In this post I link to and excerpt from #226 Kidney Boy on Acute Kidney Injury: Myths & Musings [Link is to the podcast and show notes. JULY 20, 2020 By DR CYRUS ASKIN: AKI TIPS AND TRICKS FROM JOEL TOPF MD, KASHLAK’S CHIEF OF NEPHROLOGY.
Summary
Get a grip on acute kidney injury (AKI) with Dr. Joel Topf (AKA @kidney_boy), Kashlak’s Chief of Nephrology! We’ve put together an AKI highlight reel – focusing on practical tips and tricks to help you identify, diagnose and manage AKI, plus how to recognize AIN and random myths and musings on vancomycin, NSAIDS, contrast nephropathy, and the risk of NSF from gadolinium.
Here are excerpts:
Acute Kidney Injury (AKI) Pearls
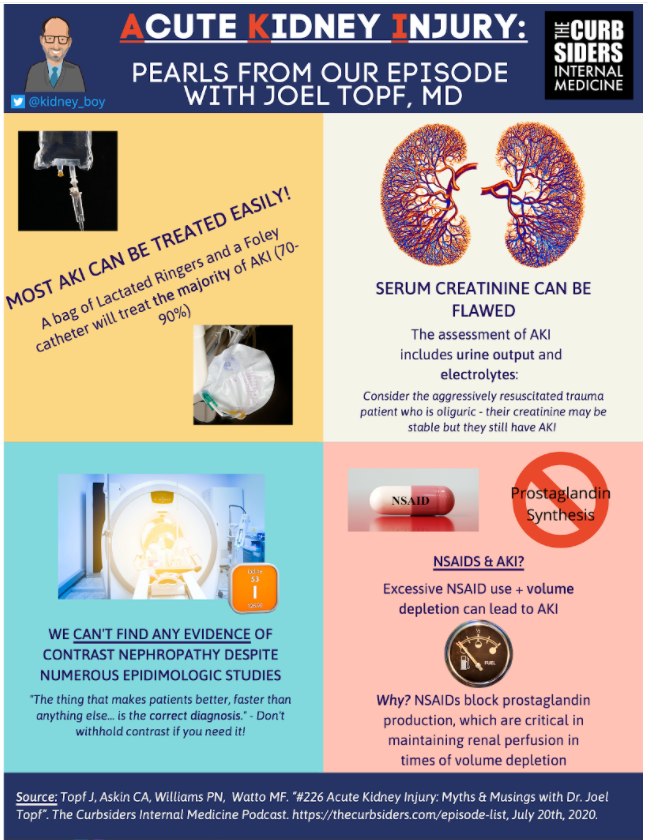
- The KDIGO definition of AKI is helpful for research… not so helpful from a clinical/practical standpoint
- sCr is a somewhat flawed / lagging indicator of renal function, keep an eye out for electrolyte disturbances and oliguria / anuria
- The majority of AKI can be fixed by “a bag of LR & a Foley Catheter”… tincture of time may be all that’s necessary in many other cases
- History is king: ask about NSAIDs, cocaine for rhabdo, antibiotics for sepsis/AIN, PPI for AIN, diarrhea, vomiting, ACEi, hypotension, new diuretics.
- Urine electrolytes and urine eosinophils are often unhelpful, may lead to more confusion than anything else
- A good chart biopsy can be very helpful in AKI: identify nephrotoxins and avoid them
- Acute interstitial nephritis (AIN) is a diagnosis of exclusion with biopsy generally reserved for cases where steroids are being considered
- Don’t fear contrast in AKI! There is no signal in the literature suggesting that contrast causes AKI
- Save the renal ultrasound for part two of your AKI work up – consider an in-and-out catheterization and bladder scan if obstruction is on the differential
Dr. Topf underlines the importance of urine output (UOP) – often overlooked in favor of sCr. He uses the example of a patient being aggressively resuscitated who does not have an appreciable elevation in sCr but does have anuria or oliguria. Resuscitation can “dilute” the sCr.
- This is not seen in volume-overloaded heart failure patients suffering from cardiorenal syndrome – this condition is almost always associated with a rise in sCr.
Buckets of AKI
- Dr. Topf encourages folks to think of AKI in terms of pre-renal, intra-renal and post-renal etiologies:
PRE-RENAL
Pre-Renal: Inadequate renal perfusion due to volume deficiency (vomiting, diarrhea, decreased PO intake, bleeding, non-hemorrhagic shock) or heart-failure (volume overload with venous congestion resulting in poor renal perfusion)
INTRA-RENAL
Intra-Renal: Acute Tubular Necrosis – the most common, acute interstitial nephritis / drug toxicities (vancomycin, aminoglycosides, etc.), glomerulonephritis, vasculitis (Makras & Spanou, 2016)
- See the Clinical Problem Solver’s schema and discussion of intrinsic renal failure here
POST-RENAL
Post-Renal: Malignancy (ex: cervical cancer in women, prostate cancer in men), BPH, a kidney stone in a solitary kidney or retroperitoneal fibrosis
KEY INSIGHT FOR MANAGING AKI
Somewhere between 70 & 90% of AKI is pre-renal or post-renal in etiology, meaning that fluid resuscitation and removal of the obstruction (i.e. a Foley) will resolve 70-90% of AKI! (Dr. Topf & Kaufman et al, 1991)
For intrinsic AKI, which is generally ATN, Dr. Topf says the treatment is also straight forward:
- Time
- Avoid nephrotoxins
- Avoid hypotension
- More time
AKI WORKUP
Dr. Topf’s initial approach to a pathologic elevation in sCr – it doesn’t end with a high creatinine!
- Is the patient non-oliguric, oliguric or anuric?
- Was this patient hypotensive?
- Does this patient have severe heart failure and possible cardiorenal syndrome?*
- Was this patient on NSAIDs? Vancomycin? Other potential culprits?
- Does the patient have significant electrolyte derangements?
- Do they have a significant acid-base derangement?
*See Link To Dr. Rola’s Grand Rounds “POCUS Assessment Of Venous Congestion” With Link To An Additional Resource
Posted on November 2, 2020 by Tom Wade MDTo FeNa or not to FeNa [Fractional Excretion of Sodium – Link is to MDCalc]: Depends on what you are looking for, may lead to discordant information
- Ex: AKI associated with rhabdomyolysis is a cause of intrarenal AKI presenting with a low FeNa while most other intrinsic renal diseases should present with an elevated FeNa. (Corwin et al., 1984)
If you suspect the patient’s AKI is unlikely due to intrinsic renal disease – it is much faster to give fluid / assess for obstruction than to get urine electrolytes and other studies that take time and could lead to confounding results
HYALINE CASTS?
Dr Topf: Can be seen in dry patients (urinalysis with high specific gravity, acidic urine) but generally, you’ll have assessed the patient and determined they are dry before looking at their urine for casts
AKI in the Previously Healthy Patient:
Significant exercise history? Consider rhabdomyolysis. (Chavez et al. & Cabral et al.)
- Step 1: talk to the patient – See if their history is consistent with rhabdomyolysis
- The cornerstone of treating rhabdomyolysis is fluid resuscitation to increase renal perfusion – Dr. Topf recommends ~200cc/hr of LR
- Urine dipstick will be POSITIVE for blood, microscopy shows NO BLOOD – this is due to release of heme containing myoglobin from muscle tissue and the dipstick detecting free heme
VOLUME STATUS
Heat? Exercise? Poor PO-intake? Consider volume depletion and pre-renal azotemia.
- Dr. Topf recommends that, in an otherwise healthy patient who we suspect has pre-renal azotemia, it’s reasonable to give them 3-4 liters of fluid and recheck a sCr
- The sCr should show a true improvement, assuming the patient has appropriate urine output (i.e. the volume should not dilute their sCr but instead truly improve renal perfusion)
- Volume status exam: One of the most challenging things in medicine, but important! (van der Mullen et al.)
- Dr. Topf recommends we favor “over-resuscitation” as young patients are at low-risk of suffering from clinically significant volume-overload, but the likelihood that they respond well to volume is high
- Particularly important to remember in cases of rhabdomyolysis, as discussed above
NSAIDS? CONSIDER NSAID-ASSOCIATED AKI.
- Talk to the patient!
- NSAIDs block prostaglandins. During volume depletion, prostaglandins help ensure adequate renal blood flow. NSAIDs, thus, can reduce the ability of the kidneys to maintain adequate perfusion during times of volume depletion, resulting in AKI. (Faubel & Topf, 1999; Imig, 2002; Zhang, 2017)
- Treatment: volume resuscitation, hold NSAIDs, hold other nephrotoxins
YOUNGER PATIENTS? OBSTRUCTION IS GENERALLY LOW ON THE DIFFERENTIAL.
- Dr. Topf tells us that these patients do, occasionally need RRT, however, they generally have an excellent prognosis if they don’t have prior kidney disease and/or have few-or-no medical comorbidities
AKI In the Critically Ill:
In Dr. Topf’s experience, 9/10 of these patients are suffering from acute tubular necrosis (ATN)
- These patients are almost always adequately resuscitated (unlikely to be pre-renal azotemia)
- Obstruction is unlikely – almost all these patients have foley catheters
- Bloody intubation? Think about ANCA vasculitis and pulmonary-renal syndrome
- Did patient recent vancomycin? And pip-tazo?
- Could this be AIN?
ACUTE INTERSTITIAL NEPHRITIS (AIN)
- A very difficult diagnosis – so many other usual suspects normally at play (vancomycin, pressors, contrast, etc.)
- AIN should be considered a diagnosis of exclusion, in many cases
- Urine eosinophils? In some cases, more common in ATN than AIN, certainly no utility in differentiating between different forms of intrinsic kidney injury (Lusica et al.) (Muriithi et al.)
- Takeaways: (Moledina & Perazella)(Fernandez-Juarez et al.)
- White blood cells (pyuria) and red blood cells are reliably found in AIN, per Dr. Topf
- PPIs, certain antibiotics and NSAIDs are common causes
- AKI + Fever + Rash (classic triad) comes from beta-lactam literature
- Treatment: remove offending agent, consider getting a biopsy, consider using steroids
WHO NEEDS RENAL REPLACEMENT THERAPY?
- Does it make sense to start RRT earlier in a patient’s course? Well, the AKIKI trial would suggest that there is no benefit to this strategy, and you may save resources by going with a delayed approach to RRT. (Gaudry et al.)
- Based on this data, Dr. Topf does not recommend early dialysis – instead, he will look for dangerous electrolyte abnormalities or volume overload to prompt initiation of RRT
- Update! – the START-AKI trial just published in NEJM also found no mortality benefit with early dialysis.