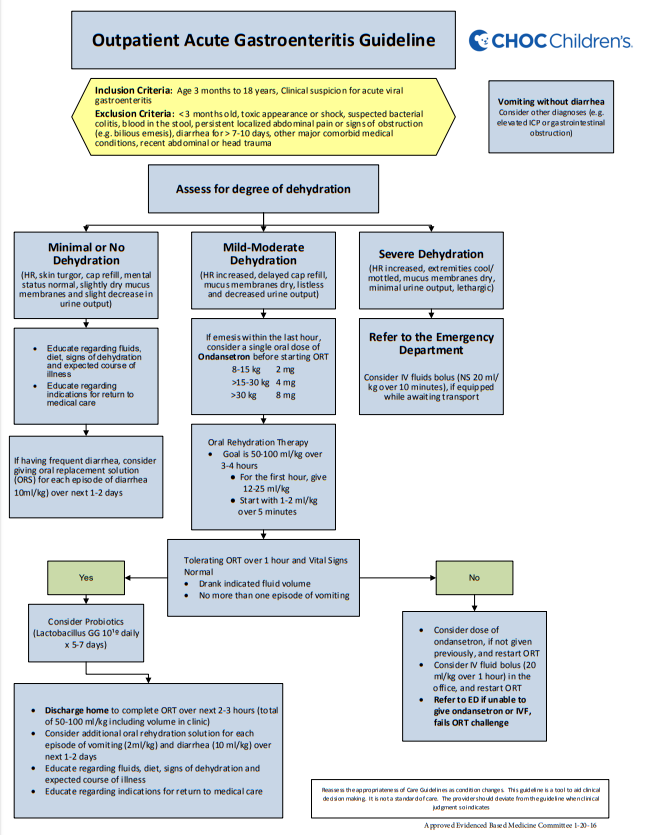
Here is a copy of the flow chart of CHOC Children’s 8 page PDF on their Outpatient Acute Gastroenteritis Guideline [Link is to flow chart and PDF]:
Here is the rest of the CHOC Children’s 8 page PDF:
Outpatient AGE Care Guideline Notes
Summary of Key Recommendations
1. The ability to discriminate degrees of dehydration clinically is limited. Categorize dehydrated
children into 3 groups:• No or minimal dehydration
• Mild-moderate dehydration
• Severe dehydration = shock or impending shock2. Laboratory studies are not needed in most patients with acute viral gastroenteritis. Consider testing if the patient has severe dehydration, will be admitted to the hospital, or testing is
needed to rule out another diagnosis.3. Oral Rehydration Therapy should be used for all patients with mild-moderate dehydration. The initial hour can be done in the office, with completion of the last 2-3 hours at home if the family
is reliable and able to return to the hospital if needed.4. As soon as the child is rehydrated, reintroduce the child’s normal diet (with the exception of no juice or soda). Avoid unnecessary dietary restrictions (e.g. giving a BRAT diet, diluting milk or formula, using lactose free formulas routinely).
5. Antibiotics, Zinc, anti-secretory medications, anti-motility agents and absorbents are not indicated in viral gastroenteritis.
6. Ondansetron should be used sparingly. It can be used in patients with mild-moderate diarrhea if there is a history of frequent vomiting. Only give a single oral dose. Do not give other antiemetics.
7. Probiotics may be helpful to decrease the duration of diarrhea for rotavirus infections, but more evidence is needed before probiotics can be routinely recommended in all cases of
gastroenteritis.Estimating the degree of dehydration
1. Classically, divided into 3 groups: mild (0-5%), moderate (5-10%), and severe (>10 %).
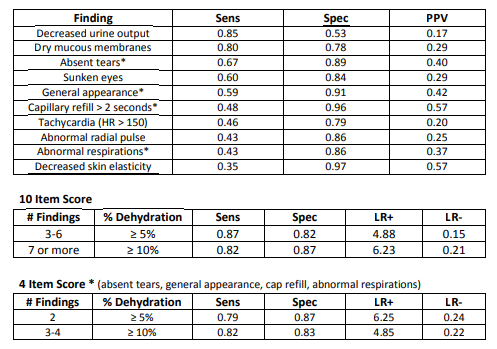
2. Performance of physical findings for ≥ 5% dehydration include:
3. Many studies assessing the degree of dehydration suffer from small numbers of patients, selecting for patients that were already thought to be dehydrated or were admitted to the
hospital, low inter-observer reliability and inappropriate reference (“gold”) standards.Because of the difficulty differentiating between mild and moderate diarrhea in clinical settings,
most experts now suggest using the following categories:• Minimal or no dehydration (0-3%)
Well appearing infants with no signs of dehydration or only 1-2 mild physical exam findings (e.g. history of decreased urine output, and slight decrease in tears).
• Mild-Moderate dehydration (3-9%)
Patient has increasing physical signs of dehydration. The severity of the findings also increases with increasing dehydration (e.g. decreased tears to absent tears).
• Severe dehydration (≥ 10%)
This should be considered shock or impending shock, and requires urgent IV fluids and transfer to an Emergency Department.
Symptoms could include altered mental status, marked tachycardia (or bradycardia), marked tachypnea, absent tears and dry mucous membranes, weak or thready pulses, cool or mottled extremities, and capillary refill > 4 seconds. Remember hypotension is a late finding in shock.
4. Lab studies (including electrolytes, BUN, creatinine, urine specific gravity and urinary ketones) do not reliably distinguish between mild and moderate diarrhea.
Laboratory Studies
1. Laboratory studies are not indicated in patients with mild-moderate dehydration who are able to be treated with ORT and discharged home.
2. Consider electrolytes, BUN and creatinine in patients with severe diarrhea, those needing IV fluids and those who will be admitted.
3. Consider CBC and blood culture when sepsis suspected.
4. Stool cultures should not routinely be obtained. They can be considered in the following patients:
• Infants < 3 months old
• Stools with gross blood or mucus
• Patients with persistent diarrhea (not improving after 7 days)
• Recent foreign travel
• Immunocompromised children
• Recent antibiotic treatment (C-difficile)
• Diarrhea after exposure to a known community bacterial outbreak
• Concern for Hemolytic Uremic Syndrome (E coli 07H157)5. Be careful when the only sign of acute “gastroenteritis” is vomiting. This is a non-specific sign and can be seen in many other diagnoses (e.g. UTI, sepsis, appendicitis, meningitis, and
intussusception). Lab studies or imaging should be done, when indicated, to rule these
diagnoses out.Oral Rehydration Therapy
1. First line therapy for mild to moderate dehydration
2. Choice of Oral Rehydration Solution (ORS):
• Solutions with equimolar concentrations of glucose and sodium maximize water reabsorption. Corresponds to solutions with glucose < 20g/dL, and sodium between
60-90 meq/L.
• Commercially available OTC solutions (e.g. Pedialyte, Enfalyte) are as effective as the revised WHO solution in developed countries.
• Both fluids with a high glucose load (sodas and fruit juices) and those with increased sodium with a low glucose amount (e.g. chicken broth) will lead to increased diarrhea
and should be avoided.3. Rehydration Phase involved giving 50-100 ml/kg (for mild-moderate diarrhea) over 3-4 hours.
• Give small volumes or ORS frequently. As an initial guide, give 1-2 ml/kg (up to 30 ml) every 5 minutes to start. Can double the volume and begin to space out frequency after 20-30 minutes if tolerated. Recommend trying to get in ¼ of the desired total volume in
the first hour (12-25 ml/kg).
• If continuing to have ongoing vomiting or diarrhea, give additional ORS at 2 ml/kg for each emesis and 10 ml/kg for each diarrheal stool.
• If vomits during this phase, can hold off on additional liquids for 20-30 minutes and restart at a lower initial volume.
• Breastfeeding can be continued during this phase.
• A child who tolerates the first hour of ORT could be sent home depending on the reliability of the parents, their ability to return if the child gets sick, and the degree of
dehydration.
• Failure of ORT would occur if the child is unable to drink the desired volume or having multiple episodes of vomiting during the rehydration phase. If having persistent vomiting, consider giving a single dose of ondansetron, if not given earlier.4. Oral Challenge in the Office
• Some patients will present with no to minimal dehydration, but have a history of frequent vomiting at home.
• In these patients, consider giving 10ml/kg of ORS over an hour as described above.5. Maintenance Phase (Recommendations for diet and fluids at home)
• If a patient continues to have frequent vomiting or diarrhea at home, they can be given
additional ORS for each episode as described above.
• Can restart formula or milk. Do not dilute formula. Do not give “clear liquids.” • No need to routinely withhold lactose containing fluids (milk, formula) in patients treated as outpatients.
• Avoid soda and juice.
• Restrictive diets (e.g. BRAT diet) are not recommended.
• Can give food as soon as the child is rehydrated. Withholding food for longer periods of time (e.g. 24 hours) is appropriate.* Fresh fruit, vegetables, complex carbohydrates (rice, wheat, bread, cereal) and lean meat are good choices.*This sentence in the guideline is an error , I believe. It should read “Withholding food for longer periods of time (e.g. 24 hours) is inappropriate.”
Medications
1. Ondansetron has shown to be effective and safe when the child is persistently vomiting.
• Decreases the risk of further vomiting (RR 0.4, 95% Cl 0.28-0.62).
• Decreases hospital admissions (RR 0.52, 95% Cl 0.28-0.62).
• A single oral dose is recommended if there is significant vomiting:8-15 kg – 2 mg
16-30 kg – 4 mg
>30 kg – 8 mg• Ondansetron has been associated with increased diarrhea in some studies but does not
seem to be clinically important.
• The FDA issued a warning that ondansetron can prolong the QTc, putting the child at
risk for fatal arrhythmiasA systematic review in the Ann Emerg Med in 2013 did not find any cases of arrhythmias related to a single oral dose of ondansetron, but did find 60 reports involving multiple
doses, IV use, pre-existing cardiac disease or concurrent use of other drugs which prolong the QTc interval.2. Because of lack of efficacy and/or the risk of significant side effects, other anti-emetics (e.g.
metoclopramide, promethazine, dimenhydrinate, grannisetron and dexamethasone) are not
recommended.3. Anti-motility agents (loperamide), absorbents (kaolin-pectin and attapulgite-activated charcoal)
and anti-secretory agents (bismuth subsalicylate) are not recommended.4. Zinc supplementation, in developing countries, leads to a decrease in the duration of diarrhea
and a decrease in persistent diarrhea. Studies in the U.S. and Europe do not show any improvement with zinc.5. Antibiotics are not indicated in acute viral gastroenteritis.
Probiotics
1. The effects seen are dependent on the strain of probiotic and dose.
2. Low quality evidence shows that Lactobacillus GG (>10 ¹⁰ CFU per day for 5-7 days) and Saccharomyces boulardii (250-750 mg/day for 5-7 days) may decrease the duration of diarrhea by 1 day and the risk for diarrhea lasting more than 3-4 days (RR 0.37-0.6).
3. The predominant cause of gastroenteritis in most of these studies was Rotavirus. In the US, where the incidence of Rotavirus has decreased significantly since vaccination was introduced, Norovirus has become the most common viral pathogen. The efficacy of probiotics against
Norovirus is not clear.4. Cost may be an issue for some families.
References
Outpatient Acute Gastroenteritis Care Guideline
1. Managing acute gastroenteritis among children: oral rehydration, maintenance, and nutritional
therapy. King CK, Glass R, Bresee JS, Duggan C; Centers for Disease Control and Prevention. MMWR Recomm Rep. 2003 Nov 21;52(RR‐16):1‐16.2. Treatment of acute gastroenteritis in children: an overview of systematic reviews of interventions
commonly used in developed countries. Freedman SB, Ali S, Oleszczuk M, Gouin S, Hartling L. Evid Based Child Health. 2013 Jul;8(4):1123‐37.3. Ondansetron and probiotics in the management of pediatric acute gastroenteritis in developed countries. Schnadower D, Finkelstein Y, Freedman SB. Curr Opin Gastroenterol. 2015 Jan;31(1):1‐6
4. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition/European Society for
Pediatric Infectious Diseases evidence‐based guidelines for the management of acute gastroenteritis in children in Europe: update 2014. Guarino A, Ashkenazi S, Gendrel D, Lo Vecchio A, Shamir R, Szajewska H. J Pediatr Gastroenterol Nutr. 2014 Jul;59(1):132‐52.5. Evidence base for probiotic products for the pediatric population. Ringel‐Kulka T. J Pediatr Gastroenterol Nutr. 2012 May;54(5):578‐9.
6. The applicability and efficacy of guidelines for the management of acute gastroenteritis in
outpatient children: a field‐randomized trial on primary care pediatricians. Albano F, Lo Vecchio A, Guarino A. J Pediatr. 2010 Feb;156(2):226‐30