Today, I review, link to, and excerpt from the Obesity Association‘s “Pharmacologic Treatment of Obesity in Adults: Standards of Care in Overweight and Obesity”. [PubMed Abstract] [Full-Text HTML] [Full-Text PDF]. BMJ Open Diabetes Res Care. 2026 Jan 13;13(Suppl 1):e005729. doi: 10.1136/bmjdrc-2025-005729.
All that follows is from the above resource
Abstract
Obesity medications may be part of a comprehensive care plan for adults with obesity. The Obesity Association, a division of the American Diabetes Association (ADA), developed comprehensive, evidence-based guidelines on the pharmacologic treatment of obesity in adults. When used in conjunction with lifestyle modifications, obesity medications have demonstrated efficacy in inducing and sustaining weight reduction while concurrently improving clinical outcomes of obesity and obesity-related diseases and complications. Healthcare professionals should engage people with obesity in a person-centered, shared decision-making approach when selecting an obesity medication to optimize health outcomes while emphasizing individual needs and preferences. The ADA’s Obesity Association encourages healthcare professionals to adopt these guidelines for treatment of obesity in adults.
Keywords: Drug Therapy; Obesity; Pharmacology; Practice Guideline.
© 2026, American Diabetes Association, Inc., and BMJ Publishing Group Limited.
Obesity medications are an essential component of a
comprehensive approach to obesity management, offering significant benefits beyond lifestyle interventions
alone for many people. Research indicates that pharmacotherapy can lead to greater weight reduction and improved weight maintenance than placebo in adults with
obesity (1). Of note, randomized controlled trials (RCTs)
of obesity medications include lifestyle intervention in
both placebo and study drug arms, which most typically comprises a 500-calorie deficit meal plan and behavioral intervention that usually produces 2.6% weight
reduction in the placebo group (2), and the placebo
group receives a placebo pill or injection, as appropriate. In meta-analyses of RCTs, people treated with obesity medications experienced substantial weight
reduction compared with placebo (2,3). Participants
treated with obesity medications also had improvements
in cardiometabolic markers, such as glycemia and lipid profiles (2,3), and some obesity medications demonstrated
improvements in cardiovascular outcomes and other obesity-related diseases (4–7). When used appropriately, obesity medications offer a favorable risk-benefit profile in
many cases, making them a viable option for many people
with obesity.As obesity medications induce clinically meaningful
weight reduction relative to placebo (2,18), the choice of
therapy should prioritize medications most likely to improve obesity-related diseases and complications and to
achieve and maintain weight reduction goals. Therefore,
this section outlines an approach focused on reducing
risk of and burden from obesity-related diseases rather
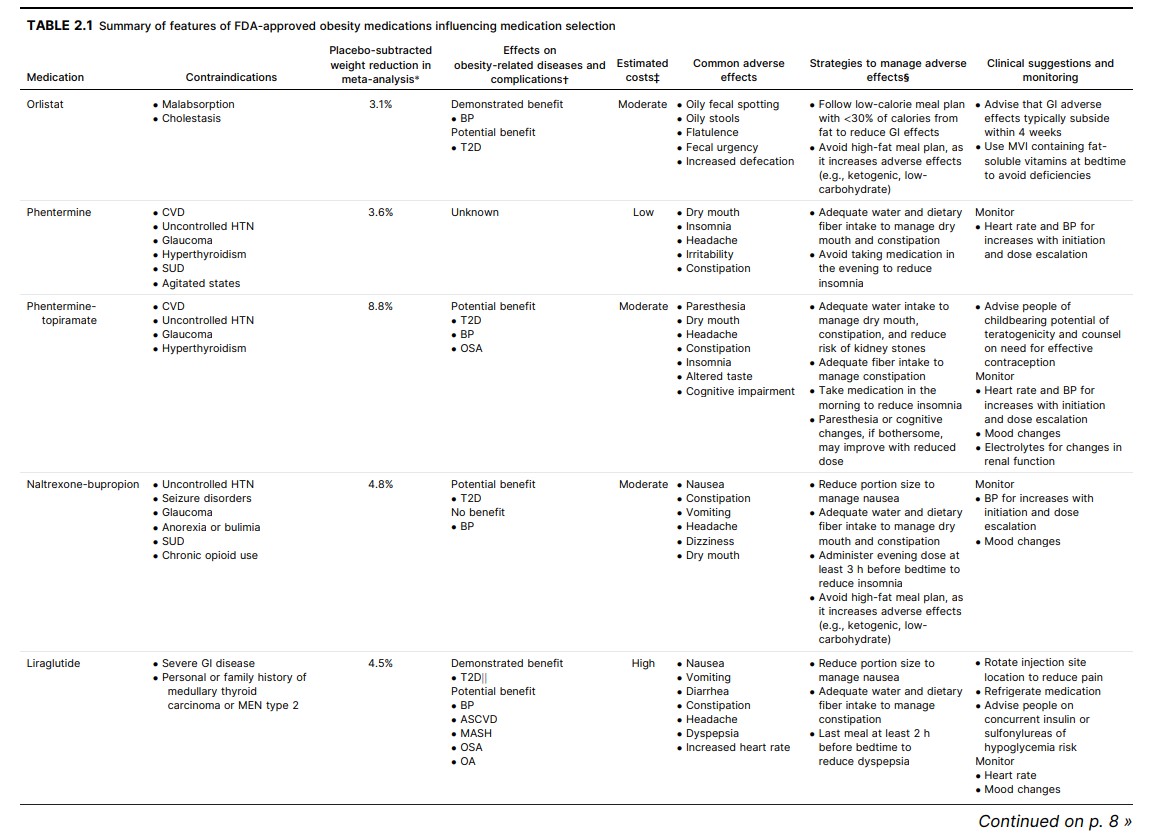
than solely focusing on weight reduction goals. Table 2.1
contains a summary of treatment and weight-reduction
outcomes as well as adverse effects reported in RCTs of
obesity medications. The risk of medication-related adverse effects should be evaluated for each individual, as
this risk may vary depending on their medical history and
concomitant medications. In a network meta-analysis
(132 RCTs), adverse events leading to drug discontinuation were significantly higher for all obesity medications
compared with placebo (2). Health care professionals
should present the potential benefits, risks, and expected
outcomes of obesity medications. Medication selection
should be guided by person-centered factors, including evidence, treatment burden, costs, and individuals’ therapeutic
goals and preferences.Behavioral Lifestyle Therapy With Obesity
MedicationsNutrition, physical activity, and behavioral therapy must be
components of all comprehensive obesity treatment plans,
including those with obesity medications. The efficacy of
obesity treatment interventions that include nutrition and
physical activity have been documented in meta-analyses
(1,19,20), and the addition of obesity medications to lifestyle
interventions improves weight reduction and weight loss
maintenance (1,19,21).
Greater weight reduction may be achieved when a lifestyle
behavioral modification program is administered with an
obesity medication among individuals with obesity (22–25).
For example, in a 1-year RCT, participants in a naltrexonebupropion plus intensive behavioral program group lost
9.3% of baseline weight compared with 5.1% in the placebo
plus intensive behavioral program group (22). The degree of
weight reduction achieved is related to program intensity
(20), and engagement in behavioral components is also associated with greater weight reduction (26). Some obesity
medications are associated with increased fullness as well
as decreased hunger, food preoccupation, dietary disinhibition, and binge eating compared with placebo (27,28),
which may aid individuals in achieving and sustaining the
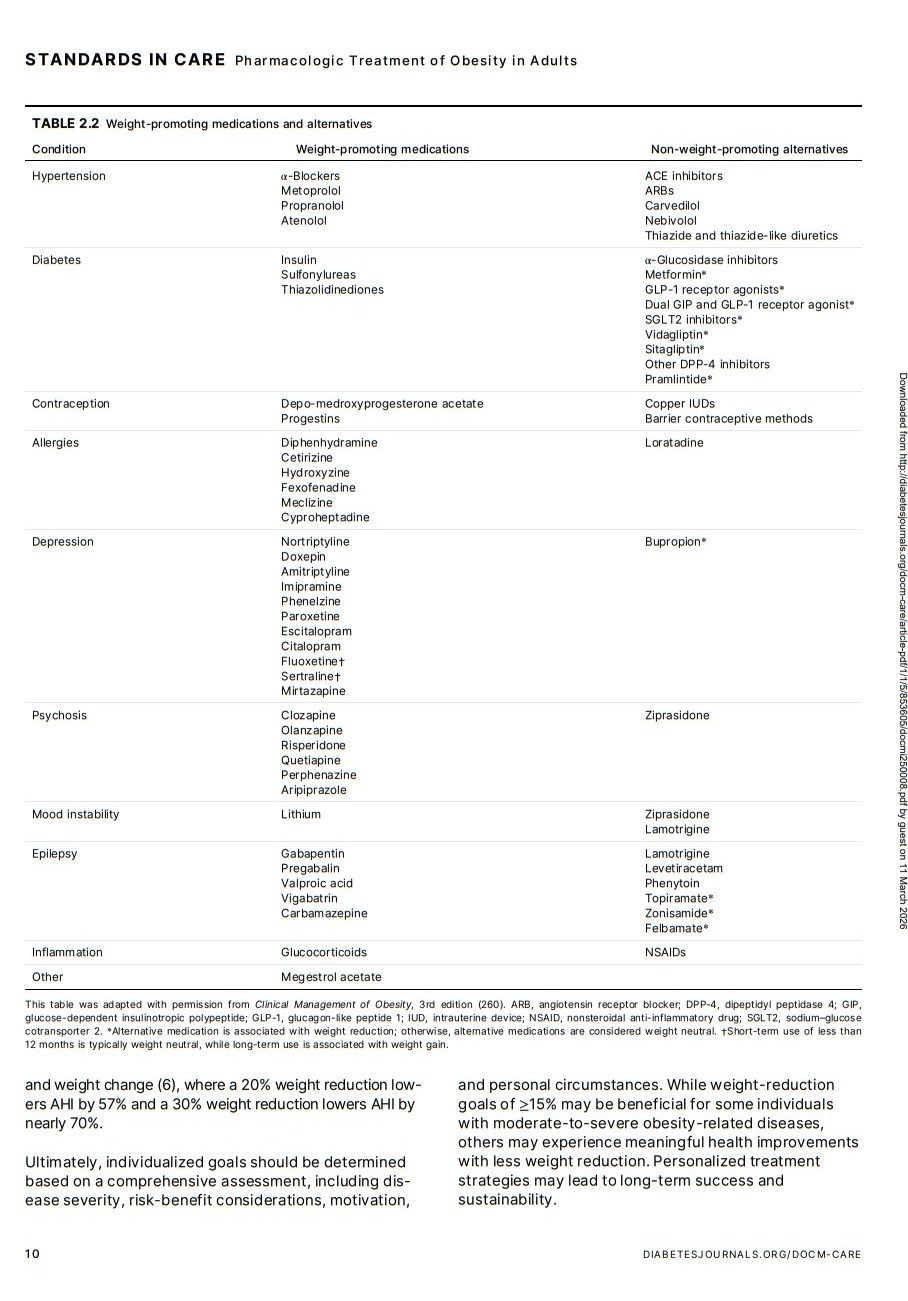
lifestyle treatment plan.Weight-Promoting Medications
Before prescribing an obesity medication, health care professionals should carefully review individuals’ concomitant
medications, as studies have found that medications
that promote weight gain are commonly used, including
among individuals with obesity (29–31). Health care professionals should be aware of weight-promoting medications (Table 2.2) (32). As the number of prescribed
weight-promoting medications increases, individuals’
BMI and waist circumference are more likely to have
greater increases over time (33). In addition, studies
have found that participants using weight-promoting
medications have reduced weight loss efficacy with lifestyle interventions to treat obesity (34,35).Whenever possible and clinically appropriate, avoid initiating
a prescription for weight-promoting medications among
individuals with obesity and instead select a non-weightpromoting alternative. For example, ACE inhibitors, angiotensin receptor blockers, calcium channel blockers, and
diuretics should be prioritized to treat hypertension rather
than some β-blockers, as these options are weight-neutral
rather than weight-promoting. When diabetes medications
are needed for glycemic management, medications most
likely to cause weight gain should be discouraged in favor
of alternatives that are non-weight-promoting (Table 2.2).
However, health care professionals should not refrain from
prescribing weight-promoting medications when needed to
achieve treatment goals for that condition (e.g., blood pressure in hypertension).
For individuals with obesity who may already be using
weight-promoting medications, health care professionals
should determine whether a dose reduction or nonweight-promoting alternative may be clinically appropriate. If so, individuals should be engaged in determining
whether their medication plan should be modified, accounting for the benefits and risks of all treatment options.
Health care professionals should be aware that cessation
of weight-promoting medications does not necessarily result in weight reduction. For individuals who must remain
on weight-promoting medications, obesity medications
may be beneficial in achieving treatment and weight goals.Setting Treatment Goals With Obesity
MedicationsAchieving a ≥5% reduction in baseline body weight for
adults treated with obesity medications is associated with
health benefits (18,38–42). Research demonstrates that a
5% weight reduction improves cardiometabolic risk factors
(2,43,44). For example, the CONQUER RCT demonstrated
that over 60% of participants treated with phenterminetopiramate achieved ≥5% weight reduction as compared with 21% with placebo, and weight reduction was accompanied by improvements in metabolic risk factors (40). In
the SURMOUNT-1 RCT, over 85% of participants who received tirzepatide achieved a ≥5% reduction goal compared with 35% with placebo; notable improvements in
cardiometabolic measures were also observed (18).For individuals with obesity-related diseases and complications, achieving ≥10% reduction in baseline body weight
has consistently demonstrated improvements in these
conditions. In general, 10–15% weight reduction results in
significantly greater improvements in glycemia, blood
pressure, and lipids compared with 5–10% loss (45). In
most cases, the magnitude of weight reduction is directly
proportional to risk factor improvements; therefore, sustained weight reduction of ≥10% is often required for optimal
cardiometabolic benefits. Obesity-related diseases and conditions that benefit from this weight loss magnitude include
metabolic dysfunction–associated steatohepatitis with moderate-to-severe fibrosis (7,46), heart failure with preserved
ejection fraction (47,48), and knee osteoarthritis (49).Emerging data suggest that ≥15% weight reduction may be
needed to achieve clinical benefits in some cases (45), particularly for individuals with moderate-to-severe obesityrelated conditions. In the SURMOUNT-OSA trial, participants with obesity and moderate-to-severe obstructive sleep apnea who received tirzepatide achieved mean weight reduction exceeding 15% and experienced significant reductions
in the apnea-hypopnea index (AHI), hypoxic burden, and
systolic blood pressure compared with placebo (6). A metaanalysis found a dose-response relationship between AHI and weight change (6), where a 20% weight reduction lowers AHI by 57% and a 30% weight reduction lowers AHI by
nearly 70%.Ultimately, individualized goals should be determined
based on a comprehensive assessment, including disease severity, risk-benefit considerations, motivation,
and personal circumstances. While weight-reduction
goals of ≥15% may be beneficial for some individuals
with moderate-to-severe obesity-related diseases,
others may experience meaningful health improvements
with less weight reduction. Personalized treatment
strategies may lead to long-term success and
sustainability.Selecting Obesity Medications
The FDA has approved several obesity medications as adjuncts to a reduced-calorie meal plan and increased physical activity in individuals with obesity or individuals with
overweight and the presence of at least one obesity-related
disease or complication. Historically, label-defined eligibility criteria were BMI ≥30 kg/m2 or ≥27 kg/m2 with one or more
obesity-related disease or complication; however, these
thresholds are no longer included in the labels of several
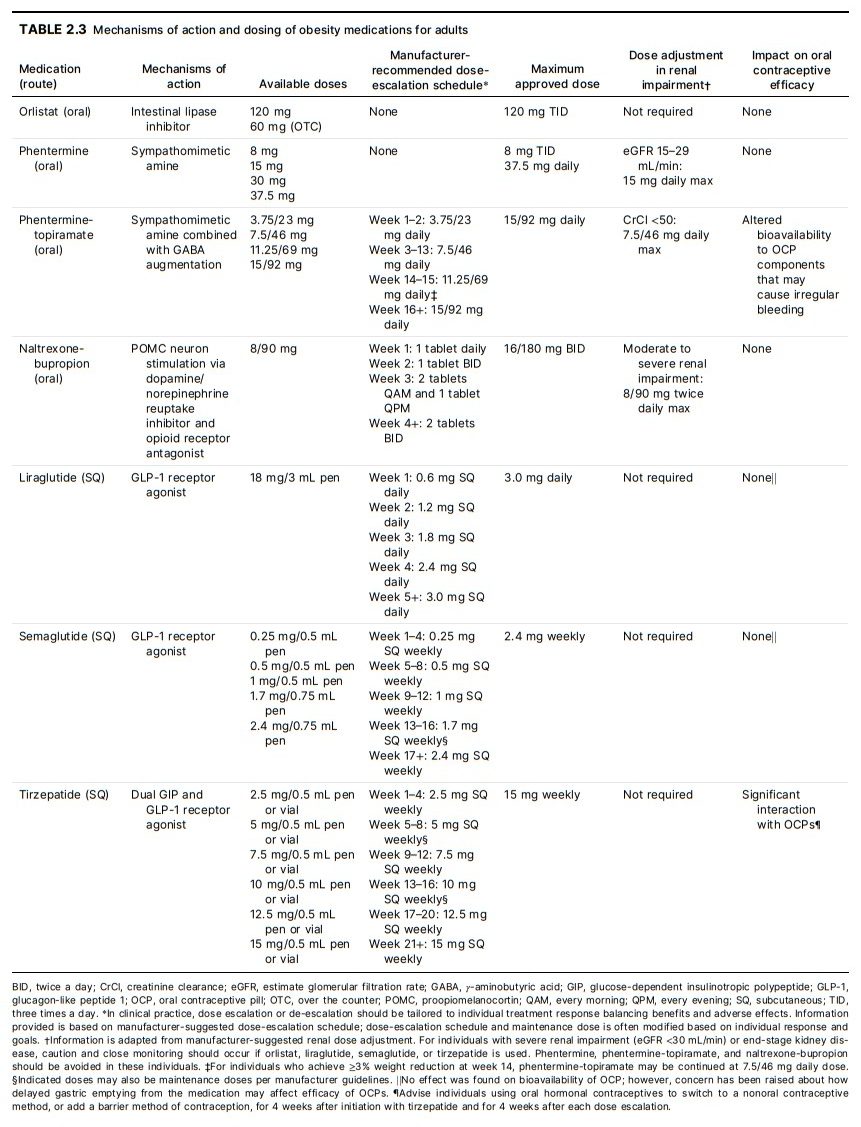
obesity medications (i.e., phentermine-topiramate, liraglutide, semaglutide, and tirzepatide). Table 2.3 describes
the mechanisms of action and dosing for all FDAapproved obesity medications. Most obesity medications are approved for long-term treatment; the
exception is monotherapy with phentermine or other
adrenergic agents (e.g., diethylpropion) that were approved for short-term use. Obesity medications are invaluable components of initial treatment in adults who
already have or who are at high risk of developing obesity-related diseases or complications, as they can significantly mitigate the progression of these conditions
(4,6,47,48). If individuals with obesity-related diseases
or complications initially decline obesity medication, it
may be appropriate to reevaluate their interest in this
treatment option at follow-up.All FDA-approved obesity medications have demonstrated
weight-reduction efficacy in clinical trials (18,38–42,50). In
a network meta-analysis, in comparison with placebo at
study end point, orlistat was associated with 3.1% greater
weight reduction (22 RCTs), phentermine-topiramate was
associated with 8.8% greater weight reduction (5 RCTs),
naltrexone-bupropion was associated with 4.8% greater
weight reduction (2 RCTs), liraglutide was associated with
4.5% greater weight reduction (10 RCTs), semaglutide was
associated with 11.9% greater weight reduction (13 RCTs),
and tirzepatide was associated with 16.2% greater weight
reduction (6 RCTs) (51). Of note, these meta-analyses include RCTs of adults with obesity who do and do not have
obesity-related diseases and complications, and other
network meta-analyses have reported similar results (2,3)To date, phentermine has only been studied in shortterm RCTs (28 weeks or less) (50,52–54); however, a
2019 observational study demonstrated that long-term
phentermine use was associated with significant weight
reduction: individuals prescribed the medication for
≥12 months experienced a 7.4% greater weight reduction
compared with those with ≤3 months (55). This analysis
also showed no significant increase in the risks of cardiovascular disease or death among individuals prescribed
phentermine long term. In individuals without cardiovascular disease, serious psychiatric disease, or substance
use disorder, phentermine monotherapy may be an effective and safe option for achieving sustained weight
reduction, with the caveat that no long-term efficacy
and safety trial data currently exist (56,57). Long-term
phentermine monotherapy is an off-label use, as this
medication is only FDA-approved for short-term use,
and health care professionals should be aware that some
geographic areas may legally restrict this approach.
Long-term phentermine monotherapy may be considered
after discussion of the benefits and risks of all obesitymedications, including those FDA-approved for longterm use. Phentermine is a schedule IV controlled substance, and therefore health care professionals should
perform an in-person examination and evaluation, preferably with electrocardiogram, prior to prescribing this
medication. The starting dose of phentermine should be
15 mg daily or less, and the dose may be increased based
on tolerability and treatment response. Health care professionals should consider monthly follow-up, including
monitoring of blood pressure and pulse, until the phentermine dose is stable, with follow-up every 3–6
months for long-term treatment to maintain health goals.
Phentermine is one of the most prescribed obesity medications in the U.S. (58).Prior research has shown that adverse effects from obesity
medications contribute to individuals discontinuing the
medication (2,59). Therefore, health care professionals
should advise individuals of the adverse effects associated
with each obesity medication and discuss strategies tominimize or manage these effects (Table 2.1). For example,
glucagon-like peptide 1 receptor agonists (GLP-1RAs) and
a dual glucose-dependent insulinotropic polypeptide and
glucagon-like peptide 1 receptor agonist (dual GIP/GLP-1RA)
are associated with gastrointestinal adverse effects including nausea, diarrhea, constipation, and dyspepsia (60–62),
which may be managed by decreasing portion sizes, decreasing intake of high-fat foods, increasing intake of
high-fiber foods, avoiding long intervals between meals, and
eating the last meal ≥2 h before bedtime (63,64).For an individual with obesity who does not have any
obesity-related diseases or complications, the primary
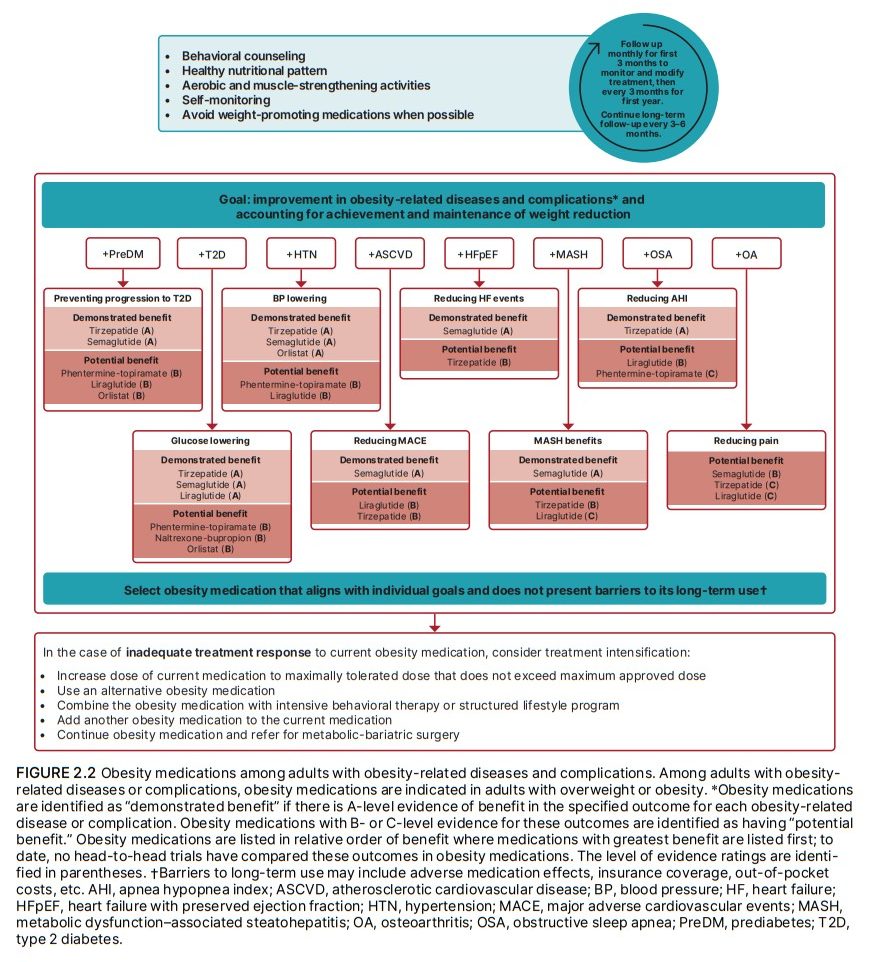
treatment goal is to prevent the development of these conditions (Fig. 2.1). Health care professionals should
perform a comprehensive medical evaluation before selecting a medication to ensure safety and efficacy. Pharmacotherapy is an important tool for managing obesity,
particularly when lifestyle interventions alone are insufficient to achieve and sustain weight reduction (53). The
amount of weight reduction necessary to prevent obesity-related diseases and complications varies from person to person, and the health care professional should
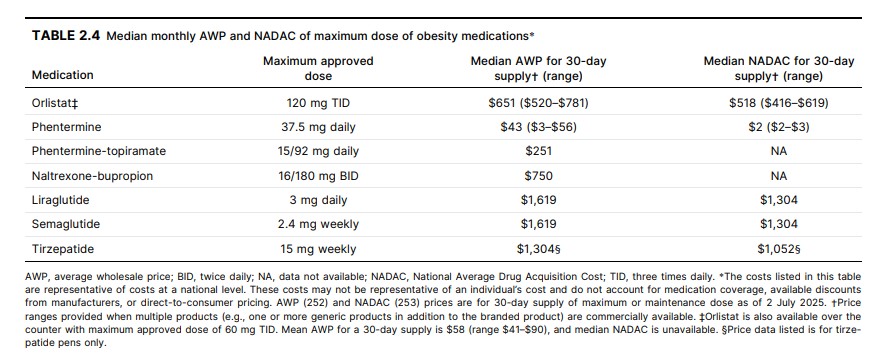
consider cost, access, tolerability, and individual preferences in the decision-making. Current costs of each obesity
medication are summarized in Table 2.4.Prediabetes
In individuals with obesity, weight reduction is highly effective in preventing or delaying progression to type 2
diabetes (T2D), particularly in high-risk people with prediabetes or metabolic syndrome. T2D prevention is dependent on weight-reduction magnitude, as evident from
the Diabetes Prevention Program that demonstrated
near-maximal T2D prevention was observed at ∼10%
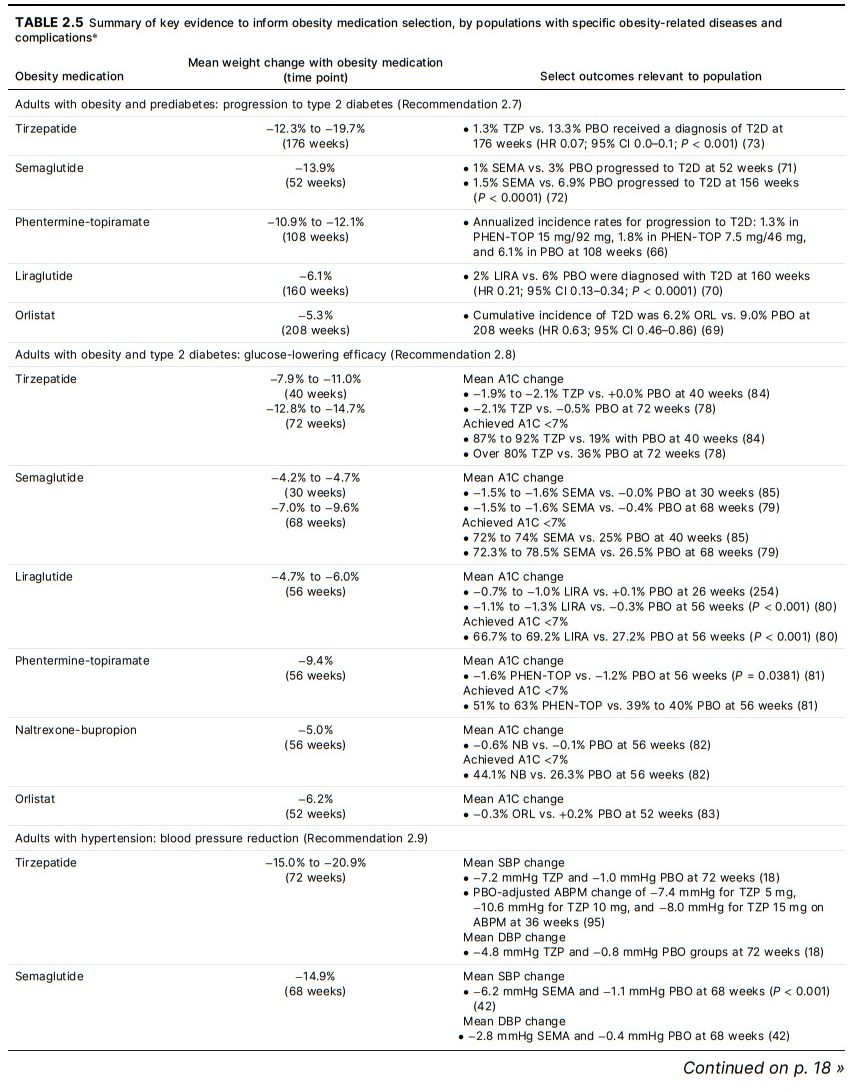
weight reduction with lifestyle intervention (65). Figure
2.2 and Table 2.5 summarize outcomes of obesity medications among individuals with prediabetes. For example,
an RCT employing phentermine-topiramate achieved
10% weight reduction and reduced incident T2D by 79%
(66), which is a relatively similar T2D reduction to that
achieved with metabolic-bariatric surgery (67,68). Orlistat also reduces incident T2D by 37% (69).More recent studies with GLP-1RA obesity medications
are consistent with a weight-reduction goal of ∼10% to
prevent T2D progression. An RCT of liraglutide 3 mg produced 6.1% weight reduction and reduced T2D progression by 79% (70). Semaglutide 2.4 mg was highly
effective in converting prediabetes to normoglycemia
(71), and 9.7% weight reduction with semaglutide was
associated with 73% reduction in risk of developing T2D
compared with placebo over ∼4 years of observation in
the SELECT RCT (72). Tirzepatide 15 mg led to a 93%
T2D reduction associated with 19.7% weight reduction in
the 3-year SURMOUNT-1 extension study (73); this T2D
prevention benefit may reflect incretin effects of the
GLP-1 component and GIP bioeffects in addition to
weight reduction (74,75).Type 2 Diabetes
While the relationship between obesity and pathogenesis
of T2D is complex, weight reduction represents highly effective therapy for both glycemic management and amelioration of obesity-related diseases and complications.
The value of weight reduction as a primary treatment approach in T2D, whether at initial diagnosis or in conjunction with glucose-lowering therapy at any time over the
disease course, is emphasized in the ADA’s “Standards of
Care in Diabetes” (76). Health care professionals should
be aware that people with T2D typically lose less weight than individuals without this diagnosis (77). All obesity
medications FDA-approved for long-term treatment
have been studied in RCTs that enrolled people with T2D
(Fig. 2.2 and Table 2.5) (2,78–83). Weight reduction with
obesity medication consistently lowers A1C and reduces
the need for diabetes medications when compared with
lifestyle alone (78–84). With obesity medications, individuals with T2D and obesity also experience blood pressure reductions, improvements in lipids, decreased
hepatic transaminases, and improvements in cardiovascular risk biomarkers. Three medications approved for
obesity (i.e., liraglutide, semaglutide, and tirzepatide)
also have an indication for glycemic management
(84–86), although maximum approved doses for T2D are
lower for liraglutide and semaglutide than approved
doses for obesity.Given their weight reduction and glucose-lowering benefits, health care professionals should strongly consider a
GLP-1RA or dual GIP/GLP-1RA obesity medication as part
of the treatment plan in people with obesity and T2D. In
the SURMOUNT-2 RCT involving people with T2D and
obesity, tirzepatide 15 mg produced weight reduction of
14.7% and resulted in a 2.1% A1C reduction, with nearly
half of people achieving normal A1C values (<5.7%) (78).
In the STEP 2 RCT, semaglutide 2.4 mg resulted in a 9.6%
weight reduction and A1C lowering of 1.6% (79). The
SCALE Diabetes trial in people with obesity and T2D
demonstrated that liraglutide 3 mg produced a 6.0%
weight reduction and 1.3% A1C reduction (80). Severe
hypoglycemic episodes were uncommon in individuals
randomized to tirzepatide, semaglutide, and liraglutide in
these trials (78–80).If a GLP-1RA or dual GIP/GLP-1RA obesity medication
cannot be used, then other obesity medications may be
considered. In people with T2D and obesity, phenterminetopiramate 15/92 mg resulted in 9.4% weight reduction and
A1C lowering of 1.6% (81). Naltrexone-bupropion 32/360
mg led to a 5% weight reduction in people with obesity and
T2D and A1C lowering of 0.6% (82). Orlistat resulted in 6.2%
weight reduction and 0.3% A1C decrease in this population
(83).Hypertension
Hypertension is an obesity-related disease (87,88), and
one of the benefits of weight reduction is lowering blood
pressure. Greater weight reduction leads to greater improvements in systolic and diastolic blood pressure with
intensive lifestyle intervention (45). Blood pressure lowering
in individuals with and without hypertension is observed
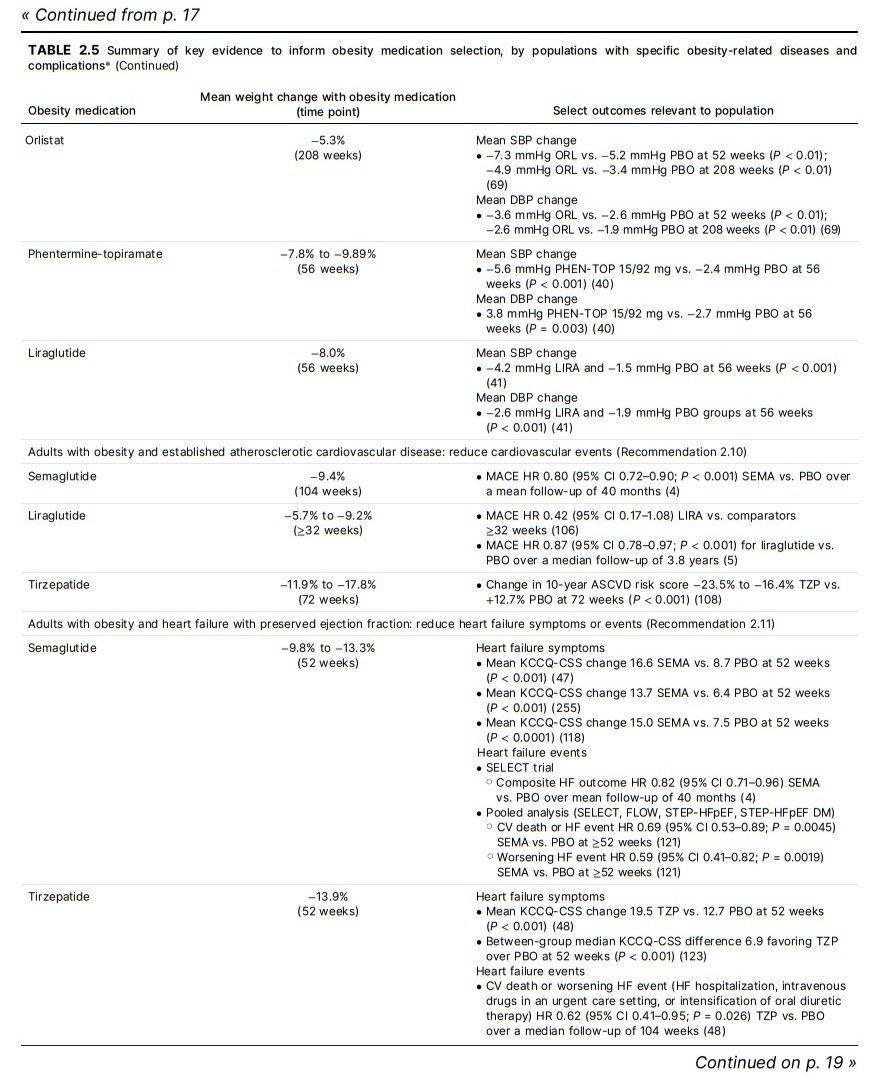
with most obesity medications (Fig. 2.2 and Table 2.5)
(18,41,42,89–94). For example, tirzepatide 15 mg reduced
systolic and diastolic blood pressure by 7.2 mmHg and
4.8 mmHg, respectively (18), and a substudy using 24-h
ambulatory monitoring found that tirzepatide substantially
reduced daytime and nighttime blood pressures in individuals with elevated baseline systolic pressure (95). It shouldbe noted that tirzepatide, semaglutide, liraglutide, and
phentermine-topiramate, on average, cause small increases
in resting heart rate (∼2–4 bpm), although some individuals
may experience larger increases (∼10–20 bpm). This heart
rate change is not clinically detrimental to most individuals,
as the rate-pressure product (heart rate × systolic blood
pressure) (96–98), which indicates workload on the heart,
remains favorable due to blood pressure reductions. Health
care professionals should be aware that naltrexonebupropion does not reduce blood pressure commensurate
with weight reduction (89), and both naltrexone-bupropion
and phentermine are contraindicated in individuals with uncontrolled hypertension.Atherosclerotic Cardiovascular Disease
Obesity contributes to atherosclerotic cardiovascular
disease (ASCVD) development through multiple mechanisms (99), and the duration of obesity and visceral adiposity are strong predictors of coronary artery disease in
epidemiologic studies (100). The magnitude of weight reduction achieved may be particularly critical for ASCVD
(99), as modest weight reduction with intensive lifestyle
intervention did not reduce the rate of cardiovascular
events in adults with T2D and obesity (101); a post hoc
analysis of this RCT found that only participants who
achieved ≥10% weight reduction had significant reductions in cardiovascular events (102). In adults with obesity and established ASCVD, the treatment plan should
include a GLP-1RA with demonstrated benefits or dual
GIP/GLP-1RA with potential benefits in reducing cardiovascular events as well as weight reduction (Fig. 2.2 and
Table 2.5).Semaglutide reduces cardiovascular events among highrisk individuals (e.g., those with prior ASCVD or T2D)
(103). In the SELECT RCT, semaglutide 2.4 mg resulted in
a 20% reduction in adverse cardiovascular outcomes
(cardiovascular death, nonfatal myocardial infarction, or
nonfatal stroke) in adults with BMI ≥27 kg/m2 and established cardiovascular disease without diabetes (4).
Further, semaglutide produced durable weight reduction
(104) and significantly reduced new-onset diabetes and
a composite kidney outcome (driven by reductions in
new-onset macroalbuminuria) (105). While a cardiovascular outcomes trial evaluating liraglutide 3.0 mg has not
been performed, liraglutide 1.8 mg (T2D-approved dose)
reduces cardiovascular events in people with T2D at increased cardiovascular risk, including among participants
with obesity (5). Similar findings were observed with liraglutide 3.0 mg versus pooled comparators in the SCALE
trials (HR 0.42; 95% CI 0.17–1.08) (106), but this analysis
was underpowered due to low event rates.A cardiovascular outcomes trial of dual GIP/GLP-1RA
obesity medication has not yet been published; however,
there is evidence to support potential ASCVD benefits.
Tirzepatide has been shown to significantly improve cardiovascular risk factors (e.g., blood pressure, cholesterol,
systemic inflammation, and glycemic management) in
people with obesity (18,107). In a SURMOUNT-1 trial post
hoc analysis, tirzepatide significantly reduced the predicted 10-year risk of ASCVD events compared with placebo (108). Moreover, in a prespecified participant-level
pooled analysis of the SURPASS program, tirzepatide did
not increase the risk of major adverse cardiovascular
events in people with T2D versus pooled comparators
(HR 0.80; 95% CI 0.57–1.11) (109).The LIGHT trial, which randomized people with BMI
27–50 kg/m2 at increased cardiovascular risk to naltrexone-bupropion, was designed to evaluate cardiovascular
outcomes. In the primary analysis (conducted after 50%
of planned events), naltrexone-bupropion did not reduce
major adverse cardiovascular events compared with placebo (110). However, due to unplanned early termination
after public release of confidential interim data, the
LIGHT trial was unable to rigorously establish the safety
and efficacy of naltrexone-bupropion compared with placebo. Moreover, most participants permanentlydiscontinued study medication by 1-year post-randomization. There are no cardiovascular outcomes trials for
orlistat, phentermine, or phentermine-topiramate.Heart Failure With Preserved Ejection Fraction
Obesity is a leading driver of heart failure with preserved
ejection fraction (HFpEF) onset and progression
(111–115), and individuals with obesity and HFpEF display
unique clinicopathological features (e.g., increased
plasma volume and greater cardiac remodeling) when
compared with individuals with HFpEF without obesity
(116). Modest weight reduction among individuals with
obesity and HFpEF through lifestyle change improves
aerobic capacity, New York Heart Association functional
class, and quality of life (117). In adults with obesity and
HFpEF, the treatment plan should include a GLP-1RA or
dual GIP/GLP-1RA with demonstrated improvements in
heart failure (HF)-related symptoms or events (Fig. 2.2
and Table 2.5).In the STEP-HFpEF RCT, semaglutide 2.4 mg significantly
improved HF-related symptoms and functional limitations
and reduced body weight (11% placebo-subtracted reduction) in people with BMI ≥30 kg/m2 and chronic symptomatic HF with left ventricular ejection fraction (LVEF)
≥45% (46). In pooled analyses, semaglutide improved
exercise function and natriuretic peptide levels (118), reduced diuretic requirements (119), and attenuated adverse myocardial remodeling (120). In a prespecified
pooled analysis of the STEP-HFpEF Program, SELECT,
and FLOW trials, semaglutide reduced cardiovascular
death or HF events by 31% compared with placebo in
participants with HF and LVEF ≥40% (121).Tirzepatide has also been shown to reduce HF events in
people with obesity and stable chronic HFpEF. Among individuals with BMI ≥30 kg/m2 and HF with LVEF ≥50% enrolled in the SUMMIT RCT, tirzepatide improved HF-related
health status and reduced the rate of cardiovascular death
or a worsening HF event by 38% (48), although number of
events was relatively low (36 with tirzepatide and 56 with
placebo). Additional benefits on body weight (12% placebo-controlled reduction), exercise function, left ventricular mass, and paracardiac adipose tissue were also
observed (122,123).Metabolic Dysfunction–Associated Steatohepatitis
Metabolic dysfunction–associated steatohepatitis
(MASH) is defined as the presence of steatohepatitis and
no alcohol consumption or consumption in amounts unlikely to directly cause adverse liver outcomes (alcohol
intake <20 g/day for women and <30 g/day for men)
(124). Fibrosis is a predictor of disease progression (125)
and is strongly linked to liver-related outcomes and death
(126). Histologically, fibrosis is staged based on severity
and distribution of scar tissue. Clinically significant fibrosis is defined as stage ≥F2 (moderate; sinusoidal and
portal fibrosis), advanced fibrosis is F3 (i.e., bridging fibrosis, usually central-to-portal or central-to-central
bridges), and F4 is cirrhosis (124). Among people with
MASH, weight reduction of ≥10% may be needed to reverse steatohepatitis and improve fibrosis (127). In adults
with obesity and MASH with moderate or advanced
fibrosis, a GLP-1RA or dual GIP/GLP-1RA is preferred for
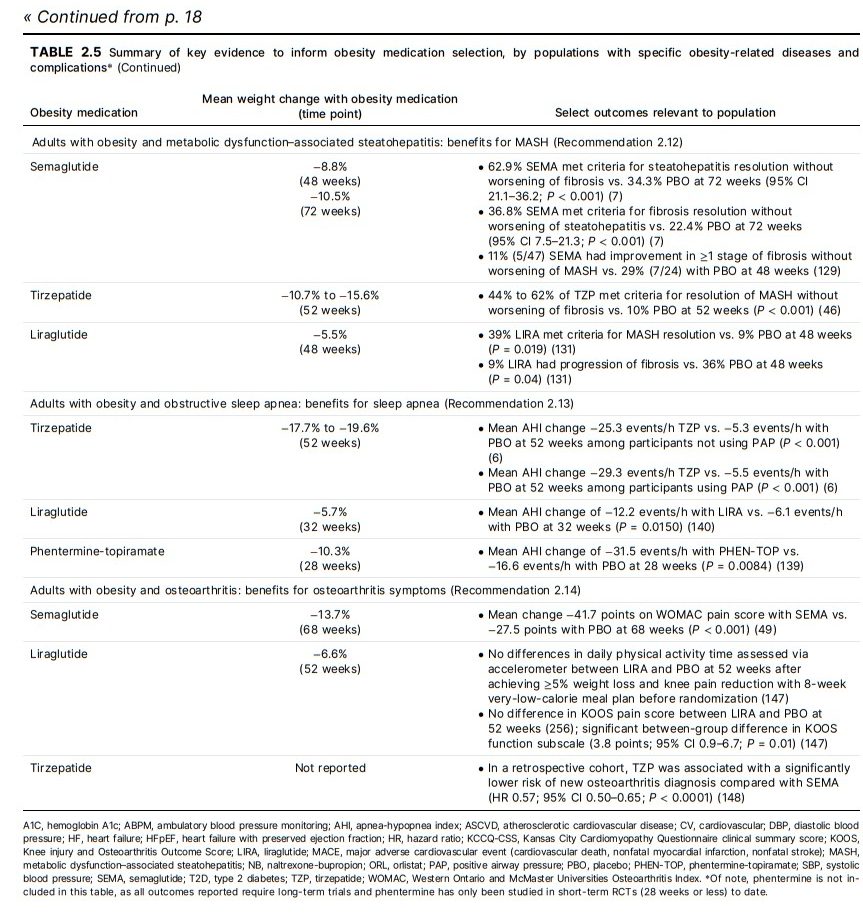
obesity treatment because of the benefits or potential
benefits on MASH (Fig. 2.2 and Table 2.5). While not specifically tested among individuals with MASH, health care
professionals should be aware that the dose of naltrexone-bupropion needs to be adjusted in the setting of
moderate hepatic impairment.Obstructive Sleep Apnea
Obstructive sleep apnea (OSA) is an obesity-related disease
characterized by partial or complete upper airway obstruction during sleep, leading to sleep fragmentation and hypoxemia (114). The prevalence of moderate-to-severe OSA,
defined as AHI ≥15 events/h on polysomnography, increases with increasing body weight (134). OSA is associated with increased mortality in people with obesity
(135,136). A 10% body weight reduction is a ssociated with a
26% lower AHI (134). Weight reduction with intensive lifestyle intervention significantly decreases AHI and increases
the likelihood of OSA remission (137,138).Several obesity medications improve OSA in RCTs (Fig. 2.2
and Table 2.5) (6,139,140), and AHI reduction is strongly related to the weight-reduction magnitude achieved with
the obesity medication (139,140). To date, the greatest
magnitude of benefit has been with tirzepatide. . . . In addition to established OSA treatment approaches (e.g., PAP,
airway stimulation, and splinting devices), the evidence supports obesity medications as part of the treatment plan for
people with obesity and OSA.Osteoarthritis
Osteoarthritis is an obesity-related disease, particularly
knee osteoarthritis (143,144). Weight gains of ≥10% are
associated with worsening pain and physical function
(145), and weight reduction of similar magnitude may
result in symptom improvements (146). In adults with
obesity and moderate osteoarthritis, the treatment plan
should prioritize a GLP-1RA or dual GIP/GLP-1RA with potential to improve osteoarthritis symptoms (Fig. 2.2 and
Table 2.5). . . . A retrospective cohort
study found that individuals prescribed semaglutide, liraglutide, or tirzepatide had a reduced risk of osteoarthritis
diagnosis (148). Individuals prescribed tirzepatide had a
significantly lower risk of osteoarthritis compared with individuals prescribed semaglutide or liraglutide.Long-term Management of Obesity Medications
Start here.