Today, I reviewed, link to and excerpt from An Updated Approach to Antiobesity Pharmacotherapy: Moving Beyond the 5% Weight Loss Goal [PubMed Abstract] [Full-Text HTML] [Full-Text PDF]. J Endocr Soc. 2023 Jan 10;7(3):bvac195. doi: 10.1210/jendso/bvac195. eCollection 2023 Jan 6.
There are 92 similar articles in PubMed.
All that follows is from the above resource.
Abstract
Despite professional society guidelines recommending that obesity be treated as a chronic disease by emphasizing the use of lifestyle modification in conjunction with pharmacotherapy, antiobesity medications are uncommonly prescribed in most clinical practices. The recent Food and Drug Administration approval of semaglutide 2.4 mg weekly to treat obesity—as well as other forthcoming advancements in diabetes and antiobesity medications—highlights the potential of pharmacotherapy to significantly augment weight loss efforts. In this Expert Endocrine Consult, we review the evolving role of antiobesity pharmacotherapy in clinical practice and suggest a framework for the use of these medications.
The prevalence of obesity has trended upwards in recent decades in the United States. In 2017-2018, the adult obesity rate passed 40% nationally for the first time, and it is predicted to eclipse 50% of US adults by 2030 [1]. Obesity increases the risk for type 2 diabetes (T2DM), hypertension, cardiovascular disease (CVD), chronic kidney disease, osteoarthritis, nonalcoholic steatohepatitis (NASH), and death from several cancers [2–8].
The rise in obesity and its comorbidities highlights the need for improvements in obesity treatment. Despite the availability of antiobesity medications (AOMs) and data supporting their efficacy and safety, their rate of use is low, with less than 2% of patients who qualify for treatment receiving prescriptions for AOMs [9,10]. Possible explanations include physicians’ lack of training in obesity management and use of AOMs, limited time during clinical visits, the high cost of many AOMs with limited insurance coverage, and hesitancy stemming from a history of failure of AOMs after regulatory approval [11].
Exciting advances in weight loss pharmacotherapy are taking place. The significantly greater efficacy of semaglutide and other emerging pharmacologic options compared with previously Food and Drug Administration (FDA)–approved AOMs highlights the need for clear guidelines and clinician and patient education regarding the potential for these therapies to significantly augment weight loss efforts. The purpose of this Expert Endocrine Consult is to introduce an updated approach to the medical treatment of obesity considering recent advances in the field of AOMs. This updated approach is aimed at both the endocrinologist and the primary care physician looking to expand their practice to include pharmacological treatment of obesity.
Clinical Case
A 55-year-old man with a past medical history of T2DM, obesity (body mass index [BMI] 35.5 kg/m2), and depression presents to your primary care clinic. He was diagnosed with T2DM 6 years ago. Current therapy includes semaglutide 1.0 mg weekly and metformin 1000 mg twice daily. He is also on paroxetine 30 mg daily for depression. Before starting semaglutide 1 year ago, his hemoglobin A1c was 8.8%. Since starting semaglutide, his hemoglobin A1C has decreased to 7.3% and he has lost 24 lbs, 10.9 kg. (−4.2% total body weight loss). He is very pleased with these results and is interested in losing more weight. He has worked on lifestyle changes including a calorie-restricted diet but has never enrolled in a structured weight loss program. Although he reports his depressive symptoms are well controlled on paroxetine, he identifies a lack of energy as a barrier to adhering to a daily exercise program. What pharmacologic options might be appropriate as a next therapeutic step?
Lifestyle
Lifestyle modification serves as the foundation for weight loss treatment. Comprehensive lifestyle interventions including a reduced calorie diet, increased physical activity, and behavioral support should be promoted and have been discussed in detail in several guidelines [12–14]. From a dietary perspective, several approaches have been studied, including low-fat, low-carbohydrate, low-glycemic-load, low-fat vegan-style, lacto-ovo-vegetarian, Mediterranean-style, and higher-protein diets, among others, without evidence of the superiority of one approach to another [15, 16]. Dietary interventions show efficacy over the short-term, with studies showing maximal weight loss at 6 months, with small losses maintained up to 2 years. Over the long-term, patients tend to regain most of the weight that was lost.
Intensive lifestyle interventions for patients with T2DM, including both group and individual counseling sessions, a reduced calorie intake with meal replacement products, and at least 175 minutes per week of moderate-intensity physical activity, have been shown to produce modest weight loss over the long-term (6% vs 3% in the placebo group in the 10-year Look AHEAD trial) [17]. However, even these intensive lifestyle interventions failed to maintain goal weight loss (>7%) over the long-term and have not been shown to change cardiovascular outcomes.
The challenges in maintaining long-term weight loss are due to compensatory biological mechanisms that decrease energy expenditure (out of proportion to reduction in body mass) and increase appetite in response to weight loss [18–22]. Because these changes persist after weight loss, lifestyle interventions often need to be augmented by therapies such as AOMs and bariatric surgery that target these compensatory mechanisms [23]. Pharmacotherapy for obesity should be considered for patients who have not met weight loss goals with lifestyle modification alone and in those who have lost weight but struggle to maintain their weight loss [24, 25].
Medications that Cause Weight Gain
In addition to lifestyle counseling, a central point of caring for patients with overweight or obesity involves reviewing their use of medications that promote weight gain and prescribing drugs that are weight neutral or that promote weight loss [24,26]. Common medication classes associated with weight gain include steroids, antipsychotics, antiepileptics, glucocorticoids, and gabapentinoid medications. These medications are often indicated and effective for treating their respective primary disorders. However, if patients experience significant weight gain on these medications, particularly to a degree where the negative effects of weight gain may exceed the positive treatment effects, clinicians should consider switching patients to alternative medications that are weight neutral or that promote weight loss. Adjustment of these medications should be made in a shared decision making process including the patient and prescribing provider (eg, psychiatry, neurology, or other specialists).
Type 2 Diabetes Mellitus
For patients with T2DM and overweight or obesity, clinicians should consider weight-losing medications (ie, metformin, sodium–glucose cotransporter 2 inhibitors, and glucagon-like peptide-1 (GLP-1) receptor agonists) and weight-neutral medications (ie, dipeptidyl peptidase-4 inhibitors), and, when possible, avoid agents associated with weight gain such as pioglitazone, sulfonylureas, and insulin [14, 24, 27]. For patients requiring insulin therapy, there are strategies to mitigate insulin-associated weight gain. The addition of metformin and GLP-1 agonists has been shown to reduce or, in the case of GLP-1 agonists, even nullify insulin-associated weight gain [28–31]. Clinicians should add 1 of these 2 agents when starting a patient with T2DM on insulin therapy. Among insulin therapies, basal insulin is associated with less weight gain than biphasic or prandial short-acting insulin and should be the first-line option [32, 33].
Psychiatric Disorders
For patients with psychiatric disorders, such as major depression or bipolar disorder, clinicians should choose weight-neutral medications when appropriate. The antidepressant medications paroxetine, amitriptyline, and mirtazapine have been associated with weight gain and should be avoided in favor of bupropion and fluoxetine when possible as they are associated with weight loss [26, 34–36]. Atypical antipsychotics are the drug class associated with the most weight gain. Among this drug class, olanzapine, quetiapine, and risperidone are associated with the largest weight gain and should be avoided in patients with overweight or obesity when possible [26]. While no atypical antipsychotics are associated with weight loss, ziprasidone may be weight neutral and appears to be a better option for patients who are overweight and obese [37].
Anticonvulsants and Mood Stabilizers
For patients with seizure disorders, clinicians should engage in shared decision-making with patients to make an informed decision about antiepileptic drug (AED) choice. When appropriate, clinicians should avoid divalproex, carbamazepine, and gabapentin, which are all associated with weight gain. Conversely, zonisamide and topiramate are associated with weight loss and should be considered in patients with recent weight gain or if overweight, when appropriate. Lithium and lamotrigine appear to be weight neutral and should also be favored over AEDs associated with weight gain [26].
Inflammatory Rheumatic Diseases
Obesity is a common comorbidity in patients with inflammatory rheumatic diseases, with a hypothesized causal role due to the proinflammatory nature of adipose tissue [38, 39]. Patients with obesity have higher disease scores and are less likely to respond to treatment with disease-modifying antirheumatic drugs [40]. In patients with inflammatory rheumatic diseases, clinicians should minimize or avoid corticosteroids in favor of nonsteroidal anti-inflammatory drugs and disease-modifying antirheumatic drugs given the tendency of corticosteroids to promote weight gain [41, 42].
Antiobesity Pharmacotherapy
Pharmacotherapy is indicated as an adjunct to caloric restriction and physical activity in adults with a BMI ≥30 kg/m2 or BMI 27 to 29.9 kg/m2 with at least 1 weight-related comorbidity such as diabetes, hypertension, hyperlipidemia, obstructive sleep apnea, nonalcoholic fatty liver disease, or osteoarthritis. Most AOMs target the compensatory increase in appetite that occurs in the setting of weight loss. In counseling patients on AOMs, patients should be educated that the addition of AOMs to a lifestyle program enhances weight loss (as has been shown in many clinical trials) [43–48].
Overview of AOMs
Phentermine
Phentermine was the first FDA-approved AOM and is the most widely prescribed AOM in the United States. Phentermine acts as a sympathomimetic, increasing norepinephrine in the central nervous system, thereby suppressing appetite [49]. It was first approved in 1959 for short-term use for weight loss based on a 36-week trial that showed both continuous and intermittent administration of phentermine led to more weight loss than placebo (−12.2 kg, −13.0 kg, −4.8 kg, respectively) [50]. Two more recent randomized controlled trials from Korea have confirmed the efficacy of phentermine for short-term weight loss, both showing significant weight reduction compared with placebo over the course of 12 weeks [51, 52]. While phentermine is the only AOM that is not FDA-approved for long-term use (approval for ≤3 months), many patients are prescribed the medication long-term [9]. Available phentermine doses are 8 mg 3 times daily, and 15 mg, 30 mg, and 37.5 mg taken once daily.
Statistically significant adverse effects of phentermine in clinical trials include dry mouth (55%) and insomnia (34%), without significant differences in systolic or diastolic blood pressure, insomnia, headache, or palpitations between the treatment and placebo group [51]. Common reasons for discontinuation in the phentermine group included dry mouth and nausea/vomiting, although the phentermine group did not have a higher discontinuation rate than the control group. Although there are no long-term cardiovascular outcome studies assessing the safety of phentermine, a 2019 retrospective analysis of real-world data did not find an increase in cardiovascular risk with long-term use [53]. Phentermine is not recommended for patients with CVD and is contraindicated in patients with hyperthyroidism, glaucoma, or in patients taking monoamine oxidase (MAO) inhibitors. Relative contraindications include patients with poorly controlled hypertension or a history of addiction given its amphetamine-like properties.
Orlistat
Orlistat is an AOM that was FDA-approved for long-term use for weight management in 1999 and approved for over-the-counter use since 2007. Unlike other AOMs, orlistat does not act on appetite, but rather it inhibits triglyceride absorption in the gut. In one of the longest trials on orlistat, the 4-year XENDOS trial randomly assigned 3304 people to placebo or orlistat, with the orlistat-treated patients losing 11% of their bodyweight after 1 year compared with 6% in the placebo-treated group. Both groups experienced weight regain over the following 3 years, with the orlistat group losing 6.9% of their bodyweight at the end of 4 years compared with 4.1% for the placebo-treated group [54]. In addition to effects on weight, the study showed a reduced progression from prediabetes to diabetes in subjects treated with orlistat. Orlistat is available in 60-mg capsules over-the-counter and 120-mg capsules by prescription, both dosed 3 times daily.
Adverse effects of this medication are predictable based on its mechanism of action: gastrointestinal (GI) adverse effects including fatty/oily stool, fecal urgency, and oily spotting are common, with frequency rates ranging from 15% to 30% in most studies [55]. These GI adverse effects are the main reason for discontinuation in studies, with discontinuation rates of 5% in orlistat-treated patients (2% higher than placebo, 95% CI 1-3%). These adverse effects can be reduced by avoiding high-fat diets. It is recommended to supplement multivitamins and beta carotene due to the risk for malabsorption of fat-soluble vitamins.
Phentermine/Topiramate Extended Release
Phentermine can be combined with topiramate for additional weight loss effects. Topiramate is an AED that was FDA approved for seizures in 1996 and migraine prevention in 2004. Although its mechanism of action for weight loss is not entirely known, it is thought to suppress appetite by increasing dopamine release and inhibiting glutamate receptors, as well as modulating neuropeptide-Y, a hormone that increases food consumption [56]. Combined with phentermine, the 2 drugs have additive effects on weight loss. This combination was approved for use in 2012.
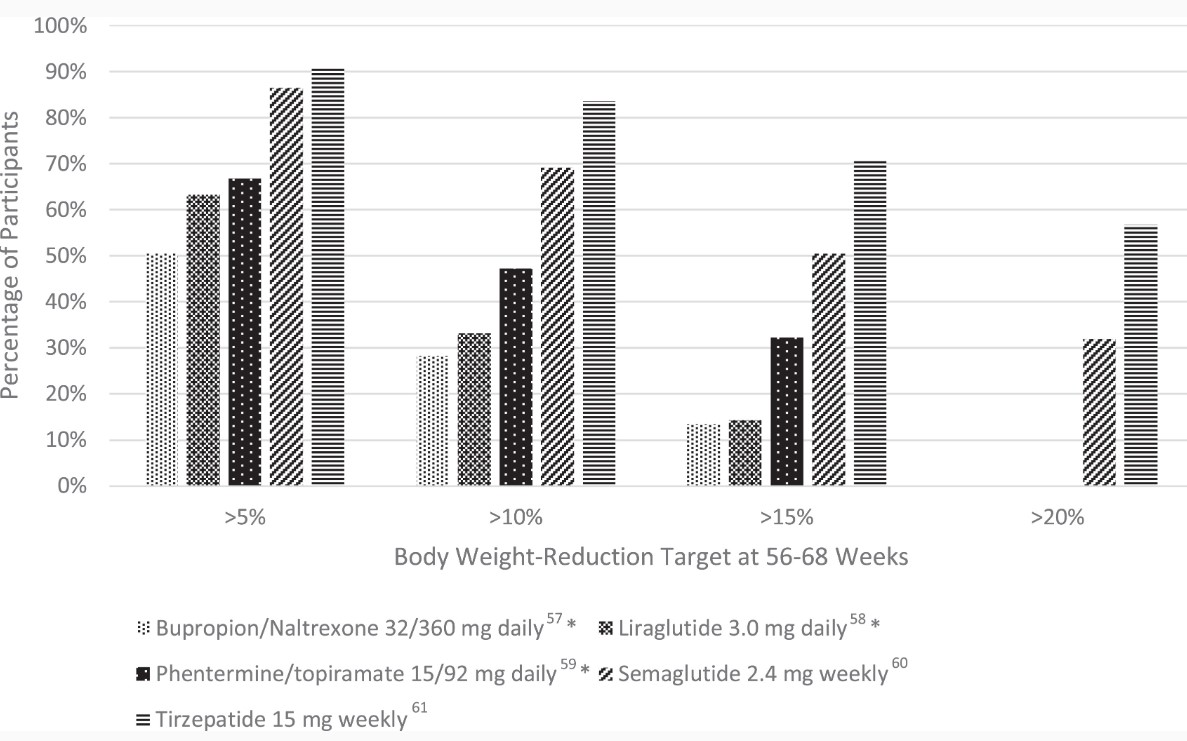
The CONQUER trial was a 56-week, phase 3 trial of 2487 adults who were overweight or obese (age 18-70 years, BMI 27-45 kg/m²) and 2 or more comorbidities (hypertension, dyslipidemia diabetes, or prediabetes, or abdominal obesity) [47]. Patients were randomized to placebo, once daily phentermine 7.5 mg/topiramate ER 46 mg, or once daily phentermine 15 mg/topiramate ER 92 mg in a 2:1:2 ratio. At 56 weeks, body weight change was −1.4 kg, −8.1 kg, and −10.2 kg, respectively. Five percent weight loss was achieved in 21% of patients with placebo, 62% of patients with phentermine 7.5 mg/topiramate 46 mg daily, and 70% of patients with phentermine 15 mg/92 mg daily; the corresponding numbers for 10% weight loss were 7%, 37%, and 48% (see Fig. 1).
The most common adverse effects include dry mouth (13% for the 7.5/46 mg dose, 21% for the 15/92 mg dose), paresthesia (14%, 21% respectively), and constipation (15%, 17% respectively), as well as a small increase in heart rate (0.6-1.6 beats/minute higher than placebo). The CONQUER trial also showed a dose-related increase in anxiety, depression, and cognitive impairment. Similar to phentermine monotherapy, phentermine/topiramate ER is not recommended for patients with CVD and is contraindicated in patients with hyperthyroidism, glaucoma, or in patients taking MAO inhibitors.
Topiramate poses a risk for orofacial clefts in infants exposed in utero. Clinicians should be aware of the risk evaluation mitigation strategy for phentermine/topiramate ER that informs clinicians and patients of the potential for this teratogenic effect, the importance of pregnancy prevention in females of reproductive age receiving this medication, and the need to discontinue the medication immediately if pregnancy occurs.
Naltrexone/Bupropion Sustained Release
The combination tablet of bupropion and naltrexone has been FDA approved for weight loss since 2014. Bupropion is a reuptake inhibitor of dopamine and norepinephrine that is FDA approved for depression and smoking cessation. Among antidepressants, it is least likely to produce weight gain. Naltrexone is an opioid antagonist approved for the treatment of opioid and alcohol dependence. While naltrexone as monotherapy does not appear to cause weight loss, when combined with bupropion it appears to diminish the mu-opioid receptor autoinhibitory feedback loop on anorexigenic hypothalamic neurons activated by bupropion, leading to reduced food intake and weight loss [57, 58]. The initial dose is 1 tablet (8 mg naltrexone and 90 mg of bupropion) daily for 1 week, which over the course of 4 weeks is increased stepwise up to the treatment dose of 2 tablets twice daily (32 mg naltrexone and 360 mg bupropion). Typical weight loss seen with this combination drug is in the 5% to 6% range [59].
The cardiovascular safety of naltrexone/bupropion SR combination was tested in the LIGHT trial, a randomized, placebo-controlled, noninferiority, cardiovascular outcomes trial. The trial was terminated prematurely after the study sponsor publicly released confidential favorable interim results after only 25% of expected vascular events had accrued, making it difficult to interpret the cardiovascular safety of this combination drug [60].
Statistically significant adverse effects of bupropion/naltrexone include nausea (30%), headache (14%), and constipation (15%), without significant differences in depression or suicidality events, insomnia, dizziness, or dry mouth between the treatment and placebo groups [57, 59]. The most common adverse effects leading to discontinuation were nausea (6%) and headache (2.6%) [57]. Contraindications include pregnancy, uncontrolled hypertension, seizure disorder, eating disorder, chronic opioid use, severe hepatic dysfunction, and in patients taking MAO inhibitors.
Liraglutide 3.0 mg Daily
Liraglutide was the first GLP-1 agonist to be approved for weight loss treatment in December 2014. GLP-1 agonists promote weight loss by delaying gastric emptying and suppressing subjective hunger and food intake [61]. The SCALE Obesity and Prediabetes and SCALE Diabetes were both 56-week, randomized, double-blind, placebo-controlled, clinical trials examining the effect of liraglutide 3.0 mg daily on patients with normoglycemia, prediabetes, and diabetes. Both trials demonstrated significantly greater weight loss with liraglutide. In SCALE Obesity and Prediabetes, weight loss was −8.0 ± 6.7% with liraglutide vs −2.6 ± 5.7% with placebo (P < .001), and in SCALE Diabetes weight loss was 6.0% with liraglutide vs 2.0% with placebo (P < .001) [62, 63]. In the former trial, more participants in the liraglutide group achieved a weight reduction of ≥5% (63.2 vs 27.1%), ≥10% (33.1 vs 10.6%), and ≥15% (14.4 vs 3.5%) (see Fig. 1) [62]. Liraglutide is administered subcutaneously once daily, with an initial dose of 0.6 mg daily for 1 week with weekly increases in dose (1.2, 1.8. 2.4, 3 mg) to the recommended 3.0 mg dose.
Similar to other GLP-1 agonists, GI adverse effects are common, including nausea (40%), diarrhea (20%), constipation (20%) and vomiting (16%). GI events were the most common reason for discontinuation in the liraglutide group (6.4% vs 0.7% in placebo group). Serious but less common adverse effects include gallbladder disease (2.5%) and pancreatitis (0.4%). While liraglutide was associated with benign and malignant thyroid C-cell tumors in rodent studies, human trials have not shown evidence of these tumors.
Semaglutide 2.4 mg Weekly
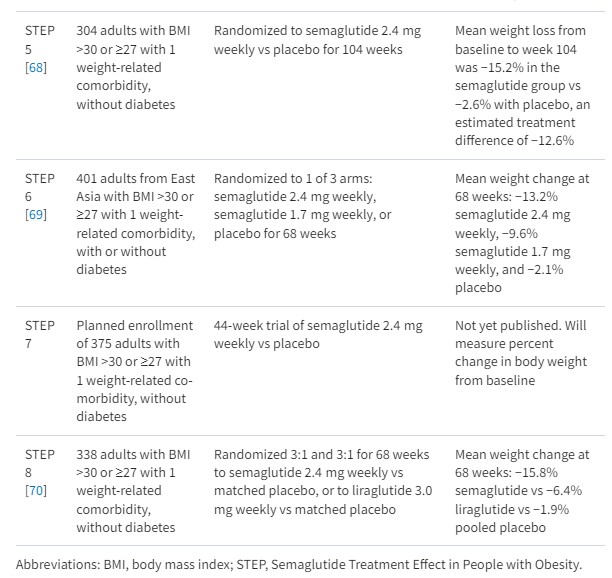
Semaglutide is the newest FDA-approved GLP-1 agonist for chronic weight management. FDA approval was based on results from the STEP trials (see Table 1). In the STEP 1 trial, a total of 1961 adults with obesity or overweight with a weight-related comorbidity (excluding patients with diabetes) were randomized to semaglutide 2.4 mg weekly or placebo as well as lifestyle intervention [64]. The study found the mean change in body weight from baseline to week 68 was −14.9% for the semaglutide group and −2.4% for the placebo group, for an estimated treatment difference of −12.4% (P < .001). When compared with placebo, more participants in the semaglutide group achieved a weight reduction of ≥5% (86.4 vs 31.5%), ≥ 10% (69.1 vs 12.0%), ≥ 15% (50.5 vs 4.9%), and ≥20% (32.0 vs 1.7%) (see Fig. 1). Among participants with prediabetes at baseline, 84% reverted to normoglycemia in the intervention group vs 38% in the control.
The STEP 4 trial demonstrated the beneficial effect of long-term treatment with semaglutide for weight loss [67]. The trial included a 20-week lead-in period in which all trial participants received semaglutide 2.4 mg weekly, followed by a 48-week period where the intervention group continued semaglutide 2.4 mg weekly and the control group received a placebo. The semaglutide group continued to lose and then sustained weight loss over the following 48 weeks, whereas the placebo group regained weight and had worsening of cardiovascular risk factors. Semaglutide is administered subcutaneously once weekly, with an initial dose of 0.25 mg daily once weekly for 4 weeks, with the dose increased at 4-week intervals (0.5, 1.0, 1.7, 2.4 mg) to the recommended 2.4 mg dose.
Like other GLP-1 agonists, GI adverse effects are common, occurring more frequently in participants receiving semaglutide than those receiving placebo in the STEP 1 trial (74.2 vs 47.9%). GI events were the most common reason for discontinuation in the semaglutide group (4.5% vs 0.8% in placebo group). Serious but less common adverse effects include gallbladder disease (2.6%) and pancreatitis (0.2%). There was no difference between treatment and placebo groups in the incidence of benign and malignant neoplasms.
Other Medications that Produce Weight Loss
Setmelanotide is a selective agonist of the melanocortin-4-receptor (MC4R) that was FDA approved in November 2020 for the treatment of monogenic obesity due to pro-opiomelanocortin, leptin receptor deficiency, or proprotein convertase subtilisin/kexin type 1 deficiency in individuals ages 6 or older, and was approved in June 2022 for Bardet–Beidel syndrome. The mechanism of action involves the binding of leptin to its receptor, which leads to agonism of the MC4R through the cleavage of peptides. Genetic mutations in this pathway lead to hyperphagia, impaired pubertal development, obesity, and insulin resistance. Setmelanotide acts as an agonist to the MC4R, bypassing the effects of these genetic mutations. A recent trial showed a mean weight loss of 25.6% among individuals with pro-opiomelanocortin deficiency and 12.5% among those with leptin receptor deficiency [71].
Zonisamide is another AED that can produce modest weight loss, although it is not FDA approved for this indication. Studies ranging from 16 weeks to 1 year show 6% to 7% weight loss for zonisamide 400 mg daily [72, 73]. Common reported adverse effects include nausea/vomiting, headaches, anxiety, impaired memory, and language problems.
Recommended Approach to AOMs
Considerations When Selecting an AOM
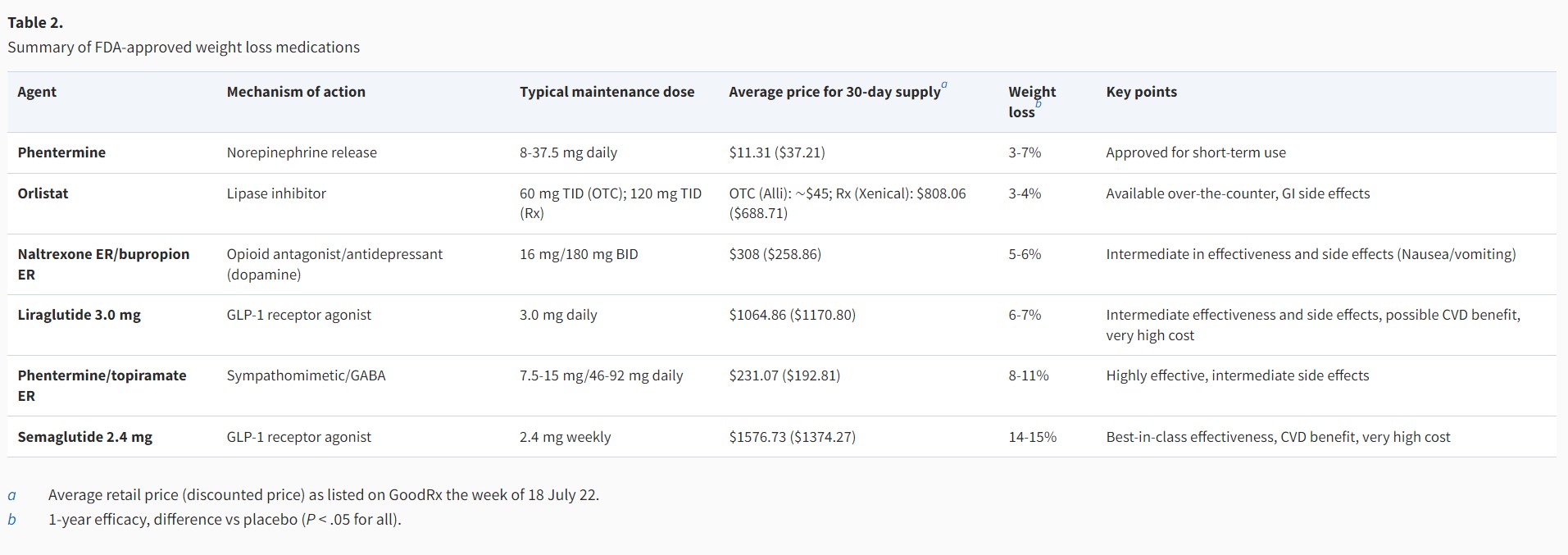
Unfortunately, medication cost and insurance coverage are the primary drivers in selecting AOMs for an individual patient (see Table 2 for average medication costs). A 2018 review paper examined 136 marketplace health insurance plans, finding only 11% had coverage for AOMs [74]. Medicare excludes drug therapy for obesity, and only 7 state Medicaid programs have AOM coverage. A recent opinion article by Roser et al posits that, with the annual cost of overweight and obesity estimated at $990 billion per annum, it likely would be more cost-effective for insurance companies to cover AOMs than to continue the current limited coverage of obesity therapies [75, 76]. Patient advocacy groups are currently lobbying insurers to pay for AOMs and lobbying Congress to pass a bill (the Treat and Reduce Obesity Act of 2021) that would require Medicare to pay for AOMs [77, 78].
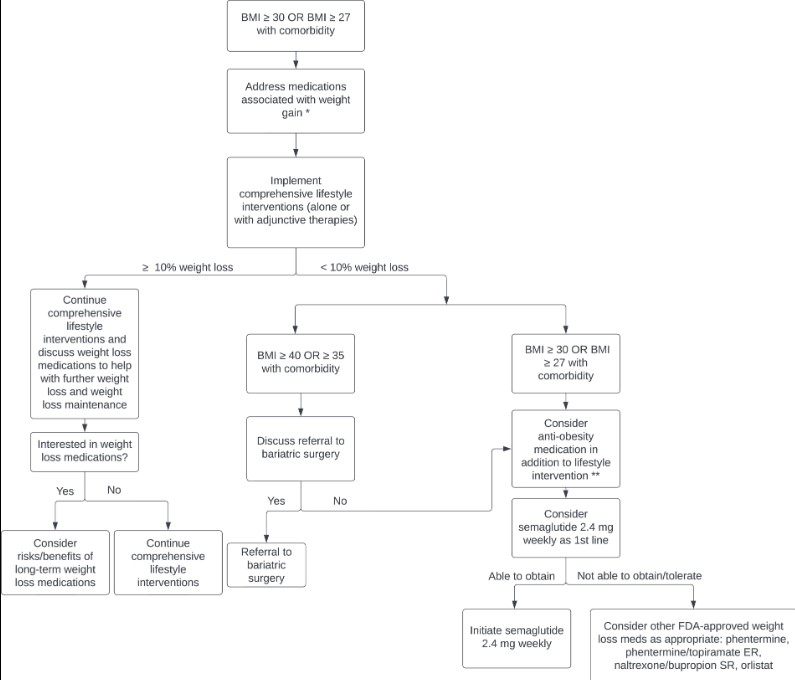
Ideally, cost would not play a significant role in AOM selection, and clinicians would primarily consider the degree of weight loss desired and the effect on comorbid conditions when selecting an AOM. Guidelines produced by the American Heart Association, American College of Cardiology and The Obesity Society (AHA/ACC/TOS), and the Endocrine Society both recommend a goal of ≥5% weight loss [12, 24]. However, data show that higher magnitudes of weight loss (≥10%) lead to greater and more clinically meaningful improvements in weight-related comorbidities, including greater relative risk reduction for cardiovascular events, improvements in NASH histology, decreased disease activity in patients with inflammatory rheumatic disease, and improvements in osteoarthritis, obstructive sleep apnea, and cancer risk [79–83]. With recent data showing a significantly greater degree of weight loss with semaglutide (∼15%% vs 5-10% for the other FDA-approved AOMs, with 69% of participants achieving ≥10% weight loss and 50% of participants achieving >15% weight loss), we recommend semaglutide 2.4 mg weekly as the first line AOM for obesity management (see Fig. 2). In addition, we propose that weight loss goals for most individuals with obesity should be ≥10%, as this is now achievable with current AOMs.
Figure 2.
Recommended approach to initiating AOM therapy in patients with BMI ≥30 kg/m2 or ≥27 kg/m2 with a weight-related comorbidity. *For treatment strategies specific to patients with T2DM, please see “Medications that Cause Weight Gain” “Type 2 Diabetes Mellitus”. **Engage in shared decision-making, considering patient-specific factors such as preferences on modes of delivery.
Practical Tips for Success With GLP-1 Agonists
When starting GLP-1 agonists, there are several strategies that promote success and decrease risk of discontinuation. Strategies to minimize adverse effects include slow dose escalation, counseling on expected adverse effects and their duration, and utilizing a multidisciplinary team approach (primary care provider, pharmacists, nurses, and medical assistants) to provide regular follow-up and guidance as patients initiate the medication. It is particularly important to discuss GI adverse effects, as patients who are not expecting these adverse effects may prematurely discontinue the medication.
Routine follow-up can come in many forms, including virtual visits, phone calls, pharmacist check-ins, or even portal messages at routine intervals. This type of follow-up can increase communication with the patient, normalizing adverse effects and allowing tighter dose titration, while also reducing the number of clinical visits a patient has to make, thereby reducing primary care provider burden and overall health care costs. Other strategies include a dose escalation period, with 1-week dose pauses when adverse effects are encountered, which may minimize nausea/vomiting. GI adverse effects may also be reduced by avoiding high fat foods and focusing on small meals.
Future Directions in Antiobesity Pharmacotherapy
Tirzepatide
Tirzepatide is a dual glucose-dependent insulinotropic polypeptide and GLP-1 receptor agonist that was FDA approved for T2DM in May 2022. Two sets of trials have examined the efficacy of tirzepatide in treating T2DM and reducing body weight. The SURPASS trials examined the effect of tirzepatide in patients with T2DM. SURPASS-1 compared 3 different doses of tirzepatide (5 mg, 10 mg, 15 mg) to placebo for 40 weeks, finding significant mean hemoglobin A1C (−1.87%, −1.89%, −2.07%) and body weight reductions (−7.9%, −9.3%, −11.0%) for all tirzepatide doses compared with the placebo group [84]. SURPASS-2 compared the same doses of tirzepatide (5 mg, 10 mg, 15 mg) with semaglutide 1.0 mg weekly, finding more effective and dose-dependent reductions in body weight, blood pressure, and hemoglobin A1C [85].
The recently published SURMOUNT-1 trial examined the efficacy of tirzepatide in producing weight loss in patients with obesity who do not have diabetes [86]. The phase 3, double-blind, randomized controlled trial assigned 2539 patients in a 1:1:1:1 ratio to receive weekly tirzepatide (5 mg, 10 mg, or 15 mg) or placebo for 72 weeks. Average weight loss at week 72 was of an unprecedented magnitude: −15.0% for the 5-mg weekly dose, −19.5% for the 10-mg dose, −20.9% for the 15-mg dose, and −3.1% for the placebo (P < .001 for all comparisons with placebo). Notably, 50% and 57% of participants in the 10- and 15-mg groups had a reduction in body weight of 20% or more, compared with 3% in the placebo group (P < .001 for all comparisons with placebo) (see Fig. 1).
Together, these trials show promise for tirzepatide as an effective and safe medication for both weight reduction and glycemic control in patients with obesity and/or T2DM. Typical adverse effects are similar to GLP-1 agonists and include nausea, vomiting, and diarrhea. No clinically significant hypoglycemia was reported in any of the trials.
Cagrilintide
Cagrilintide, a long-acting amylin analogue, is another emerging AOM. The medication mimics natural amylin, a pancreatic hormone that induces satiety. When used alone at higher doses of 2.4 and 4.5 mg weekly, resulting weight loss was 9.7% and 10.8% at 26 weeks, respectively, compared with 3.0% with placebo and 9.0% with liraglutide 3.0 mg/day [87]. Common adverse effects included nausea, diarrhea, and injection site reactions. Cagrilintide also shows promise as an adjunct to current AOMs; recent phase 1b trials show promising results for cagrilintide (0.16-4.5 mg weekly) combined with semaglutide 2.4 mg weekly when compared with placebo combined with semaglutide 2.4 mg weekly [88]. Larger trials of this combination are planned.
Emerging Therapies
Several medications with multi-receptor action are under development and in early trials, including oxyntomodulin, cotadutide, and retatrutide. Oxyntomodulin is a peptide hormone that activates both the glucagon-like peptide-1 receptor and the glucagon receptor and, when administered exogenously, can improve glucose tolerance and result in weight loss [89]. Phase 1b trials have shown favorable safety profiles and body weight and glucose-lowering effects using synthetic peptide analogues of oxyntomodulin [90]. Cotadutide is another GLP-1 and glucagon receptor agonist with a similar safety and efficacy profile currently undergoing phase 2 trials [91]. Retatrutide is a “triple-G agonist” (GLP-1, glucagon receptor, and glucose-dependent insulinotropic polypeptide receptor agonist) that is currently undergoing 2 phase 2 trials, 1 for adults with overweight or obesity and 1 for adults with T2DM. Phase 1 trials showed the drug was well tolerated with promising weight loss after 12 weeks of treatment in patients with T2DM [92].
Weight Loss Maintenance
The AHA/ACC/TOS guidelines outline 4 pillars of weight maintenance therapy:
monthly or more frequent face-to-face or telephone contact [12, 93];
200 to 300 minutes of physical activity per week;
weekly or more frequent monitoring of body weight;
continued consumption of a reduced-calorie diet.
Frequent Face-to-Face or Telephone Contact
Forms of frequent face-to-face or telephone contact with medical professionals, such as monthly multidisciplinary programs focusing on lifestyle change, can prevent weight regain [93]. Evidence supports the aphorism coined by Hall and Kahan [94]: “long term benefits require long term attention.” Continuing a comprehensive weight loss program (on site or by telephone) for periods of up to 2.5 years after initial weight loss reduction reduces weight regain, as compared with the provision of minimal intervention (ie, usual care) [94]. Example programs include community weight management groups like the Diabetes Prevention Program and commercial programs like Weight Watchers or Jenny Craig.
Physical Activity for Weight Maintenance
Physical activity plays an important role in mitigating weight regain after weight loss, more so than for initial weight loss [20, 95]. However, a significant quantity is necessary—evidence shows that high-volume exercise (>1800-2500 kcal per week) is associated with sustained weight loss [96–98]. The type of exercise (aerobic vs resistance training) appears to matter less than the quality, although aerobic exercise may have a small, if clinically insignificant, advantage over resistance training [99].
Frequent Monitoring of Body Weight
Self-weighing is associated with weight loss maintenance, with consistent self-weighing (≥6 or all 7 days/week) associated with successful weight loss maintenance [100]. Successful weight loss maintainers use self-weighing as a method to set goals and acceptable boundaries for weight variation and to prompt small corrective actions for slight weight regain before significant weight gain has occurred [101]. Other themes from qualitative studies highlight self-weighing as a tool that fosters a sense of remaining on track, reinforces healthy habits, and serves as a prompt for clear relapse protocols [102].
Continuing Dietary Changes and AOMs
If weight loss is successful with pharmacotherapy, it is recommended to continue AOMs long-term to ameliorate comorbidities and augment adherence to behavioral change [24]. Crossover trials of AOMs have shown that the weight loss effects of these medications attenuate if discontinued, like medications for other chronic disease like hypertension and T2DM [103, 104].
Patients should be encouraged to sustain the dietary changes that led to initial weight loss. Other strategies that have been associated with weight maintenance include higher dietary protein intake—which may increase satiety and possibly increase overall energy expenditure—and adhering to a diet that aligns with a patient’s food preferences [105–108].
Bariatric Surgery
For a subset of bariatric surgery patients, lifestyle change may not be sufficient to prevent weight regain and worsening of obesity-related comorbidities like T2DM. For this subset of patients, pharmacotherapy may attenuate weight regain and manage hyperglycemia [109, 110]. Several AOMs, including phentermine (alone or with topiramate), sodium–glucose cotransporter 2 inhibitors, and GLP-1 agonists have been shown to promote weight loss and reduction in hemoglobin A1C in patients with T2DM after bariatric surgery [111–116]. These therapies should be considered in patients who have not achieved goal weight reduction or are experiencing weight regain after bariatric surgery.
Weight Loss Maintenance—Final Thoughts
For clinicians, it is important to acknowledge that weight management is a lifelong challenge and to be prepared to assist patients with addressing small weight gains before they become larger ones, emphasizing the above strategies.
Clinical Case Resolution
This patient with T2DM and obesity has several possible options for AOM therapy. His current GLP-1 agonist, semaglutide 1.0 mg weekly, provides some weight loss benefit (−6.0 kg in SUSTAIN FORTE) [117] but there are AOMs that could be added to his regimen for greater weight loss potential. For instance, recently available semaglutide 2.0 mg weekly for T2DM has slightly greater weight loss than 1.0 mg weekly (−6.9 kg vs −6.0 kg, respectively) [117]. This dose increase could be considered to augment his weight loss, although it is not clear from available trial data how likely it is that he might reach his ≥10% weight loss goal. You check with your pharmacist, who finds the patient’s insurance will not cover semaglutide 2.4 mg or a switch to tirzepatide.
In this scenario, a good option would be phentermine/topiramate, which has been shown to result in 7% to 11% weight loss [47]. This patient does not have a history of CVD or hypertension. His depression is well controlled and he denies suicidal thoughts. You decide to start him on phentermine/topiramate ER 7.5/46 mg daily. You discuss with the patient that AOM therapy should be done in conjunction with a comprehensive lifestyle intervention, focusing on sustainable physical activity with a goal of at least 150 minutes per week, as well as dietary changes that increase protein and fiber intake and decrease processed food. After discussions with the patient, he decides to try Weight Watchers.
You also re-evaluate his antidepressant paroxetine, which is associated with weight gain and anticholinergic effects that may contribute to his lower energy levels. More activating antidepressants associated with weight loss include bupropion and fluoxetine. While bupropion has more data regarding weight loss, it has sympathomimetic properties similar to phentermine that may increase the risk of hypertension, palpitations, and tachyarrhythmias when taken together. You decide to switch the patient from paroxetine to fluoxetine.
At 16-week follow-up he has achieved an additional 4% body weight loss on phentermine/topiramate ER 7.5/46 mg daily (−8.2% overall since starting semaglutide for glycemic control), so you continue at this dose. At 6-month follow-up, he reports he has lost 7 kg (−9.3%) body weight. You congratulate him on his success and encourage continued sustainable lifestyle changes, emphasizing the importance of finding an exercise program that he enjoys and can continue long-term. You schedule follow-up in another 6 months.
Conclusion
Exciting recent advancements in the field of AOMs brings hope for a new paradigm in medical obesity management. With the emergence of more effective medications, we have proposed an updated goal of ≥10% weight loss as the threshold to gauge success with AOM treatment. Further work is needed to make these drugs more affordable and easily implementable, and to study their use over longer time periods. With further research, clinician education, and advocacy regarding AOM accessibility, the above paradigm can become widely implemented.