In this post, I link to and excerpt from Core EM‘s March 4, 2021 podcast and show notes, Episode 181.0: Subarachnoid Hemorrhage.
In addition to the above outstanding resource, consider reviewing Radiopaedia’s resources on subarachnoid hemorrhage [Link is to SAH search-314 results found].
Please review the excellent Radiopaedia article, Subarachnoid hemorrhage, by Dr Mostafa El-Feky and Assoc Prof Frank Gaillard et al. The following is from this Radiopaedia article:
Subarachnoid hemorrhage (SAH) is a type of extra-axial intracranial hemorrhage and denotes the presence of blood within the subarachnoid space.
Epidemiology
Patients tend to be older middle age, typically less than 60 years old 2. Subarachnoid hemorrhage accounts for 3% of stroke and 5% of stroke deaths 2.
Risk factors
Risk factors include 2:
- family history
- hypertension
- heavy alcohol consumption
- abnormal connective tissue
- female gender: ~1.5x baseline risk 10
- African descent: ~2x baseline risk ref
- Japanese or Finnish descent 12
Clinical presentation
Patients typically present with a thunderclap headache, described as a sudden-onset headache that is the worst headache of their life. It is often associated with photophobia and meningism. Focal neurological deficits often present either at the same time as a headache or soon thereafter 2.
In a substantial number of patients (almost half 2), it is associated with collapse and decreased or loss of consciousness, even in those patients who subsequently regain consciousness and have a good grade.
Patients can be graded into five groups based on their clinical presentation, using the commonly employed Hunt and Hess grading system, which is predictive of outcome.
Pathology
Three distinct patterns of subarachnoid hemorrhage have been described each with its own etiology and treatment/prognostic implications 4:
- suprasellar cistern with diffuse peripheral extension
- perimesencephalic and basal cisterns
- isolated cerebral convexity
Etiology
Causes include 1:
- trauma
- spontaneous
- ruptured berry aneurysm: 85% 1
- perimesencephalic hemorrhage: 10% 4
- arteriovenous malformation
- cerebral amyloid angiopathy
- ruptured mycotic aneurysm
- reversible cerebral vasoconstriction syndrome
- dural arteriovenous fistula
- spinal arteriovenous malformation
- venous infarction 1
- intradural arterial dissection
- pituitary apoplexy
- sympathomimetic drugs (eg. cocaine)
- cerebral vasculitis 6
- anticoagulation therapy 9
Radiographic features
Although MRI is thought to be more sensitive, CT is frequently performed first due to wider availability. As well as being more sensitive to hemorrhage, MRI has greater sensitivity to the wide range of causative lesions.
A description of the radiographic features of each causative underlying lesion is clearly beyond the scope of this article; these are discussed separately (see above).
CT
The sensitivity of CT to the presence of subarachnoid blood is strongly influenced by both the amount of blood and the time since the hemorrhage.
The diagnosis is suspected when a hyperdense material is seen filling the subarachnoid space. Most commonly this is apparent around the circle of Willis, on account of the majority of berry aneurysms occurring in this region (~65%), or in the Sylvian fissure (~30%) ref needed.
Small amounts of blood can sometimes be appreciated pooling in the interpeduncular fossa, appearing as a small hyperdense triangle, or within the occipital horns of the lateral ventricles 5.
Subarachnoid hemorrhages are grouped into four categories according to the amount of blood on unenhanced CT by the Fisher scale. This scale has been updated to the modified Fisher scale, which more accurately correlates the risk of vasospasm.
MRI
MRI is sensitive to subarachnoid blood and is able to visualize it well in the first 12 hours typically as a hyperintensity in the subarachnoid space on FLAIR 3.
Susceptibility weighted sequences are also exquisitely sensitive to blood products.
MR angiography and MR venography are also able to detect a causative aneurysm or another source of bleeding, although in general MRI suffers from poor availability (compared to CT), longer scan times, and greater difficulty in transferring and looking after patients who are often unstable and intubated.
In aneurysm-associated subarachnoid hemorrhage, diffusion weighted imaging may demonstrate early ischemic changes (within 0-3 days) in more than half of all patients 8. Additionally delayed ischemia detected on DWI, associated with vasospasm developing 4-21 days after ictus, may develop in about half of all patients 8.
DSA
Digital subtraction catheter angiography remains the gold standard for diagnosis and characterization of vascular abnormalities and in many centers, even if the causative lesion is identified on MRA or CTA and it is thought to require surgical management, a catheter study is carried out.
The benefit of DSA is two-fold:
- higher spatial resolution: better able to delineate small vessels and characterize vascular morphology (e.g. aneurysm neck and incorporation of adjacent vessels)
- temporal resolution: contrast can be seen to wash into and out of vascular malformations, giving important information in regards to the feeding vessels (e.g. arteriovenous malformations or dural arteriovenous fistulas)
Additionally, depending on the cause, endovascular therapy (e.g. aneurysm coiling) may be appropriate.
Treatment and prognosis and Differential diagnosis
Please see the Radiopaedia article for details on the above.
For an excellent discussion of traumatic subarachnoid hemorrhage, please see the excellent Radiopaedia article, Traumatic subarachnoid hemorrhage, by Dr Subhan Iqbal and Dr Henry Knipe et al. The following is excerpted from this Radiopaedia article:
Traumatic subarachnoid hemorrhage (tSAH) is a common injury, and trauma is the most common cause of subarachnoid hemorrhage (SAH) 5.
Epidemiology
Traumatic subarachnoid hemorrhage occurs in ~35% (range 11-60%) of traumatic brain injuries 1.
Pathology
Traumatic subarachnoid hemorrhage is more commonly seen in the cerebral sulci than in the Sylvian fissure and basal CSF cisterns 1. When in the basal cisterns, it has an affinity for the quadrigeminal cistern and ambient cistern 2. tSAH is also commonly seen adjacent to skull fractures and cerebral contusions 3.
The exact mechanism of tSAH remains uncertain although it is clear that a number of etiologies exist and these will determine, at least to a degree, the distribution of blood. Causes of tSAH include 4:
- direct extravasation of blood from an adjacent cerebral contusion
- arterial dissection
- direct damage to small veins or arteries
- sudden increase in intravascular pressures leading to rupture
Radiographic features
CT
CT of the brain is almost always the first scan obtained in the setting of trauma, often as part of a CT panscan.
Although the sensitivity of CT to the presence of subarachnoid blood is strongly influenced by both the amount of blood and the time since the hemorrhage, in the setting of trauma scans are almost always obtained early, often mere minutes or hours from head injury, making even small amounts of blood readily visible.
The distribution and amount of blood varies greatly depending on the underlying mechanism (see above) and from patient to patient.
Often a small amount of blood is seen filling a few sulci, sometimes with an adjacent cerebral contusion. Small amounts of blood can also sometimes be appreciated pooling in the interpeduncular fossa, appearing as a small hyperdense triangle, or within the occipital horns of the lateral ventricles.
Occasionally, and worrying for an underlying arterial dissection or an aneurysmal hemorrhage that preceded trauma, larger amounts of blood may be seen around the circle of Wilis and within the posterior fossa.
Treatment and prognosis
Traumatic subarachnoid hemorrhage has a better prognosis than aneurysmal SAH 2.
Complications
Differential diagnosis
It can be difficult to delineate from tSAH, particularly as in many instances the cause of head trauma may have been spontaneous subarachnoid (e.g. while driving).
The distribution of blood, particularly if closely related to cerebral contusions can suggest traumatic etiology, whereas extensive blood around the circle of Willis should prompt arterial imaging to exclude an aneurysm.
Distinguishing between aneurysmal subarachnoid hemorrhage and traumatic non-aneurysmal subarachnoid hemorrhage is not always possible, and the trauma may have in reality been precipitated by a spontaneous aneurysmal hemorrhage (e.g. while driving).
That having been said, there are helpful features in suggesting that subarachnoid hemorrhage is the result of trauma, rather than the reason for trauma. These features include:
- documented (witnessed) trauma not being preceded by a headache or loss of consciousness or seizure
- subarachnoid blood being relatively minor and associated with cerebral contusions
- subarachnoid blood located over the convexity of the brain rather than around the circle of Willis or posterior fossa
- location of subarachnoid blood deep to scalp hematoma or in a contrecoup distribution
The Core EM podcast and show notes linked to and excerpted today discuss only nontraumatic subarachnoid hemorrhage.
All that follows below is excerpted from Core EM‘s Episode 181.0: Subarachnoid Hemorrhage.
We discuss EM presentation, diagnosis, and management of [nontraumatic] subarachnoid hemorrhage.
Hosts:
Mark Iscoe, MD
Brian Gilberti, MD
Bree Tse, MDHunt-Hess grade and mortality (from Lantigua et al. 2015.)
Hunt-Hess grade Mortality (%) 1. Mild Headache 3.5 2. Severe headache or cranial nerve deficit 3.2 3. Confusion, lethargy, or lateralized weakness 9.4 4. Stupor 23.6 5. Coma 70.5
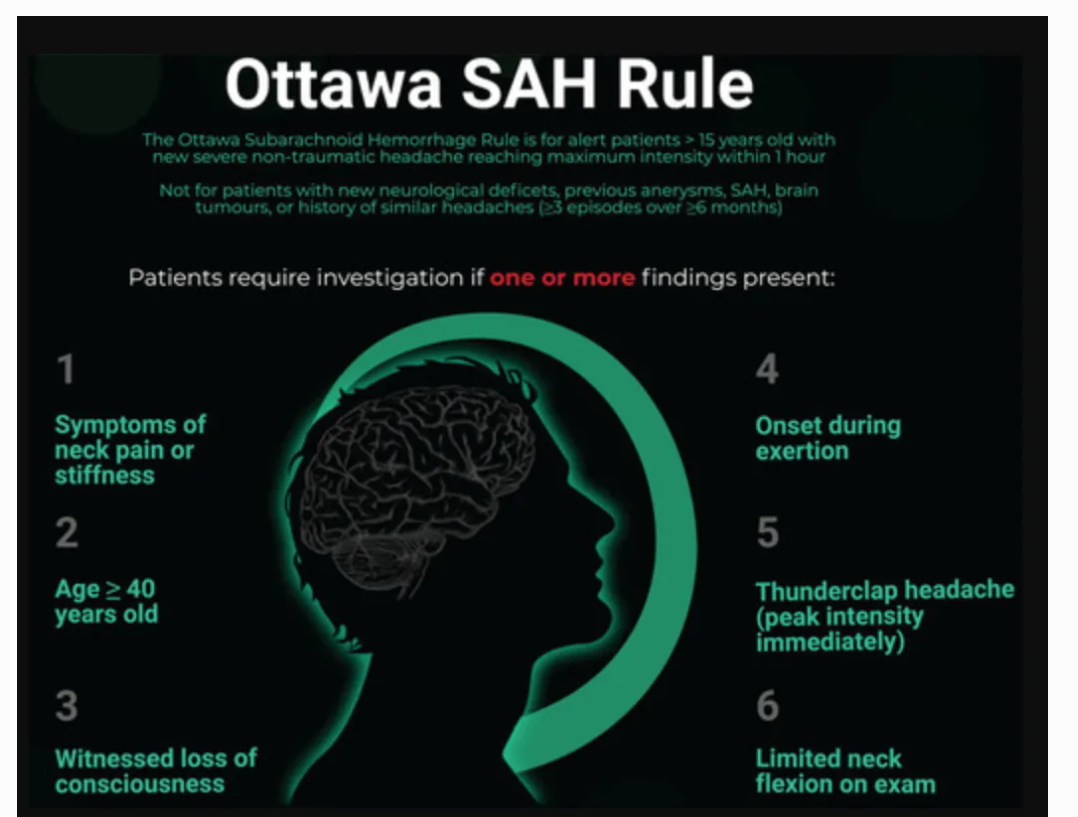
Ottawa Subarachnoid Hemorrhage Rule, and appropriate population for rule application (from Perry et al. 2017)
Apply to patients who are:
- Alert
- ≥ 15 years old
- Have new, severe, atraumatic headache that reached maximum intensity within 1 hour of osnet
Do not apply to patients who have:
- New neurologic deficits
- Previous diagnosis of intracranial aneurysm, SAH, or brain tumor
- History of similar headaches (≥ 3 episodes over ≥ 6 months)
SAH cannot be ruled out if the patient meets any of the following criteria:
- Age ≥ 40
- Symptom of neck pain or stiffness
- Witnessed loss of consciousness
- Onset during exertion
- “Thunderclap headache” (defined as instantly peaking pain)
- Limited neck flexion on examination (defined as inability to touch chin to chest or raise head 3 cm off the bed if supine)