In this post I link to and excerpt from The Curbsiders episode of May 1, 2020, #211 COVID Cakes: Remdesivir, Anticoagulation, Transmission [Link is to the podcast and show notes], by Dr. Emi Okamoto.
Here is the podcast:
Time Stamps
- 00:30 Intro, disclaimer
- 02:30 Picks of the Week
- 6:36 Remdesivir trial rundown
- 21:52 Thrombosis in COVID-19
- 29:50 Asymptomatic/Presymptomatic transmission
- 41:42 Quick takes
- 49:00 Outro
This Curbsiders podcast added following last minute update:
**Updates: 1) NIH Press Release about Remdesivir RCT with accelerated recovery from April 27, 2020. 2) Wang Y et al article from Lancet published April 29, 2020.**
And Dr. Josh Farkas gives us a thoughtful review of the Wang et al article from Lancet, PulmCrit- First placebo-controlled RCT on remdesivir for COVID-19
April 30, 2020 by Dr. Josh Farkas.
Here are the show notes on COVID and Thrombosis:
The first section of this episode reviews the article:
Grein, J et al. Compassionate Use of Remdesivir for Patients with Severe Covid-19. NEJM. April 10, 2020 [https://www.nejm.org/doi/full/10.1056/NEJMoa2007016]
For a step-by-step breakdown of the study, follow along with Dr Ganatra (@rbganatra) on his twitter learning thread, #HowIReadThisPaper.
I wasn’t sure how you follow tweetorial. I clicked on #HowIReadThisPaper above.
The click took me to Dr. Ganatra’s first tweet on the series of tweets (the tweetorial). When I clicked anywhere in the first tweet, I was taken to the entire thread of 27 tweets.
Bottom line of Dr. Ganatra’s analysis of the study was:
27/ Bottom line: Without a control group, it is impossible to tell whether the observed outcomes in this selected population are due to RDV. Selection bias limits both comparison to other cohorts and the generalizability of these results. Well-designed RCTs are needed. (End)
Basically, if I understand Dr. Ganatra’s conclusion, after he has carefully reviewed the study in his tweetorial, is that no conclusions can be drawn from the study due to the study’s numerous methodological flaws.
Here are more show notes from Curbsiders #211 – Thrombosis in COVID-19:
COVID and Thrombosis
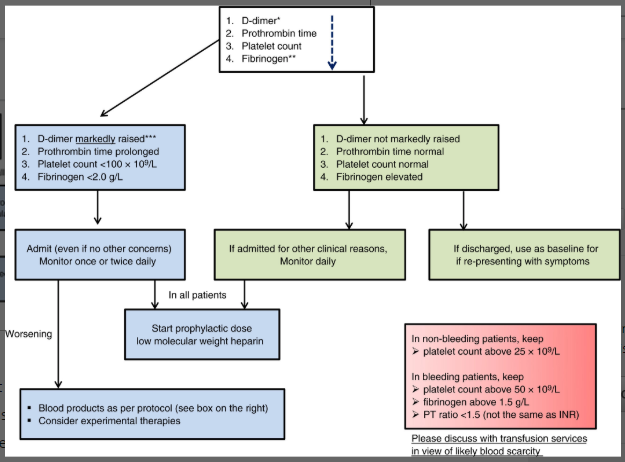
Thachil J et al. ISTH interim guidance on recognition and management of coagulopathy in COVID19. J Thromb Haemost. 25 March 2020 [https://onlinelibrary.wiley.com/doi/epdf/10.1111/jth.14810]
Flow chart and caption are from the above citation.
Tang, N et al. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 27 March 2020. [https://onlinelibrary.wiley.com/doi/abs/10.1111/jth.14817]
What is everyone’s hospital policy on this?
Anticoagulation policies vary widely, from prophylactic to treatment dosing for hospitalized patients, depending on institution. We recommend that you follow your institutions guidelines until we have more data to guide practice.
International Society of Thrombosis and Hemostasis Interim Guidelines Review
- Check D-dimer, PT, and platelet count on all COVID admitted patients, can use to guide care (who needs to be considered for ICU, more aggressive care, or improvement). D-dimer may predict severe disease and mortality.
- Recommend prophylactic LMWH in all patients without contraindication.
- Related Commentary to ISTH- therapeutic anticoagulation with LMWH should be considered in severe illness, and even consideration of fibrinolytics as salvage therapy. More research is needed.
Wuhan China observational data on heparin use (Tang Paper)
- 449 patients comparing heparin users versus nonusers
- No difference in 28d mortality among all patients (30% in both groups)- very high!
- Mortality difference was seen among those D-dimer 6x ULN (33% users vs 52% nonusers) and Sepsis Induced Coagulopathy score > 4 (40% vs 64%)
- Dr Ganatra pro tip: The benefit of prophylactic anticoagulation was seen in a subgroup analysis of this retrospective study – it is hard to extrapolate to therapeutic anticoagulation based on these data. Current guidelines recommend prophylactic anticoagulation in all patients with COVID19 who are critically ill.