The following are excerpts from Influenza Antiviral Medications: Summary for Clinicians from the Centers For Disease Control and Prevention (CDC) [Be sure and review the whole page]:
Antiviral medications with activity against influenza viruses are an important adjunct to influenza vaccine in the control of influenza.
Influenza antiviral prescription drugs can be used to treat influenza or to prevent influenza.
Do not use amantadine or ramitidine as they don’t work against the current Influenza A viruses (99% resistance) and they do not work against Influenza B.
Antiviral resistance to oseltamivir, zanamivir, and peramivir among circulating influenza viruses is currently low, but this can change. Also, antiviral resistance can emerge during or after treatment in some patients (e.g., immunocompromised).
For information about antiviral drug resistance to influenza viruses and guidance on the use of influenza antiviral medications when antiviral resistance is suspected or documented this season, see Antiviral Drug-Resistance among Influenza Viruses.
For weekly surveillance data on antiviral resistance this season, see the FluView U.S. Influenza Surveillance Report.
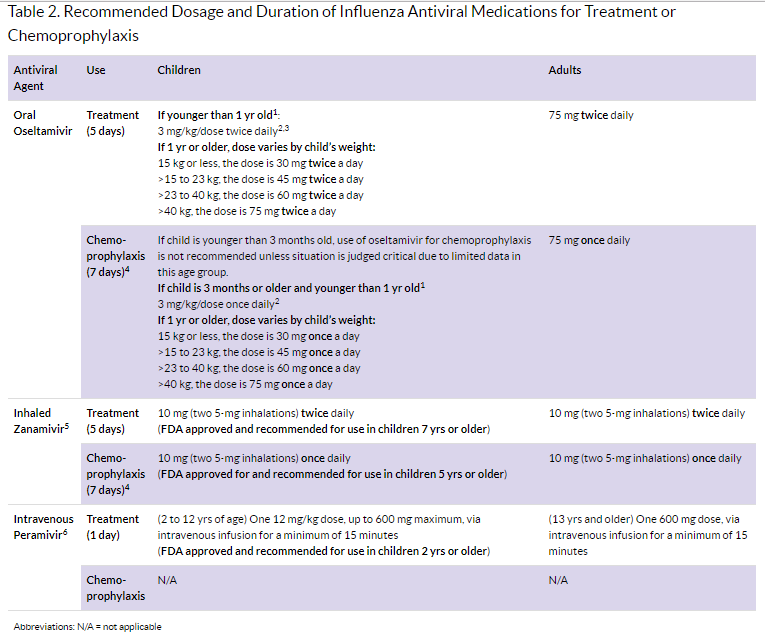
Duration of Treatment or Chemoprophylaxis
Treatment: Recommended duration for antiviral treatment is 5 days for oral oseltamivir or inhaled zanamivir. For the treatment of uncomplicated influenza with intravenous peramivir, a single dose is recommended. Longer daily dosing (oral oseltamivir or intravenous peramivir) can be considered for patients who remain severely ill after 5 days of treatment.
Chemo prophylaxis: Recommended duration is 7 days (after last known exposure). For control of outbreaks in institutional settings (e.g. long-term care facilities for elderly persons and children) and hospitals, CDC recommends antiviral chemoprophylaxis for a minimum of 2 weeks, and continuing up to 1 week after the last known case was identified. Antiviral chemoprophylaxis is recommended for all residents, including those who have received influenza vaccination.
Chemoprophylaxis
- Annual influenza vaccination is the best way to prevent influenza because vaccination can be given well before influenza virus exposures occur, and can provide safe and effective immunity throughout the influenza season.
- Antiviral medications are approximately 70% to 90% effective in preventing influenza and are useful adjuncts to influenza vaccination.
- CDC does not recommend widespread or routine use of antiviral medications for chemoprophylaxis except as one of multiple interventions to control institutional influenza outbreaks. Routine use of post-exposure chemoprophylaxis is not recommended; one reason for this is to avoid sub-therapeutic treatment dosing if infection is already established, although the possibility of whether antiviral resistant viruses could emerge is unknown.
- In general, CDC does not recommend seasonal or pre-exposure antiviral chemoprophylaxis, but antiviral medications can be considered for chemoprophylaxis to prevent influenza in certain situations, such as the following examples:
- Prevention of influenza in persons at high risk of influenza complications during the first two weeks following vaccination after exposure to a person with influenza.
- Prevention for people at high risk for complications from influenza who cannot receive influenza vaccine due to a contraindication after exposure to a person with influenza.
- Prevention for people with severe immune deficiencies or others who might not respond to influenza vaccination, such as persons receiving immunosuppressive medications, after exposure to a person with influenza.
- An emphasis on close monitoring and early initiation of antiviral treatment if fever and/or respiratory symptoms develop is an alternative to chemoprophylaxis after a suspected exposure for some persons.
- To be effective as chemoprophylaxis, an antiviral medication must be taken each day for the duration of potential exposure to a person with influenza and continued for 7 days after the last known exposure. For persons taking antiviral chemoprophylaxis after inactivated influenza vaccination, the recommended duration is until immunity after vaccination develops (antibody development after vaccination takes about two weeks in adults and can take longer in children depending on age and vaccination history).
- Antiviral chemoprophylaxis generally is not recommended if more than 48 hours have elapsed since the first exposure to a person with influenza.
- Patients receiving antiviral chemoprophylaxis should be encouraged to seek medical evaluation as soon as they develop a febrile respiratory illness that might indicate influenza.
Dosing in Adult Patients with Renal Impairment
Dose adjustment of oseltamivir is recommended for patients with creatinine clearance between 10 and 60 mL/min and patients with end-stage renal disease (ESRD) undergoing hemodialysis or continuous peritoneal dialysis receiving oseltamivir for the treatment or chemoprophylaxis of influenza. Oseltamivir is not recommended for patients with ESRD not undergoing dialysis. The recommended doses are detailed in Table 3; duration of treatment and chemoprophylaxis is the same as recommended for patients with normal renal function. No dose adjustment is recommended for inhaled zanamivir for a 5-day course of treatment for patients with renal impairment. The dose of intravenous peramivir should be reduced for patients with baseline creatinine clearance below 50 mL/min (see Table 3 on the web page below).
Resources:
(1) Influenza Antiviral Medications: Summary for Clinicians from the Centers For Disease Control and Prevention (CDC)