The appropriate interpretation of medical tests critically depends on only ordering a test when the test is indicated based on the physician’s estimation of pre-test probability.
The physician must take a careful medical history, including a complete review of systems, and a careful and appropriate physical exam to determine the pretest probability of a connective tissue disease.
For help on rheumatologic pretest probability, please see Link To Comprehensive Rheumatology Review Of Systems and History Form From The American College Of Rheumatology
Posted on November 9, 2018 by Tom Wade MD
In this post I link to and excerpt from Antinuclear antibodies: When to test and how to interpret findings [Link is to the PDF], J Fam Pract. 2015 January;64(1):E5-E8. By Habib U. Rehman, MB, FRCPC, FRCPI, FRCP (Glas), FACP.
The author states in his introduction:
Order ANA assays only when clinical features suggest a
connective tissue disorder. Let ANA immunofluorescent
patterns direct additional testing decisions.Antinuclear antibodies (ANA) are a spectrum of autoantibodies that react with various nuclear and cytoplasmic components of normal human cells. Their detection is important in the diagnosis of some connective tissue diseases (CTD)—eg, systemic lupus erythematosus (SLE), Sjögren’s syndrome (SS), scleroderma, polymyositis, or mixed connective tissue disease (MCTD). Unfortunately, ANA tests are often used indiscriminately in daily clinical practice.1
> When clinical probability of connective tissue
diseases is low, the presence of ANA in the serum can
indicate chronic infection, neoplasm, or advancing age.> Think systemic lupus erythematosus when a
patient has a photosensitive butterfly rash, arthralgia, pleuritic chest pain, fever, or urine sediment consistent with nephritis.
Here is his practice recommendation:
Practice recommendation
Reserve antinuclear antibody testing for instances of clinically suggestive connective tissue diseases (CTD) and for assessing
CTD prognosis. It can also be useful in monitoring disease progression. CStrength of recommendation (SOR)
A – Good-quality patient-oriented evidence
B – Inconsistent or limited-quality patient-oriented evidence
C – Consensus, usual practice, opinion, disease-oriented
evidence, case series
Here are excerpts:
When is ANA testing warranted?
Indiscriminate use of ANA testing can yield positive results that
falsely point to CTD in a high proportion of patients and thereby
lead to further inappropriate testing and errant management decisions. To wit: The presence of ANA in the serum can be associated with any number of factors, such as genetic predisposition (eg, through histocompatibility locus DR3), environmental agents (viruses, drugs), chronic infections, neoplasms, and advancing age.1 Therefore, the test should not be ordered in a patient with low pretest probability of CTD. Moreover, higher titers of ANA are more clinically significant than lower titers. In one multicenter study, 31.7% of healthy individuals were ANA-positive at a serum dilution of 1:40, but only 5% were ANA-positive at a dilution of 1:160.2What is the clinical significance of different immunofluorescent patterns?
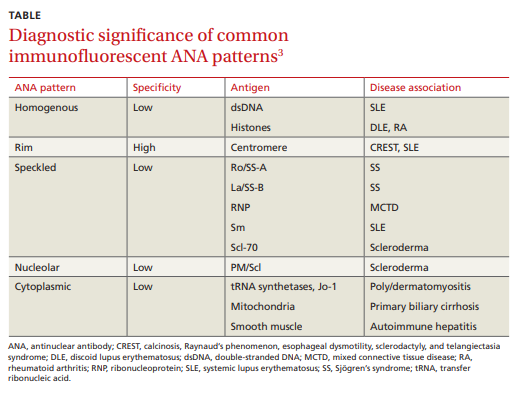
Immunofluorescent ANA testing not only determines if such
antibodies are present in a patient’s serum but also reveals informative antibody patterns. Five distinct patterns of fluorescence are possible and can help differentiate between various CTDs (TABLE3):1. Homogenous, in which the entire nucleus fluoresces, is
seen in SLE and discoid lupus erythematosus (DLE).
2. Rim, in which the nuclear perimeter fluoresces, is seen
most often in CREST (calcinosis, Raynaud’s phenomenon, esophageal dysmotility, sclerodactyly, and telangiectasia) syndrome and SLE.
3. Speckled, in which the nucleus fluoresces in a speckled pattern, can be seen in a variety of CTDs, including
Sjögren’s syndrome, MCTD, SLE, and
scleroderma.
4. Nucleolar, in which the nucleolus fluoresces, is associated with scleroderma.
5. Cytoplasmic, in which fluorescence occurs outside the nucleus, typically occurs with poly/dermatomyositis,
primary biliary cirrhosis, or autoimmune hepatitis.
What is the next step if ANA is positive?
A positive ANA result warrants additional studies to identify specific autoantibodies suggested by the fluorescence pattern and by a patient’s signs and symptoms.
Following up diagnostic clues
Most systemic autoimmune diseases have a highly characteristic profile of autoantibodies to cellular antigens. A patient’s clinical features and ANA fluorescence pattern should direct additional testing.
Pages E6 + E7 of the PDF give suggestions on further antibodies to order based on the patient’s clinical features and ANA flourescence pattern.