCC. Therefore, we recommend that clinicians screen patients with 2 or more metabolic conditions for NAFLD-related clinically significant hepatic fibrosis. The metabolic conditions include central obesity, defined by waist circumference with ethnicity-specific cutoffs; raised serum triglycerides, ≥150 mg/dL, or specific treatment for hypertriglyceridemia; reduced serum high-density lipoprotein cholesterol, <40 mg/dL in men, <50 mg/dL in women, or specific treatment; hypertension, systolic blood pressure ≥130 mm Hg or diastolic blood pressure ≥85 mm Hg, or specific treatment; and raised fasting plasma glucose, between 100 mg/dL19 and 125 mg/dL (prediabetes).
Patients with incidental finding of hepatic steatosis or elevated aminotransferases: Some patients undergoing thoracic and abdominal imaging for reasons other than liver symptoms, signs, or abnormal liver biochemistry can demonstrate unsuspected hepatic steatosis. Studies suggest that 11% of patients with incidentally discovered hepatic steatosis might be at high risk for advanced hepatic fibrosis.20 This appears to be particularly true in patients with elevated aminotransferases. A recent retrospective cohort study found that patients with hepatic steatosis and elevated alanine aminotransferase had a significantly higher risk of progression to cirrhosis or HCC than patients with hepatic steatosis and persistently normal alanine aminotransferase.21 These findings support our recommendation to evaluate patients with unsuspected hepatic steatosis detected on imaging, especially those with abnormal liver chemistries, for presence of NAFLD and clinically significant fibrosis. However, further research on the accuracy and costeffectiveness of this strategy is required.
Step 2: Conduct Standard History and Blood Tests to Obtain Key Measures
We recommend that all at-risk patients identified in Step 1 be screened for alcohol use and have liver tests (or comprehensive metabolic panel, if done as part of routine care) and a complete blood count as part of the initial screening process. The US Preventive Services Task Force recommends that all adults 18 years and older be screened for alcohol use disorders, using the Alcohol Use Disorders Identification Test, Alcohol Use Disorders Identification Test-Concise, or single-question screening tool.22 Results from standard laboratory testing can allow clinicians to calculate simple fibrosis scores (such as Fibrosis-4 [FIB-4] or NAFLD Fibrosis Score) that rely on serum levels of aminotransferases, albumin, and platelets (Step 3).23
These initial laboratory tests can also identify patients with elevated aminotransferases, all of whom should be evaluated for presence of other chronic liver and biliary diseases, including chronic hepatitis C virus infection, chronic hepatitis B virus infection, alcohol-related liver disease, and mass lesions (via liver imaging) (Figure 2). Other tests to evaluate for rare liver diseases can be performed in primary care clinics. Alternatively, patients can be referred to specialty hepatology clinics for further evaluation and management.
Evaluate for other forms of liver disease.
Abdominal ultrasound is commonly used to diagnose hepatic steatosis and has high accuracy for detecting moderate and severe steatosis,24 but it has suboptimal sensitivity for mild steatosis.25 In patients with a high pretest probability of NAFLD, such as the 3 at-risk groups identified in Step 1, moving directly to risk stratification (Step 3) is reasonable without an abdominal ultrasound to diagnosis hepatic steatosis.
Step 3: Conduct Noninvasive Testing for Liver Fibrosis Using Simple Scores
We recommend that all individuals in the target risk groups undergo a 2-tier process to assess for clinically significant liver fibrosis. The first tier involves using simple, nonproprietary fibrosis scores. Several proprietary scores are available but might not be cost-effective to use in all clinical situations. The Pathway relies on the FIB-4 score because it has been shown to have the best diagnostic accuracy for advanced fibrosis compared with other noninvasive markers of fibrosis in patients with NAFLD.23,26 FIB-4 score also correlates with clinical outcomes27,28 in patients with NAFLD. Other noninvasive tests, such as aspartate transaminase to platelet ratio index can be used in lieu of FIB-4.23 Of note, all noninvasive fibrosis scores are more accurate in distinguishing patients with from those without advanced fibrosis (F3 or higher). Therefore, the cutoffs for the noninvasive fibrosis scores, although evidence-based, might miss some patients with F2 at an initial assessment. However, these patients should be detected during future assessments as they progress.
Previous studies have shown that FIB-4 score <1.3 (<2.0 in those older than 65 years) can reliably exclude advanced fibrosis in patients with NAFLD, with a negative predictive value of ≥90%.23,26,29,30 As a result, FIB-4 provides a useful, inexpensive, first-line assessment of liver fibrosis for use in primary care. Patients with values below this cutoff do not need further evaluation, but we recommend that clinicians consider these patients for repeat testing with FIB-4 in 2–3 years. This recommendation is supported by a prospective study using serial transient elastography in patients with T2D, in which only 12% of patients had a ≥30% relative increase in liver stiffness after 3 years of follow-up.31
Patients with FIB-4 score >2.67 are at high risk for advanced fibrosis,29 with most studies reporting positive predictive values of 60%–80%.23,32–34 We recommend referring these patients to hepatology, where they can be considered for liver stiffness measurement (LSM) or liver biopsy to confirm liver fibrosis stage. The remaining 30%-40% of patients with an FIB-4 test would likely have values in the indeterminate range (ie, 1.3–2.67).1 These patients should also undergo LSM, depending on the clinical setting (see Step 4), which can be done as a point-of-care test (if available) in the primary care or endocrinology clinic, ordered by the clinician as other imaging tests to be reviewed at the next visit or as part of a referral to hepatology.
Of note, the negative and positive predictive values of the novel imaging techniques (NITs) depend on the prevalence of advanced fibrosis in the target population, with prevalence being lower in the primary care clinic populations and higher in specially clinic populations. However, recent studies have shown that the prevalence of advanced fibrosis (F3–F4) in primary care clinic populations of patients with risk factors for NAFLD (such as diabetes) is higher than previously believed, ranging from 9% to 15% in different studies.1,2 Furthermore, most NITs have high negative predictive values at the low cutoffs and can reliably rule out clinically significant fibrosis. Indeed, this 2-tier, risk-stratification process is supported in part by a study of 759 patients with biopsy-proven NAFLD showing that using noninvasive fibrosis scores followed by LSM (with FibroScan) only for patients with indeterminate or high scores was most accurate for diagnosing advanced fibrosis.35 In another study of 968 patients with biopsy-proven NAFLD, sequential testing using NAFLD Fibrosis Score or FIB-4 followed by FibroScan for patients with indeterminate score was more accurate than using tests individually.32 Sequential testing may also be justified because the performance of noninvasive fibrosis scores for the diagnosis of advanced fibrosis in NASH appears to be less optimal in patients with T2D, with a significant number of patients falling into the indeterminate group.36
For aspartate transaminase to platelet ratio index, a study from a tertiary center in India found a value of ≤0.48 had a negative predictive value of 78% and a value of ≥1.34 had a positive predictive value of 78%, with values between 0.49 and 1.33 representing the indeterminate group.37
The Pathway uses the currently available NITs. We expect the Pathway will be updated as more precise markers are developed and validated.
Step 4: Obtain a Liver Stiffness Measurement
The second tier relies on an imaging-based test for LSM, depending on the initial FIB-4 score result. To assess liver stiffness, the Pathway uses FibroScan, which is based on vibration controlled transient elastography (VCTE).38 New techniques, such as bidimensional shear wave elastography or point shear wave elastography, can also be used to assess LSM, with diagnostic performances at least as good as VCTE (with FibroScan).39,40
FibroScan (transient elastography) scores, measured in kilopascals (kPa), reflect risk for clinically significant fibrosis. In a study of 450 consecutive adults who underwent liver biopsy and FibroScan for suspected NAFLD at 7 centers, the Youden cutoff value for F≥F2 was 8.2 kPa.38 This cutoff was associated with high negative predictive values for stage 2 fibrosis in patients seen in diabetes clinics or the general population (78% and 97%, respectively), although the negative predictive value was modest in specialty hepatology clinic populations (61%). However, using VCTE, an LSM of <8.2 kPa excluded advanced fibrosis (fibrosis stage 3 and 4) with negative predictive values of >80% in all populations.38 To allow easy implementation, we recommend a simplified rounded value of 8.0 kPa as the low cutoff to exclude clinically significant fibrosis (ie, fibrosis stage F2–F4) for LSM using VCTE. Among 1073 patients with NAFLD from 10 European liver centers who had a liver biopsy and LSM within 6 months, a low cutoff of 8 kPa has a 93% sensitivity to exclude advanced fibrosis.41 A recent systematic review also provides support to the low cutoff of 8 kPa.34 Given these data, patients with LSM (using VCTE) <8.0 kPa can be considered low risk for clinically significant fibrosis and are best managed with repeat surveillance testing in 2–3 years.
A value of >12.1 kPa on VCTE indicates that clinically significant fibrosis is likely, with positive predictive values of 76% and 88% in patients seen in diabetes and hepatology clinic populations, respectively, although the positive predictive value can be low in primary care populations.38 We recommend using a rounded-off value of 12.0 kPa as the upper cutoff.41 We further recommend referring these high-risk patients (>12.0 kPa) to a hepatologist, if not already in hepatology care, for consideration of liver biopsy or magnetic resonance elastography (MRE). A liver biopsy is usually indicated, although a FIB-4 score >2.67 together with an LSM using VCTE ≥12.0 kPa is highly suggestive of advanced liver fibrosis. Additional nonproprietary and proprietary plasma tests, or imaging by MRE, can also be considered to confirm findings. Ina cross-sectional analysis of a prospective cohort of 238 consecutive patients with MRE and biopsy-proven NAFLD, MRE ≥3.3 kPa and FIB-4 score ≥1.6 ruled in stage 2 or higher fibrosis with a positive predictive value of 97.1%.42 This combination remained significant at a positive predictive value of 91.0% in a separate validation cohort, suggesting that patients meeting both cutoffs on the sequential application of noninvasive tests might not need to undergo a liver biopsy for subsequent risk stratification.42 However, this study included patients seen in specialty clinics with a high prevalence of hepatic fibrosis. These data will need confirmation in patients with NAFLD seen in other clinical settings. An LSM ≥20 kPa on VCTE or thrombocytopenia is highly suggestive of cirrhosis. These patients also have a risk of gastroesophageal varices requiring treatment and should undergo variceal screening.43
We recommend that patients with discordant or indeterminate LSM results (ie, 8.0–12.0 kPa) in primary care and endocrine clinics be referred to hepatology where, like high-risk patients, they might need to undergo either a liver biopsy or MRE for further diagnostic evaluation. We recognize the need for physician–patient shared decision making and individualized care, and it may be appropriate to follow the patients annually with repeated LSMs if this strategy is consistent with patients’ preferences.
We recommend proprietary, commercially available blood or NITs44–47 for patients considered indeterminate or high risk based on FIB-4 score or aspartate transaminase to platelet ratio index where LSM is unavailable.
Please see the article to review the following sections:
- Management of Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis
- Management of Nonalcoholic Fatty Liver Disease in Patients at Low Risk of Advanced Fibrosis
- Management of Nonalcoholic Fatty Liver Disease in Patients at High Risk of Advanced Fibrosis
- Management of Nonalcoholic Fatty Liver Disease in Patients at Indeterminate Risk of Advanced Fibrosis
- Summary