In this post I link to and excerpt from The IBCC chapter & cast – Clostridioides Difficile [Link to the chapter] [Link to the podcast]. July 4, 2019 by Dr. Josh Farkas.
Dr. Farkas gives us direct links to each part of his Clostridioides Difficile chapter:
CONTENTS
Here are excerpts from the chapter:
presentation & diagnosis
broad range of severity
- Mild disease: diarrhea only.
- Moderate disease: diarrhea plus colitis, causing fever and pain.
- Severe disease: toxic megacolon*, septic shock, peritonitis, multiorgan failure.
*See Toxic Megacolon-Background from emedicine.medscape.com. Updated: Mar 01, 2018
Author: Brian Lin“The dreaded complication of TM (TC) is perforation, even in the absence of colonic dilatation. Numerous studies have demonstrated that classic physical signs of peritonitis are absent in the majority of patients with free perforation, possibly because of the effects of steroids. (See Prognosis and Presentation.)”
when to consider testing for C. difficile
- (1) Any hospitalized patient with new-onset diarrhea (unless there is an obvious cause, such as over-enthusiastic laxative use).
- (2) Any patient admitted to the hospital for acute diarrhea (C. difficile is now being seen in patients from the community without any recent antibiotic use).
- Risk factors include inflammatory bowel disease, healthcare exposure, cancer chemotherapy, and other chronic comorbidities (e.g. liver or kidney disease).
- (3) Patients with clinical/imaging evidence of colitis or toxic megacolon.
- (4) Any patient you notice developing a profoundly elevated white blood count that you cannot explain.
- C. difficile patients may have markedly elevated white blood counts, which are disproportionate to their degree of clinical illness.
prevention
#2. know which antibiotics cause C. Difficile
- Slimmings 2014 performed a meta-analysis to determine the odds ratio (OR) of getting C. Difficile following treatment with different antibiotics. This allows antibiotics to be arranged roughly in order of increasing risk. The most problematic antibiotics are generally fluoroquinolones, clindamycin, and advanced-generation cephalosporins (29462280).
- Tetracyclines (OR 0.77)
- Macrolides (OR 1.14)
- Aminoglycosides (OR 1.17)
- Penicillins (OR 1.23)
- G1-cephalosporins (OR 1.36)
- Piperacillin-tazobactam (OR 1.5)
- Fluoroquinolones (OR 1.66)
- Trimethoprim-sulfamethoxazole (OR 1.78)
- Carbapenems (OR 1.84)
- G4-cephalosporins (OR 2.14)
- Clindamycin (OR 2.86)
- G3-cephalosporins (OR 3.2)
- A few comments:
- Macrolides and tetracyclines have very low risk, making them good choices for COPD exacerbation (where a broad spectrum antibiotic isn’t needed).
- There is a substantial difference between various cephalosporins, so when possible a first-generation cephalosporin is preferred.
- Piperacillin-tazobactam has the lowest C. difficile rate of any broad-spectrum antibiotic. Shortage of piperacillin-tazobactam has been associated with doubling the rate of C. difficile (Alston 2004).
#3. excellent hygiene
- One meaning of “first do no harm” is to try to avoid C. difficile transmission.
- C. difficile spores aren’t sterilized by ethanol-based hand cleansers. Better approaches include the use of gloves and traditional hand-washing.
- Any clothing that isn’t laundered daily can transmit C. difficile spores (e.g. white coats, ties, vests). Ideally, all external clothing in the ICU should be hospital-issued and cleaned daily (e.g. scrubs and anesthesia “warm-up” jackets).
- Hospital-laundered scrubs are cleaner than home-washed scrubs (hospitals clean them industrially at high temperatures).
#4. isolation of patients with C. Difficile
- Patients with known or suspected C. Difficile should be placed under contact isolation. Signage should remind staff to wash their hands (rather than using ethanol-based hand cleansers).
- Continuing contact isolation until hospital discharge could help limit the spread of C. difficile (carriage of spores may persist for weeks).
lab testing
the menu of various lab tests
glutamate dehydrogenase antigen
- Present in all strains of C. difficile (both toxigenic strains & benign, non-toxigenic strains).
- May be used as a screening test (sensitive for clinical infection, but nonspecific).
PCR for C. difficile toxins A or B
- More specific than glutamate dehydrogenase (found only in toxigenic strains of C. difficile).
- Not entirely specific for clinical infection (the bacterium may simply be a colonizing organism).
Enzyme immunoassay (EIA) to detect C. difficile toxin A or B
- Most specific for clinical disease (bacteria which are actually secreting toxin in the colon).
- Less sensitive (~80%), but >95% specific.
- Toxin breaks down rapidly, so samples should be refrigerated or transported rapidly to the lab.
the strategy of using different tests
Every hospital will have a preferred diagnostic strategy for detecting C. difficile. Discuss with your infectious disease colleagues which assay your hospital uses and the best strategy for its application. Some potential algorithms are as follows:
- (1) Accept only unformed stool for testing (to screen out patients with low pre-test probability). Then use the PCR to test for C. difficile toxin A or B. If positive, assume that the patient has clinical C. difficile and treat as such.
- (2) Start with glutamate dehydrogenase or PCR as a screening test. If positive, then use an ELISA assay to detect toxins as a confirmatory test.
potential pitfalls
- A patient on treatment for C. difficile may have a false-negative toxin assay (due to suppression of bacterial replication).
- After successful treatment for C. difficile, patients may continue to have positive tests (especially the PCR test). For this reason, testing to confirm cure is not recommended.
colonoscopy
not generally helpful (especially in the critically ill)
- Diagnosis can usually be secured based on stool analysis with or without CT scanning of the abdomen.
- Colonoscopy of a patient with severe colitis may risk perforation, so this should be avoided if possible.
- Endoscopic visualization (+/- biospsy) may be of benefit in situations with an unusually broad differential diagnosis:
- Infectious colitis vs. inflammatory bowel disease
- Possible immune checkpoint-mediator induced colitis
- If performed, the study should probably be limited to an unprepped sigmoidoscopy with minimal insufflation of air into the colon (especially if the CT scan shows involvement of the sigmoid colon).
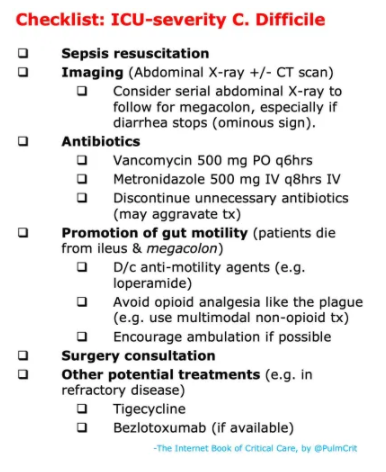
imaging
plain film for megacolon
- There should be a low threshold for abdominal X-ray to screen for toxic megacolon (e.g. in patients with significant abdominal tenderness or distension).
- Dilation > 6 cm is worrisome for the possibility of impending perforation (especially if associated with other markers of severe disease).
- May be repeated in a serial fashion to follow colonic distension.
CT scan
- Allows for identification of toxic megacolon, as well as other complications (e.g. perforation).
- Better study than abdominal X-ray, but be careful – patients may evolve rapidly. Absence of megacolon at one point in time doesn’t mean this cannot evolve over time.
For details on Risk Stratification, Treatment, and Other Potential Therapies, please see the Chapter.