In this post I link to Dr. Farkas’ Internet Book of Critical Care [Link is to the IBCC TOC] Toxic Shock Syndrome Chapter [Link is to the chapter] of Nov. 15, 2020.
Note to myself and my readers: I need to just review the whole chapter when I come back to this post in the future. The chapter is outstanding. I copy the notes because it helps me fix the material in my memory and it makes it easier to find the topic using my blog’s search function.
Another note to myself: In addition to reviewing the outstanding chapter show notes, I need again to listen to Dr. Farkas outstanding podcast on Toxic Shock. There are so many clinical pearls in the podcast.
And after reviewing the chapter, I need to next review the IBCC Septic Shock podcast and Septic Shock chapter, Aug. 8, 2019. The two chapters compliment each other and need to be reviewed together.
Here are the direct links Dr. Farkas has provided for the sections of the Toxic Shock Chapter:
CONTENTS
Here are excerpts:
Pitfalls
- Failure to recognize toxic shock syndrome as an entity distinct from septic shock, which requires unique management strategies.
- Failure to use IVIG in patients in patients with streptococcal toxic shock and high risk of morbidity and mortality (e.g., persistent vasopressor requirement with failure to respond to conventional therapy).
- Failure to aggressively investigate and drain foci of infection (even small abscesses or benign-appearing skin incisions – which may not seem to be clinically relevant).
pathophysiology
- Some Streptococcus and Staphylococcus species secrete superantigens that causes widespread activation of T-lymphocytes (figure above). This triggers a cascade of inflammatory cytokines (similar to septic shock), leading to multiorgan failure.
- Most people acquire antibodies that neutralize these toxins. A minority of people lack these antibodies, rendering them vulnerable to toxic shock syndrome.
staphylococcal toxic shock vs. streptococcal toxic shock
- Streptococcal toxic shock is much more common, potentially affecting patients of all ages.
- This occurs in the context of an invasive streptococcal infection. The most common source is a soft-tissue infection (e.g., cellulitis, myositis, or necrotizing fasciitis), but any invasive streptococcal infection can cause toxic shock.
- Toxic shock may be more common than generally recognized (affecting ~20% of patients with invasive group A streptococcal infection).(19393958)
- Toxic shock syndrome is most closely associated with group A Streptococcus (Streptococcus pyogenes) and Streptococcus dysgalactiae subspecies equisimilis (SDSE). SDSE may be identified in microbiology laboratories as either “Group C” or “Group G” streptococcus. Overall, SDSE is closely related to group A streptococcus and has similar clinical manifestations. Group B streptococcus (Streptococcus agalactiae) is less commonly associated with toxic shock, but several reports suggest that it might cause toxic shock syndrome.(32099700)
- Staphylococcal toxic shock is less common, affecting mostly younger patients. It often occurs due to mucosal colonization, without invasive infection. This may result from methicillin-sensitive Staphylococcus aureus (MSSA) or methicillin-resistant Staphylococcus aureus (MRSA).
Clinical Presentation
There are potentially two components of presentation: the primary site of infection and systemic effects from toxin production. In some cases, clinical manifestations are dominated by toxin secretion, whereas in other cases the primary focus of infection may be more obvious.
primary focus of infection
- In staphylococcal toxic shock, the focus of infection is often clinically silent (e.g., colonized tampon or nasal packing) or unimpressive (e.g., surgical site infection, skin abscess). Staphylococcal toxin suppresses neutrophil function, which may decrease the local signs and symptoms of infection!
- In streptococcal toxic shock, most patients will have an evident focus of infection (often cellulitis or necrotizing fasciitis).
- In some cases, the primary focus may be subtle (e.g., a small skin abscess or patch of cellulitis).
- Pain out of proportion to examination may signal necrotizing fasciitis.
- 🚩 A red flag suggesting toxic shock is a patient with an unimpressive focus of infection (e.g., a small patch of cellulitis) who is in septic shock. Most patients should not develop septic shock due to cellulitis.
systemic features from toxin production
- (1) “Flu-like syndrome” (~25% of patients)
- Fevers, chills, headache, myalgia, and/or sore throat.
- 💩 Vomiting, diarrhea, and abdominal pain are common (in the context of unexplained septic shock, diarrhea should prompt consideration of toxic shock syndrome).
- 🛑 Patients may be incorrectly diagnosed as having influenza or gastroenteritis. Patients may look OK and thereby elude initial diagnosis. Don’t expect patients to appear “toxic” initially!
- (2) Skin findings (~10% of patients)
- A diffuse erythematous rash is most characteristic. This may be subtle, resembling a sunburn.
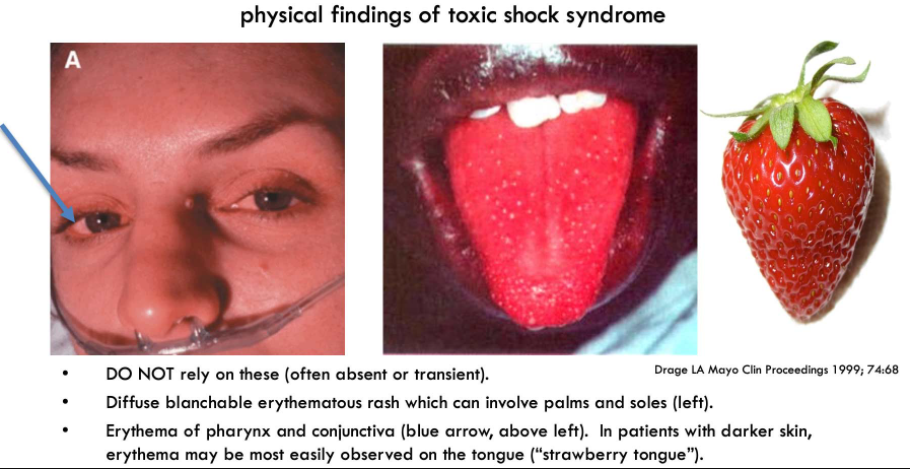
- Erythema may involve the mucous membranes (including conjunctiva and “strawberry tongue”). In patients with darker skin, mucosal changes may be the most noticeable signs (see figure below).
- A diffuse maculopapular rash may occur as well, which can be pruritic.(28299216)
- Desquamation can occur, but this is a delayed finding – it will be absent initially.
- (3) Multisystem organ failure
- Hypotension, tachycardia, and systolic heart failure (septic myocardial dysfunction) are common.
- Acute renal failure is common (often disproportionately severe compared to hemodynamic abnormalities).
- Delirium may occur.
Lab Tests
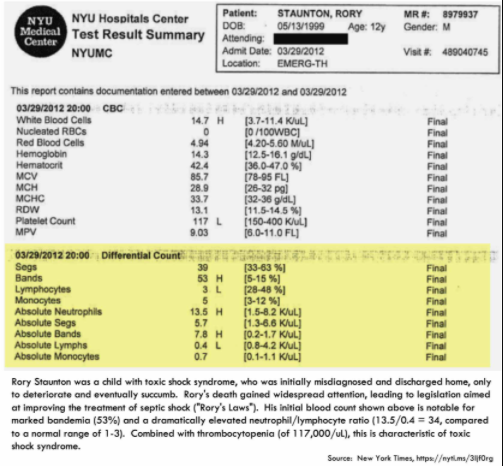
complete blood cell count and differential showing a left shift
- White blood cell count may be normal, but there is generally a left shift.
- Increased levels of immature cells (e.g., bands or immature granulocytes – depending on your assay).
- The neutrophil/lymphocyte ratio is usually quite elevated.*
* PulmCrit: Neutrophil-Lymphocyte Ratio (NLR): Free upgrade to your WBC. May 23, 2019 by Josh Farkas
“NLR increases rapidly following acute physiologic stress (<6 hours).3 This prompt response time may make NLR a better reflection of acute stress than labs which are more sluggish to respond (e.g. white blood cell count or bandemia).4 calculation & rough reference range
NLR may be calculated using either absolute cell counts or percentages, as shown here:Interpretation of NLR depends on clinical context. However, to provide some idea of how to interpret this:
A normal NLR is roughly 1-3.
An NLR of 6-9 suggests mild stress (e.g. a patient with uncomplicated appendicitis).
Critically ill patients will often have an NLR of ~9 or higher (occasionally reaching values close to 100).
acute kidney injury
- Acute kidney injury often is the first sign of organ injury. This frequently occurs at an earlier stage than in most types of septic shock, typically preceding hypotension. Consequently, patients may present with advanced acute kidney injury.(30225523)
- Urine sediment may show pyuria, in the absence of urinary tract infection.
coagulation studies
- Thrombocytopenia is characteristic. However, this is often a later and ominous sign.
- Full-blown disseminated intravascular coagulation may be seen late in the course of illness.
creatinine kinase
- Creatinine kinase more than twice normal is a component of some definitions of toxic shock syndrome. Profound cytokine elevation may cause release of creatinine kinase from muscle tissue. However, another possibility to consider is necrotizing fasciitis.
liver function tests
- Elevation of bilirubin, AST, and ALT are components of the diagnostic criteria. However, mild abnormalities in liver function tests are commonly seen in septic patients, rendering them wholly nonspecific.
cultures
- Staphylococcal toxic shock syndrome
- Culture of sterile sites are generally negative (with blood culture yield below <5%).
- Cultures are used to exclude alternative diagnoses.
- Swabs of mucosal surfaces or wounds may reveal Staphylococcus (however, this is nonspecific).
- Streptococcal toxic shock syndrome
- Blood cultures have a yield of ~60%.
- Other sterile sites may yield cultures depending on the site of infection (e.g., Group A streptococcus has a tendency to cause empyema or peritoneal infection). If necrotizing fasciitis is present with bullae, fluid from bullae may be sterilely sampled using a syringe, and sent for analysis.
Differential Diagnosis
The differential diagnosis will vary depending on specific presentations. Some closer mimics may include:
- Adrenal crisis
- Septic shock due to any other pathogen (e.g., gram-negative organisms)
- Meningococcemia and/or meningococcal meningitis
- Rocky Mountain Spotted Fever
- Leptospirosis
- Kawasaki disease
- Scarlet fever
- Streptococcal scalded skin syndrome
- Toxic shock syndrome due to Clostridium sordellii (often following obstetric procedures)
- Other forms of necrotizing soft tissue infection:
- Polymicrobial necrotizing fasciitis
- Clostridial myonecrosis and/or necrotizing fasciitis
Overall Approach To Diagnosis
- Consider the diagnosis early and often (e.g., especially in patients displaying red flags as above).
- Consider and evaluate for alternative diagnoses (e.g., meningococcal meningitis).
- When in doubt, initiate treatment for toxic shock (below).
- The treatment for toxic shock syndrome is fairly benign (and similar to the treatment for septic shock).
- If you’re wrong and the patient doesn’t have toxic shock syndrome (e.g., they simply have septic shock), you’ll still probably be doing an excellent job of caring for them.
Basic Sepsis Resuscitation
vasopressor and fluid resuscitation
- Most patients will require immediate vasopressor support, beginning with peripheral pressors and usually progressing to a central line.
- Consider a conservative fluid strategy, because patients often have leaky capillaries and won’t respond well to fluids.
stress-dose steroid
- Early use of stress-dose steroid may be a rational therapy (e.g., hydrocortisone 50 mg IV q6hr), given the tendency of these patients to develop refractory shock and their underlying immune dysregulation.
- Some patients with advanced toxic shock and disseminated intravascular coagulation may develop acute adrenal insufficiency due to adrenal gland infarction (Waterhouse-Friderichsen syndrome). Although rare, this is important to recognize, as it may lead to a chronic steroid requirement.
- Further discussion of stress-dose steroid in septic shock is found here.
septic myocardial dysfunction
- Patients with toxic shock seem to exhibit a particular tendency towards the development of myocardial dysfunction, possibly related to exuberant cytokine release and to direct effects of bacterial toxins on the myocardium.(16271055, 32099700).
- Serial echocardiography should be utilized in patients who aren’t responding well to vasopressors. Myocardial dysfunction may emerge some days after initial admission to critical care.
- Supportive care may include inotropes (e.g., epinephrine).
- With resolution of the toxic shock, myocardial recovery should occur.
More on sepsis resuscitation here.
Antibiotics
toxin-suppressive antibiotics
- Two antibiotics may be used to suppress toxin production: clindamycin 900mg IV Q8 and linezolid 600 mg IVq12.
- Clindamycin is most widely recommended.
- Clindamycin has activity against nearly all Streptococcus and most Staphylococcus (including many strains of MRSA).
- Clindamycin is supported by more robust data in the treatment of severe group A streptococcal infections.
- However, clindamycin resistance is increasing, so it cannot be relied upon to cover all strains of Streptococcus. For this reason, clindamycin should be combined with a beta-lactam.
- Linezolid has the advantages of both suppressing toxin secretion and covering all Streptococcus and Staphylococcus (including all MRSA). (16447124) Some evidence suggests that the combination of linezolid plus clindamycin is superior to either alone.(12760865, 12709354, 29366615) The combination of linezolid plus clindamycin may be reasonable initially, until it has been clarified whether the patient has staphylococcal or streptococcal infection.
beta-lactam backbone
- Beta-lactam antibiotics have the following advantages:
- (a) Uniform effectiveness against streptococcal species (with no resistance issues).
- (b) Low toxicity, which allows for prolonged antibiotic courses.
- Beta-lactams are generally combined with clindamycin for the treatment of toxigenic streptococcal infections. Clindamycin suppresses toxin synthesis, while the beta-lactam provides some coverage against clindamycin-resistant strains.
- Selection of the beta-lactam will depend on how sure you are about the focus of infection and the bacterial species. For example:
- In necrotizing fasciitis, if the bacteria involved is unclear then broad-spectrum coverage is safest (e.g., piperacillin-tazobactam or meropenem).
- If the patient is known to have a group A streptococcal infection, then penicillin G is the definitive therapy.
empiric regimen for undifferentiated toxic shock syndrome (possibly staphylococcal or streptococcal)
- For suspected toxic shock syndrome due to Staphylococcus or Streptococcus, a reasonable initial regimen is triple therapy. This will cover all potential organisms and also provide dual toxin suppression.
- (1) Linezolid 600 mg IV q6hr.
- (2) Clindamycin 900 mg IV q8hr.
- (3) A beta-lactam – selection may depend on the clinical context:
- In patients with uncomplicated cellulitis, cefazolin may be adequate.
- In patients with possible or definite necrotizing fasciitis, piperacillin-tazobactam may be a reasonable initial choice (pending culture results).
- In patients with possible meningitis, an agent with meningeal penetration may be rational (e.g., ceftriaxone 2 grams IV q12 hours or high-dose meropenem)
- Don’t be fooled by patients with “beta-lactam allergy” or “penicillin allergy” – it will always be possible to find a beta-lactam antibiotic which is safe to use. Further discussion of beta-lactam allergy here.
- Over time, these antibiotics may be de-escalated as follows:
- (1) Linezolid
- Linezolid may usually be discontinued after MRSA is excluded. Alternatively, if MRSA is the causative agent, then linezolid may be continued (with discontinuation of other agents).
- Ongoing exposure to linezolid may eventually cause thrombocytopenia (as a cumulative, dose-related adverse effect).(30567096) A few days of linezolid are probably fine, even in patients with mild thrombocytopenia (as is often the case in toxic shock). For patients with profound or worsening thrombocytopenia, exposure to more than a couple days of linezolid should probably be avoided.
- (2) Clindamycin
- Clindamycin may be continued until ~1-2 days after the resolution of sepsis syndrome.
- (3) Beta-lactam backbone
- Beta-lactam therapy is generally continued to cover the duration of therapy (which may vary depending on the site of infection).
- Following availability of official speciation and sensitivities, the beta-lactam may be narrowed appropriately.
- In group A/B/C/G streptococcal infections, the optimal beta-lactam is IV penicillin G. However, penicillin G requires frequent infusions, so for the sake of convenience it may be easier to use other agents, such as IV cefazolin or IV ceftriaxone.
DIC & purpura fulminans
purpura fulminans
- Patients with sepsis and toxic shock syndrome are at high risk for sepsis-associated DIC. Some patients will develop purpura fulminans, which is an extreme form of disseminated intravascular coagulation involving necrosis of the dermal blood vessels. In severe cases, purpura fulminans causes ischemia and autoamputation of digits or entire extremities.
- Purpura fulminans may require specific and agressive therapy to avoid limb loss – described further here.
preventing nosocomial transmission
- The Centers for Disease Control (CDC) recommends contact and droplet precautions for the first day of effective antimicrobial therapy in patients with severe group A streptococcal infection.(30225523)
- Recommendations to isolate patients are not based on high-level evidence. However, some strains of Streptococcus are definitely more toxigenic than others. Case clusters and outbreaks have been reported.
- (So yeah, we’ve used 21st century technology to verify what Semmelweis figured out in the 1800s.)