The Pediatric Guidelines from the Surviving Sepsis Campaign: Considerations for Care is a 47 minute video. It is a review of the pediatric part of Reference (5), PEDIATRIC CONSIDERATIONS IN SEVERE SEPSIS (Table 9), pp 613 to 619 and is an outstanding brief summary of the ACCM-PALS guidelines. Reference (5) is Surviving Sepsis Campaign: International Guidelines for Management of Severe Sepsis and Septic Shock: 2012.
0:49: The speaker on this video is Dr. Margaret M. Parker. Dr. Parker is the director of the Pediatric Intensive Care Unit, Stony Brook Long Island Children’s Hospital, Stony Brook, NY and is a past president of the Society for Critical Care Medicine (SCCM).
Please see references (1, 2, and 3) in the Resources section from Stony Brook for a very useful Pediatric Sepsis screening tool (1), a set of adult and pediatrics sepsis protocols (2), and a pediatric sepsis order set (3).
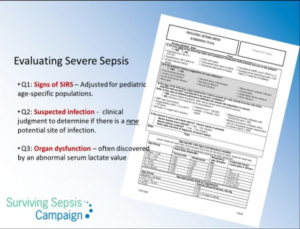
Finally, the big challenge for front-line clinicians (office, urgent care, and emergency care physicians and for ward nurses) is diagnosing sepsis early and reliably. So, here is the [Pediatric] Severe Sepsis Screening Tool from Stony Brooke (Reference 1):
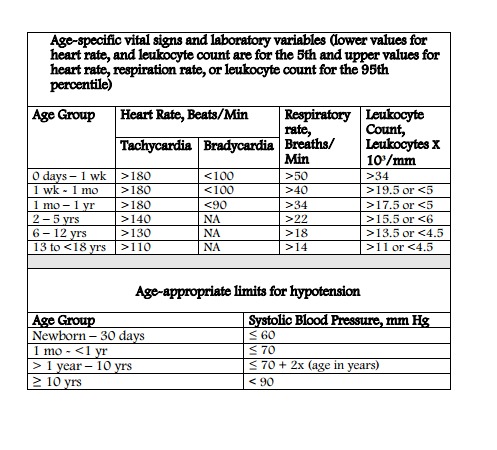
And below are the pediatric age specific vital signs to be used with the above form.
7:10: In children with severe sepsis the lactate level is much less often elevated than in adults and so is not as routinely used in pediatrics as it is in adults, Dr. Parker says. [However, it is worth noting that the Stony Brook [Pediatric] Severe Sepsis Screening Tool referenced above requires an explanation if serum lactate level is not ordered.]
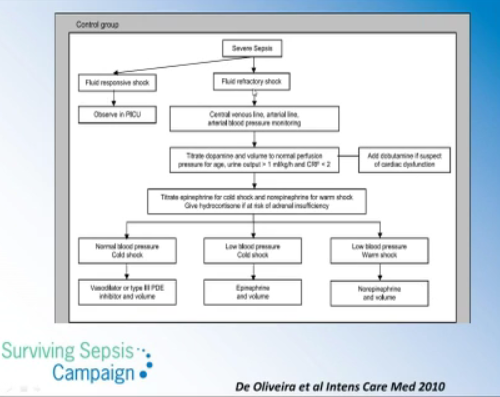
The above slide incorrectly references the article. I believe the correct reference is (4) in the Resources section.
Dr. Parker discusses the article referenced above through 16:35. (4) For primary care pediatricians (Peds, FPs, EMs, and NPs) whose critical responsibility is to promptly recognize and provide appropriate initial management of Pediatric Sepsis, this article is of limited significance, I believe.
Reference (5), PEDIATRIC CONSIDERATIONS IN SEVERE SEPSIS (Table 9), pp 613 to 619 is an outstanding brief summary of the ACCM-PALS guidelines which is worth reviewing at this time.
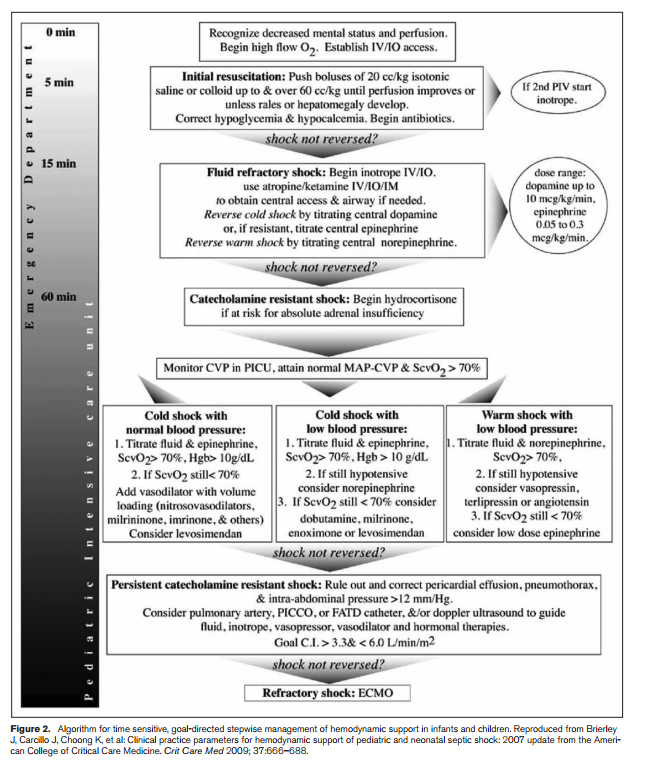
17:25: The slide (below) is Figure 2 of reference (5).
“We suggest beginning peripheral inotropic support until central venous access can be attained in children who are not responsive to fluid resuscitation (grade 2C).” (5, p 617)
“In the initial resuscitation phase, inotrope/vasopressor therapy may be required to sustain perfusion pressure even when hypovolemia has not yet been resolved. Children with severe sepsis can present with low cardiac output and high systemic vascular resistance, high cardiac output and low systemic vascular resistance, or low cardiac output and low systemic vascular resistance shock (555). A child may move from one hemodynamic state to another. Vasopressor or inotrope therapy should be used according to the hemodynamic state (555). A child may move from one hemodynamic state to another. Vasopressor or inotrope therapy should be used according to the hemodynamic state (555). Dopamine refractory shock may reverse with epinephrine or norepinephrine infusion.” (5, p 617)
From p 617 of Reference (5): “Children normally have a lower blood pressure than adults, and a fall in blood pressure can be prevented by vasoconstriction and increasing heart rate. Therefore, blood pressure alone is not a reliable endpoint for assessing the adequacy of resuscitation. However, once hypotension occurs, cardiovascular collapse may soon follow. Thus, fluid resuscitation is recommended for both normotensive and hypotensive patients in hypovolemic shock (542-554). Because hepatomegaly and/or rales occur in children who are fluid overloaded, these signs can be helpful signs of hypervolemia. In the absence of these signs, large fluid deficits can occur, and initial volume resuscitation can require 40 to 60 mL/kg or more; however, if these signs are present, then fluid administration should be ceased and diuretics should be given. Inotrope infusions and mechanical ventilation are commonly required for children with fluid refractory shock.”
After reviewing the above slide, please remember that additional quotes directly from reference 5 concerning vasopressors and inotropes are available above at 17:25.
Consider adding quotes about ECMO from Guidelines
When to Intubate the Septic Pediatric Patient: “Inotrope infusions and mechanical ventilation are commonly required for children with fluid-refractory shock.” Reference (5), p 617,
Lung protective strategies during mechanical ventilation are similar to those in adults: use a smaller tidal volume and keep the plateau pressure to less than 30.
For information on “Sedating the Intubated Patient” see reference (6) from Open Pediatrics.
Resources:
(1) [Pediatric] SEVERE SEPSIS SCREENING TOOL accessed from the Surviving Sepsis Campaign Protocols and Checklists page where it is called the Pediatric ICU Screening Tool – Stony Brook.
(2) Stony Brook Medicine Severe Sepsis/Septic Shock Recognition and Treatment Protocols (Full Text PDF) was published Aug 9, 2013 and covers both adult and pediatric cases. This was accessed from the Surviving Sepsis Campaign Protocols and Checklists page.
(3) PEDIATRIC ACUTE SEPSIS: PHYSICIAN’S ORDERS–Stony Brook Medical Center (Full Text PDF) accessed from the Surviving Sepsis Campaign Protocols and Checklists page.
(4) ACCM/PALS haemodynamic support guidelines for paediatric septic shock: an outcomes comparison with and without monitoring central venous oxygen saturation, 2008. Intensive Care Medicine. [I believe this is the correct reference for the article referenced on the slide as 2010].
(5) PEDIATRIC CONSIDERATIONS IN SEVERE SEPSIS (TABLE 9), pp. 613 to 619 of the Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012 [PubMed Abstract] [Full Text PDF] . Crit Care Med. 2013 Feb;41(2):580-637. doi: 10.1097/CCM.0b013e31827e83af.
(6) Sedating the Intubated Patient [Text] [Video], Dr. Monica Kleinman, Open Pediatrics. These resources require free registration at the site.
(7) Hemodynamic support in fluid-refractory pediatric septic shock [PubMed Abstract] [Full Text HTML] [Full Text PDF]. Pediatrics. 1998 Aug;102(2):e19. Ceneviva G et al.
(8) Bedside echocardiography is useful in assessing children with fluid and inotrope resistant septic shock [PubMed Abstract] [PMC Full Text]. Indian J Crit Care Med. 2013 Jul-Aug; 17(4): 224–230. doi: 10.4103/0972-5229.118426
(9) Multimodal monitoring for hemodynamic categorization and management of pediatric septic shock: a pilot observational study* [PubMed Abstract]. Pediatr Crit Care Med. 2014 Jan;15(1):e17-26. doi: 10.1097/PCC.0b013e3182a5589c.