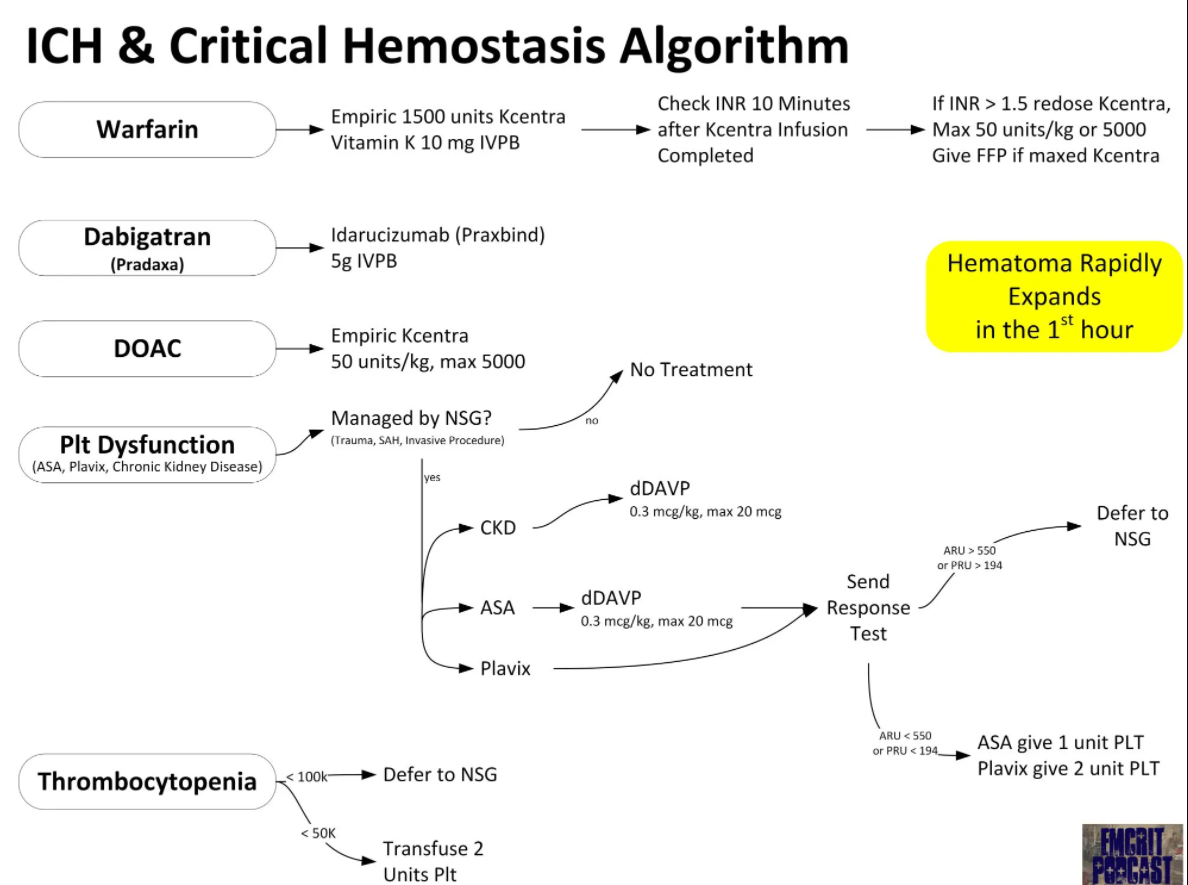
In this post I’m embedding Dr. Weingart’s flow chart from EMCrit 319 Safe and Smart Reversal of Anticoagulation / Anti-platelet Agents in 2022. March 15, 2022 by Scott Weingart, MD FCCM.*
*In order to access this podcast and show notes you need to be a member of EMCrit (I think it is definitely worth it and it helps support all of the other free resources on the EMCrit site). So, all I’ve included from his show notes is:
What follows below is resource on the the safe use of Phytonadione (Vitamin K1).
Phytonadione (Vitamin K1) from StatPearls.
Curtis J. Ingold; Shane R. Sergent. Last Update: July 22, 2021.
Phytonadione is used off-label for intracranial hemorrhage associated with vitamin K antagonist anticoagulants.
Dosing for intracranial hemorrhage associated with vitamin K antagonist anticoagulants:
Adverse Effects
Most adverse effects from phytonadione occur with IV, SQ, and IM administration[10][11]:
In a retrospective cohort study by Shields et al. published in 2001, they evaluated the efficacy and safety of IV phytonadione. Patients who met the inclusion criteria were those who had received IV phytonadione between September 1, 1994, and March 31, 1996, and were taking warfarin for two weeks or longer. Patients were excluded if they were currently bleeding or received concomitant factor replacement therapy. Out of the 105 patients assessed, 29 patients received a second dose. The researchers found adverse reactions in only two patients. Both patients experienced dyspnea and chest tightness that resolved by stopping the infusion.[10]
Phytonadione (vitamin K1) has a black box warning due to severe anaphylaxis when given IV that may cause shock, respiratory arrest, and cardiac arrest.[8]