In this post, I link to and excerpt from Long COVID in Children and Adolescents: A Systematic Review and Meta-analyses [Full-Text HTML] [Full-Text PDF]. Posted March 13, 2022.
This article is a preprint and has not been peer-reviewed [what does this mean?]. It reports new medical research that has yet to be evaluated and so should not be used to guide clinical practice.
All that follows is from the above article.
ABSTRACT
Objective To estimate the prevalence of long COVID in children and adolescents and identify the full spectrum of signs and symptoms present after acute SARS-CoV-2 infection.
Methods Two independent investigators searched PubMed and Embase in order to identify observational studies that met the following criteria: 1) a minimum of 30 patients, 2) ages ranged from 0 to 18 years, 3) published in English, 4) published before February 10th, 2022, and 5) meets the National Institute for Healthcare Excellence (NICE) definition of long COVID, which consists of both ongoing (4 to 12 weeks) and post-COVID-19 (≥12 weeks) symptoms. For COVID symptoms reported in two or more studies, random-effects meta-analyses were performed using the MetaXL software to estimate the pooled prevalence, and Review Manager (RevMan) software 5.4 was utilized to estimate the Odds Ratios (ORs) with a 95% confidence interval (CI). Heterogeneity was assessed using I2 statistics. The Preferred Reporting Items for Systematic Reviewers and Meta-analysis (PRISMA) reporting guideline was followed (registration PROSPERO CRD42021275408).
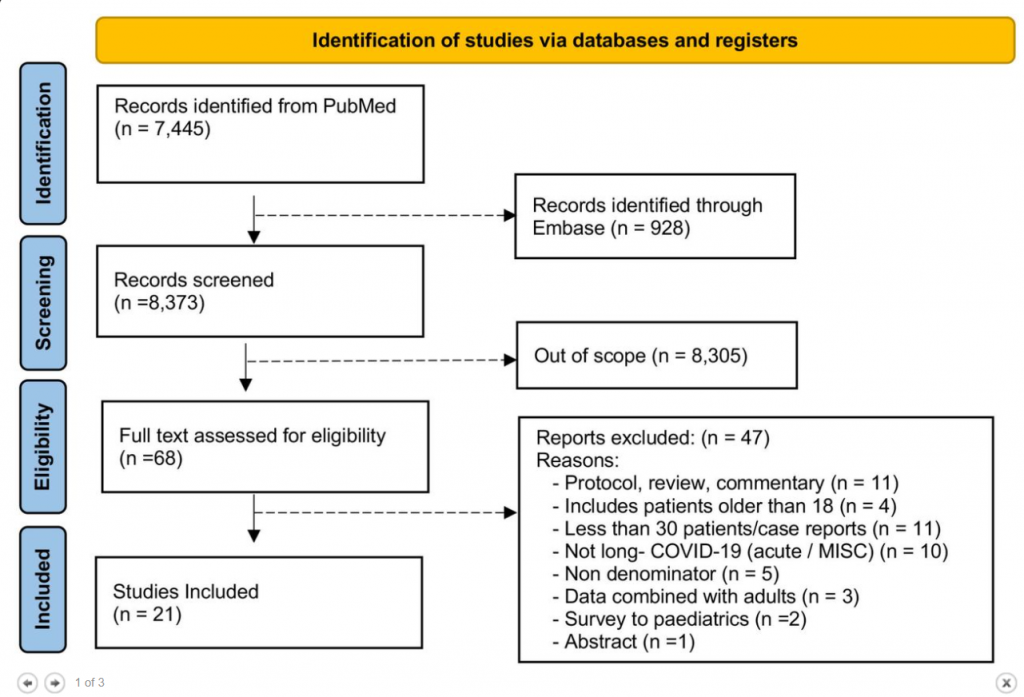
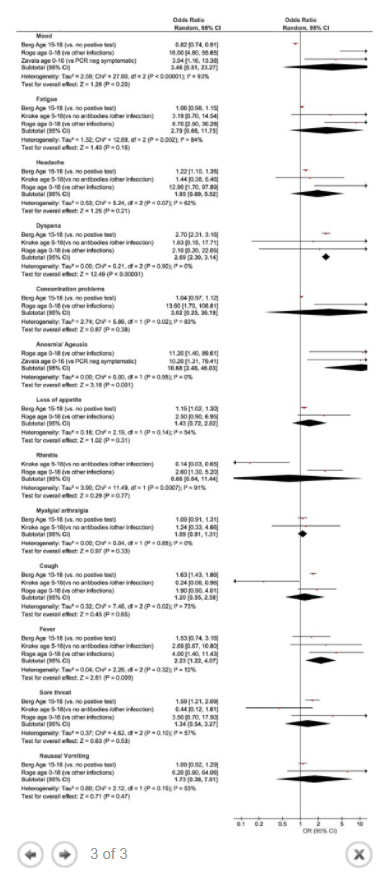
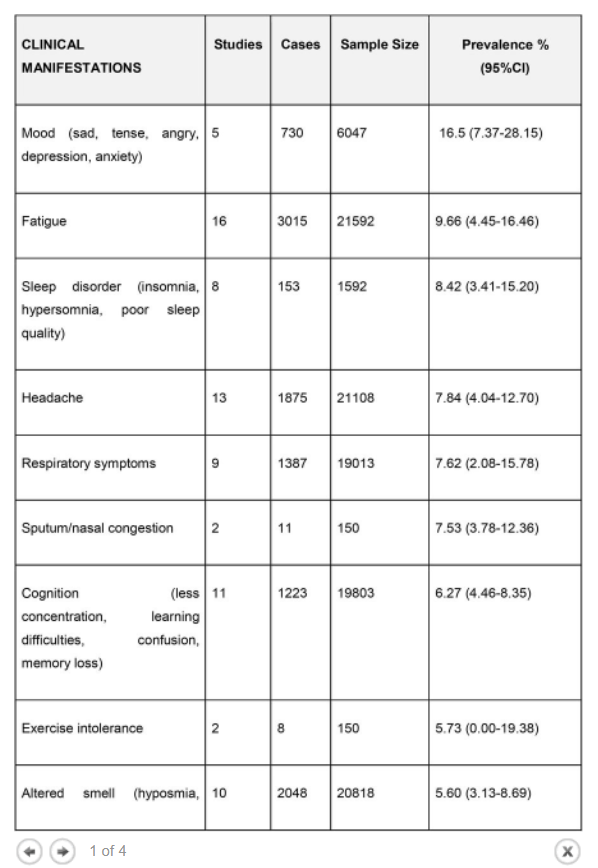
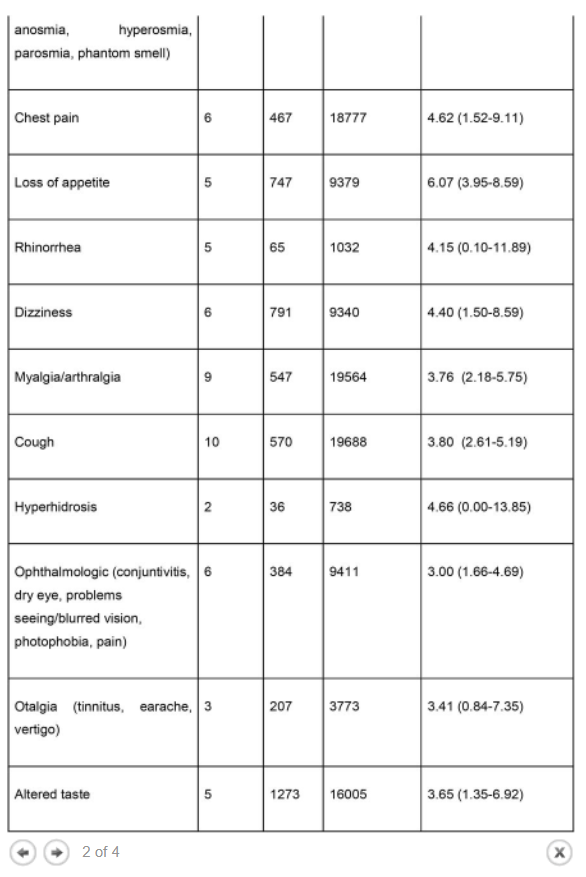
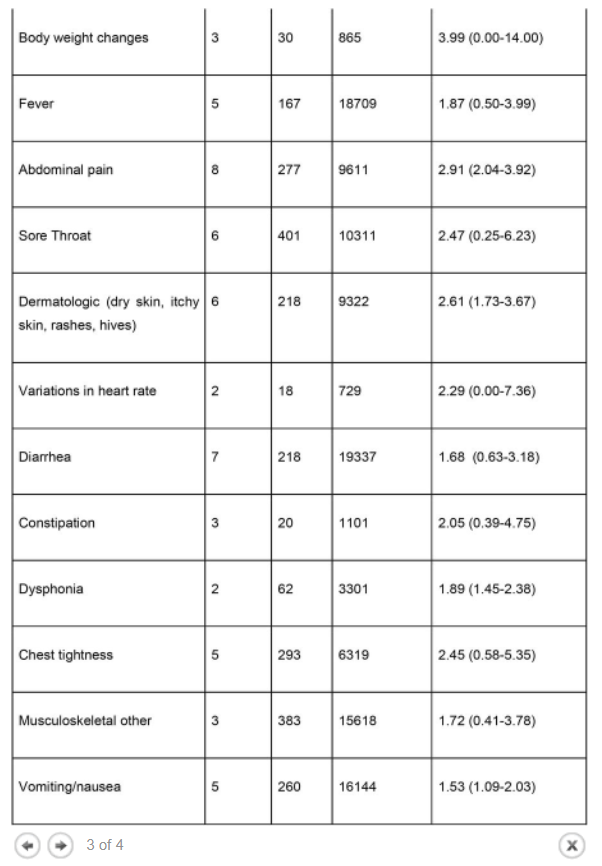
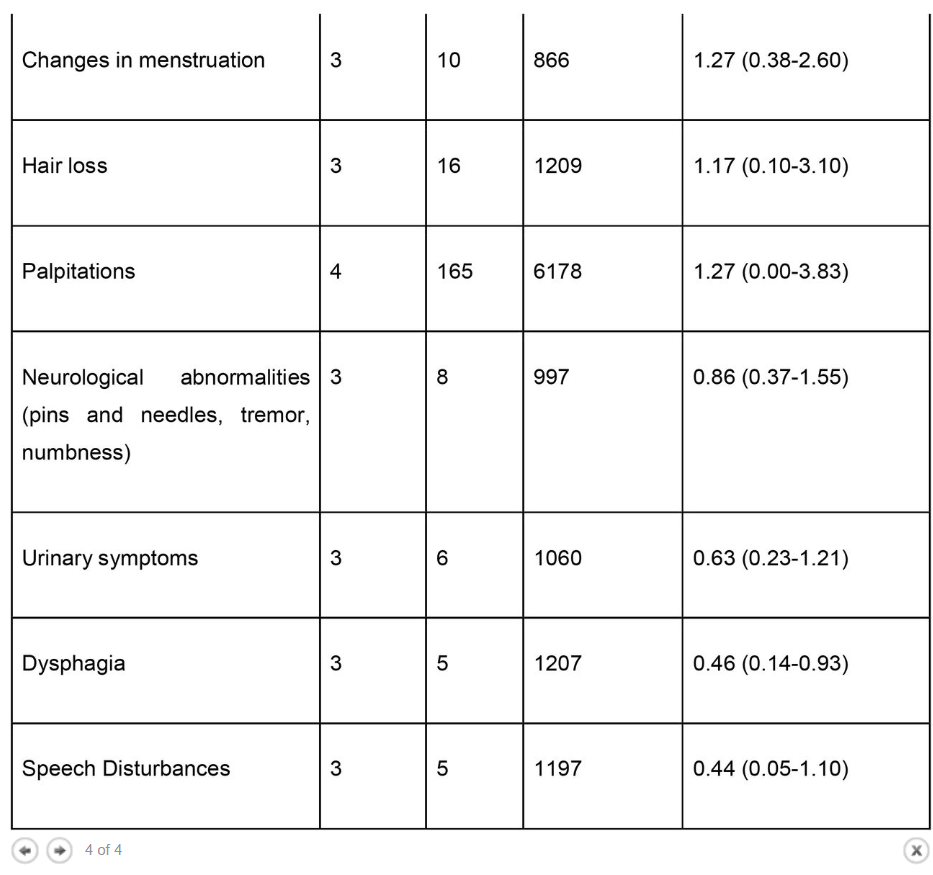
Results The literature search yielded 68 articles for long COVID in children and adolescents. After screening, 21 studies met the inclusion criteria and were included in the systematic review and meta-analyses. A total of 80,071 children and adolescents with COVID-19 were included. The prevalence of long COVID was 25.24% (95% CI 18.17-33.02), and the most prevalent clinical manifestations were mood symptoms (16.50%; 95% CI 7.37-28.15), fatigue (9.66%; 95% CI 4.45-16.46), and sleep disorders (8.42%; 95% CI 3.41-15.20). When compared to controls, children infected by SARS-CoV-2 had a higher risk of persistent dyspnea (OR 2.69 95%CI 2.30-3.14), anosmia/ageusia (OR 10.68, 95%CI 2.48, 46.03), and/or fever (OR 2.23, 95%CI 1.22-4.07). The main limitation of these meta-analyses is the probability of bias, which includes lack of standardized definitions, recall, selection, misclassification, nonresponse and/or loss of follow-up, and the high level of heterogeneity.
Conclusion These meta-analyses provide an overview of the broad symptomatology of long COVID in minors, which may help improve management, rehabilitation programs, and future development of guidelines and therapeutic research for COVID-19.
DISCUSSION
The prevalence of long COVID in children and adolescents, following a COVID-19 infection was 25.24%. The five most prevalent clinical manifestations were mood symptoms (16.50%), fatigue (9.66%), sleep disorders (8.42%), headache (7.84%), and respiratory symptoms (7.62%). It was only possible to perform meta-analyses of ORs comparing cases and controls for 13 symptoms. When compared to controls, persons with COVID-19 had a higher risk of presenting persistent dyspnea, anosmia/ageusia, and/or fever.
Interestingly, many of the symptoms identified in these meta-analyses, such as mood, fatigue, sleep disorders, orthostatic intolerance, decreased concentration, confusion, memory loss, balance problems, exercise intolerance, hyperhidrosis, blurred vision, body temperature dysregulation, dysfunction on heart, rate variability and palpitations, constipation or diarrhea, and dysphagia, are commonly present in dysautonomia 29.
Dysautonomia is defined as a dysfunction of the sympathetic and/or parasympathetic autonomic nervous system Postural orthostatic tachycardia syndrome, chronic fatigue syndrome (CFS), and myalgic encephalomyelitis (ME) are subclassifications of this condition30. Moreover, the constellation of symptoms because of long COVID can vary from patient to patient, fluctuating in their frequency and severity 31. Several viruses have been shown to trigger ME/CFS, including the Epstein Barr Virus, Ross River virus, and earlier coronaviruses (e.g., SARS and MERS) 32. However, it remains unclear whether dysautonomia may occur as a direct result of the SARS-CoV-2 infection, interaction with other viruses, or due to immune-mediated processes such as cytokines, which are known mediators of the inflammatory response 33–36.
As with other meta-analyses, the strength of this study centers on the large sample size 40 which helps provide identify the signs and symptoms present after acute SARS-CoV-2 infection.. Further, there were some limitations to our meta-analyses. The quality of the meta-analyses results depends on the quality of the studies included. Table 3 contains a list of all the methodological aspects that future studies need to consider. We can observe that all studies had a high probability of bias, including lack of standardized definitions recall, selection, misclassification, nonresponse, and/or loss of follow-up. Additionally, the included studies have the limitations inherited in all observational studies, including bias due to residual and unmeasured confounding. Another limitation relates to the high level of heterogeneity. To account for heterogeneity, we used a random-effects model 41. However, ideally one should stratify the meta-analysis to identify what is causing the heterogeneity. This was not possible because most studies did not include data on different groups. The differences between studies were likely due to differences in study designs, settings, populations, follow-up time, symptom ascertainment methods, inconsistent terminology, little details on stratification on pre-existing comorbidities, and prior receipt of COVID-19 therapeutics and vaccines. Only four studies mentioned what percentage of the population was already vaccinated 14,15,23,28 (Table 3)