In this post I link to and excerpt from Dr. Josh Farkas’ Pulmcrit [Link is to the table of contents] post, Mastering the dark arts of BIPAP & HFNC.
Note to myself: I have only excerpted a small part of Dr. Farkas post and so I need to go to link above for review. As always the excerpts are just to help me remember the information.
All that follows is from Mastering the dark arts of BIPAP & HFNC:
Links [to other posts on respiratory support]
- Optimizing respiratory drive to avoid failure (PulmCrit) – Probably most relevant companion post to this one.
- Avoiding gratuitous ABGs
- Sedation for the anxious COPD patient & more discussion of Flash COPD (PulmCrit)
- Sympathetic Crashing Acute Pulmonary Edema (SCAPE) – EMCrit’s classic inaugural podcast from 2009.
- HFNC for pneumonia (FLORALI study; PulmCrit)
- HFNC to avoid reintubation part I (contains some basic information on HFNC; PulmCrit
Summary: The Bullet
- The role of noninvasive respiratory support is generally to reduce the patient’s work of breathing, thereby avoiding diaphragmatic exhaustion.
- The goal of noninvasive respiratory support isn’t to immediately normalize the ABG.
- Serial evaluation by experienced practitioners is generally far more useful than monitoring ABG values.
- The choice of BiPAP vs. HFNC may be made on the basis of the patient’s diagnosis (e.g. pneumonia vs. heart failure), not the ABG values.
- BiPAP should never be used to “blow off” CO2 in a patient with hypoventilation due to drug intoxication.
Goals of therapy
The goal of HFNC or BiPAP is to stabilize the patient, in order to buy time for the underlying disease process to improve. Therefore, my goals are as follows:
- Maintain adequate oxygenation.
- Provide enough ventilatory support so that the patient is comfortable and doesn’t develop respiratory muscle fatigue.
- Ensure that the patient is protecting their airway.
- Serial examination (focused on #1-#3) shows that the patient’s trajectory is either stable or improving (2).
Assessment
For most patients on noninvasive respiratory support, clinical assessment is all that’s needed. This involves three or four pieces:
- Oxygenation: If the patient has a good pulse oximetry waveform, the preferred method is simply to monitor pulse oximetry (ABG is rarely needed to measure oxygenation).
- Work of breathing: The best metric is the respiratory rate. Worsening tachypnea (e.g. respiratory rate >35 b/m) suggests that the patient may eventually tire out. Additional warning signs include retractions, diaphoresis, tripoding, shallow breathing, and an abdominal paradoxical breathing pattern. Asking the patient how they feel is generally useful, but some patients minimize their symptoms.
- Mentation: A patient who is easily arousible and mentating adequately doesn’t have life-threatening hypercapnia (3).
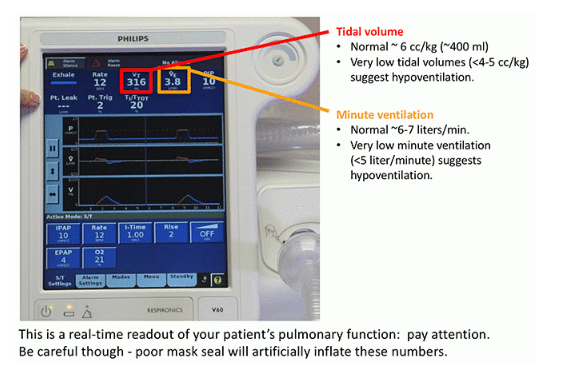
- BiPAP monitor: Low tidal volumes and/or low minute ventilation may suggest hypoventilation (4). Alternatively, adequate tidal volumes and minute ventilation suggest a satisfactory response to BiPAP.
Look at the monitor and the patient, talk to the patient, perhaps try to wake them if they are sleeping. That’s all. An experienced clinician will perform this assessment in about a minute.
The most common problem with assessment is excessive reliance on ABG values. This is problematic for many reasons: [See the post]
One situation where ABG/VBG is necessary is the somnolent patient who has received sedation (e.g. to facilitate tolerance of the BiPAP mask). Blood gas analysis is needed to sort out whether somnolence is due to hypercapnia or medication effect. Please note that VBGs are generally completely adequate to monitor a patient with respiratory failure. If the patient has a peripheral IV line with blood return, this is a humane and rapid approach to obtain VBGs.
The key to assessment is frequent assessment by multiple providers. Patients will be serially evaluated by a nurse, a respiratory therapist, and a physician. When in doubt, all providers should discuss their impressions. A team approach combines dozens of assessments by several experienced clinicians.
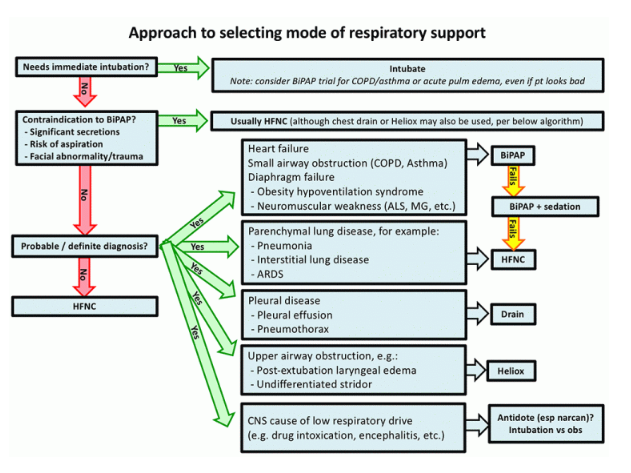
Are there contraindications to full-face BiPAP?
Contraindications include:
- Significant secretions: Positive pressure and the BiPAP mask impair expectoration. Sometimes, it is possible to maintain a patient on BiPAP with occasional breaks on HFNC for secretion clearance (e.g. a COPD patient with mild secretions). However, for a patient with copious secretions, BiPAP is contraindicated. In such cases, BiPAP may initially have excellent results, but eventually mucus plugging occurs with abrupt deterioration.
- Facial trauma, burns, or other anatomic problem with mask seal.
- Risk of aspiration: Aspiration may occur if the patient vomits and is unable to remove the BiPAP mask. Therefore, evaluating aspiration risk requires judging the likelihood of vomiting (e.g. increased with bowel obstruction or pancreatitis) versus the mental status. Note that altered mental status due to hypercapnia isn’t an absolute contraindication to BiPAP.
There are nearly no contraindications to HFNC, so any patient with a contraindication to BiPAP can be treated with HFNC (6). BiPAP with a nasal interface can also be considered here, especially if HFNC isn’t available (7).
BiPAP-sensitive conditions
BiPAP has some important advantages compared to HFNC:
- Positive pressure reduces pre-load and after-load on the heart, improving heart failure (this works similar to an ACE-inhibitor – but easier to titrate and no nephrotoxicity).
- BiPAP can provide a greater amount of mechanical support for breathing. This is desirable for patients with respiratory muscle weakness or obesity-hypoventilation syndrome (both conditions involve an imbalance between diaphragmatic strength versus work of breathing).
- For patients with small airway obstruction (e.g. COPD/asthma), BiPAP can provide mechanical support. The expiratory airway pressure (PEEP) may also help stent open airways during exhalation (8).
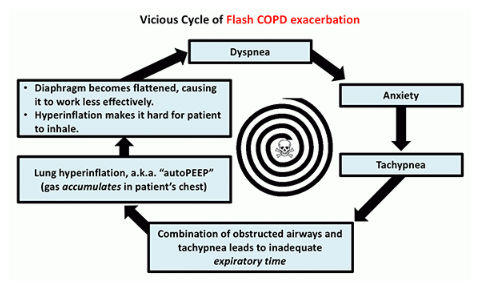
For patients with these conditions, I will generally make a real effort to use BiPAP. If the patient can’t tolerate BiPAP due to anxiety, it may be worth using sedation to facilitate BiPAP tolerance. Sedation is particularly useful for patients with COPD or asthma, who require a slow respiratory rate in order to exhale properly:
Some brief comments on various sedatives to facilitate BiPAP:
- Dexmedetomidine is very effective and safe if the patient can wait long enough to titrate this up (9). Dexmedetomidine can be up-titrated to induce light sleep, without affecting respiratory drive.
- Ketamine dissociation may be useful up-front, especially in asthma (because it provides bronchodilation). This only provides sedation for 30-60 minutes, so another agent may be needed for ongoing sedation.
- Fentanyl in tiny divided doses may be effective in patients who are very tachypneic, but requires caution and meticulous monitoring (explored further here)(10).
- IV haloperidol or olanzapine may be considered (with the advantage that they don’t suppress the respiratory drive).
- Benzodiazepines have unpredictable effects. They sometimes work, but can also cause confusion or paradoxical agitation.
HFNC for patients with parenchymal lung disease (e.g. pneumonia)
Parenchymal lung disease refers to any primary disease of the alveolar tissue itself. The advantages of HFNC here include the following:
- Reduction in work of breathing due to dead space washout
- Preserved ability to cough & clear secretions
- Excellent tolerance, including for extended periods of time (important for patients with interstitial lung disease who may take several days to recover)
- Improved ability to communicate with patients and assess their progress
Compared to heart failure and COPD, parenchymal lung disease is a less BiPAP-responsive physiology. Studies have found questionable benefit from BiPAP in this situation. The FLORALI trial showed that in a group of patients with hypoxemic respiratory failure (mostly from pneumonia), HFNC was more successful than BiPAP at avoiding intubation and improving mortality.
Treat pleural disease with drainage
Temporize upper airway obstruction with Heliox
Heliox is a fixed-ratio mixture of helium and oxygen which has lower viscosity than air. This may be used to stabilize patients with upper airway obstruction. One limitation is that these ratios contain a fixed amount of oxygen (typically 70-30 mix or 60-40 mix with 30% or 40% FiO2), so they cannot be used for patients who require >40% inspired oxygen. Heliox isn’t a cure for upper airway obstruction, but it may be useful:
- Stabilization of post-extubation laryngeal edema, giving steroid time to work.
- Stabilization of the patient with undifferentiated stridor, allowing time to gather materials and personnel for safe management (e.g. difficult airway cart, bronchoscope, colleague prepared for cricothyrotomy with double-setup).
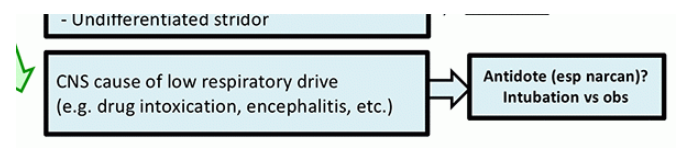
Avoid BiPAP or HFNC in primary CNS disease
One practice which drives me crazy is the use of BiPAP for treatment of hypercapnia due to drug intoxication. If the patient is so severely intoxicated that they truly need ventilatory support, then they should be intubated (aspiration risk precludes the use of BiPAP here). A reasonable approach to these patients may be as follows:
- If the patient is protecting their airway and doesn’t clearly need intubation, then monitor very carefully on room air or low-flow oxygen (12). If the pCO2 is moderately elevated, that’s OK as long as the patient is clinically doing OK (e.g. protecting airway, oxygenating, not rapidly deteriorating). Please note that mild hypercapnia is an expected feature of opioid intoxication, so it shouldn’t cause panic. Treat the patient, not the ABG.
- If the patient isn’t protecting their airway, attempt antidotal therapy (e.g. naloxone). If this fails, then intubate.
Consider HFNC if the diagnosis is unclear
For a patient with unclear diagnosis, either BiPAP or HFNC could be used. My preference is HFNC for the following reasons:
- HFNC facilitates communication, including obtaining additional history.
- BiPAP may make some patients look much worse (due to anxiety) or others look much better (due to treatment of CHF or COPD). This can confuse matters if you don’t know what is going on.
Choice of BiPAP vs. HFNC depends on the diagnosis, not the ABG
For example:
- Consider a patient with COPD or heart failure, who is in severe respiratory distress and has a normal pCO2 level. This patient will benefit from BiPAP, even though the pCO2 level is normal.
- Consider a patient with intoxication and elevated pCO2 level. BiPAP is not useful here to “blow off the CO2” – as discussed above it is actually contraindicated.
Selecting the treatment based on the diagnosis streamlines management. The diagnosis can generally be determined rapidly on the basis of history, physical exam, and bedside ultrasonography. This allows for immediate treatment, without waiting for an arterial blood gas measurement.