In this post, I link to and excerpt from Secondary Prevention for Atherosclerotic Cardiovascular Disease: Comparing Recent US and European Guidelines on Dyslipidemia [Full-Text HTML]. Circulation Volume 141, Issue 14, 7 April 2020; Pages 1121-1123
All that follows is from the above resource.
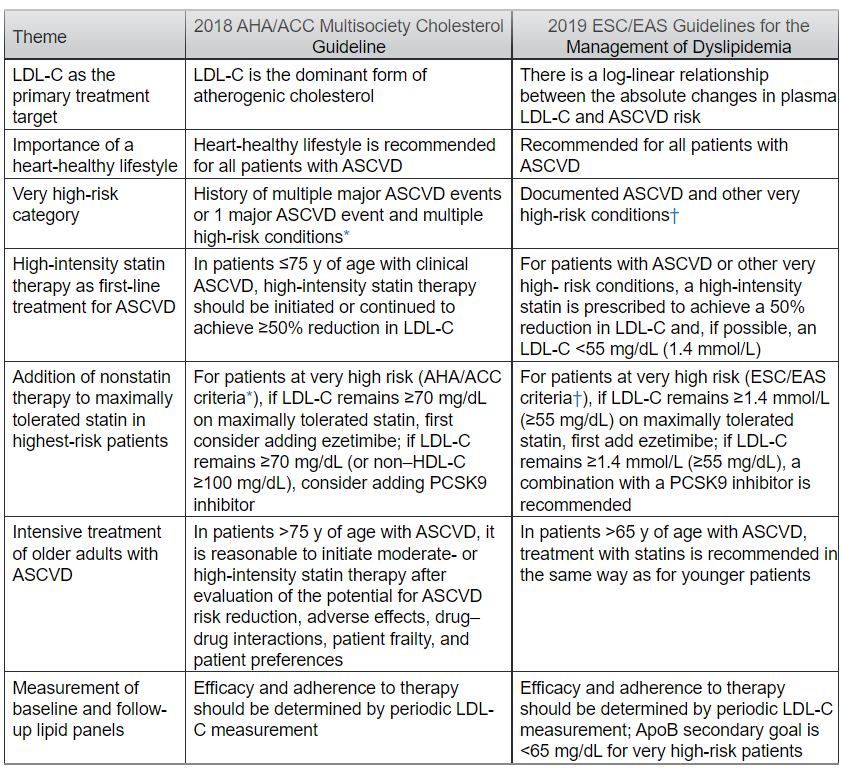
The 2018 American Heart Association (AHA)/American College of Cardiology (ACC) Multisociety Guideline on the Management of Blood Cholesterol1 divides patients with clinical atherosclerotic cardiovascular disease (ASCVD) into high-risk and very high-risk categories. The high-risk group includes those with stable ASCVD who are treated with high-intensity statins. Very high-risk patients are those with ASCVD plus other high-risk conditions; these patients are potential candidates for maximal statin therapy plus 2 nonstatins (ezetimibe and/or proprotein convertase subtilisin/kexin type 9 [PCSK9] inhibitors). This article compares and contrasts the AHA/ACC guideline with the dyslipidemia guidelines recently published by the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS)2; the latter offers a different definition of very high risk for the identification of patients deemed eligible for adding nonstatins to statin therapy (Table).
Both guidelines highlight the causal role of low-density lipoprotein (LDL) in atherogenesis, and they identify LDL cholesterol (LDL-C) as the primary target of therapy. Both guidelines recognize a heart-healthy lifestyle as the foundation for treatment. Both further recommend high-intensity statin therapy in patients with ASCVD on the basis of meta-analysis of secondary prevention randomized controlled trials of statin therapy.1,2 High-intensity statins typically reduce LDL-C by ≥50%, which supports the primary goal of therapy being a ≥50% reduction in LDL-C. Periodic monitoring of the response to therapy is generally recommended. An important addition to both guidelines is the identification of very high-risk patients who may benefit with greater absolute risk reduction from the addition of nonstatins (ezetimibe±PCSK9 inhibitor) to maximal statin therapy.
Although the 2 guidelines share many similarities, notable differences must be recognized and considered. For the definition of documented ASCVD, both guidelines include acute coronary syndrome, stable angina, coronary revascularization, stroke, transient ischemic attack, and peripheral arterial disease. The most important distinction is the definition of very high risk. The AHA/ACC guideline defines very high risk as a history of multiple major ASCVD events or 1 major ASCVD event plus multiple high-risk conditions (see footnote to the Table for details). According to the ESC/EAS, very high risk consists of documented ASCVD or various other higher-risk conditions in the absence of ASCVD (see footnote to the Table for details). These include subclinical atherosclerosis as detected by imaging and several other conditions, for example, diabetes mellitus with complications, severe chronic kidney disease, or 10-year risk of fatal CVD ≥10%. This may expand the number of patients who are candidates for nonstatin add-on therapy, especially patients without ASCVD who are classified as very high risk in the ESC/EAS guidelines. It is important to note that sufficient magnitude of benefit has not been demonstrated with randomized controlled trials, especially with nonstatin therapy in patients noted as very high risk without ASCVD.
The AHA/ACC guideline relegates subclinical atherosclerosis to primary prevention and moderate-intensity statin therapy; high-intensity statins are reserved for those also having multiple risk factors (10-year risk for ASCVD ≥20%). However, the AHA/ACC guideline does not consider subclinical atherosclerosis as an indication for nonstatin therapies. The same can be said for high-risk conditions (except familial hypercholesterolemia) without concomitant ASCVD, for example, diabetes mellitus, chronic kidney disease, or multiple risk factors. As per the AHA/ACC guideline, these conditions alone usually trigger statin therapy but do not warrant the addition of PCSK9 inhibitors.
Besides the LDL-C goal of 50% reduction in patients with ASCVD, both guidelines use numeric thresholds for enhancement of LDL-lowering therapy with nonstatins. In the AHA/ACC guideline, for very high-risk patients, when LDL-C exceeds 70 mg/dL (1.8 mmol/L) on maximal statin therapy, ezetimibe is recommended. If LDL-C remains ≥70 mg/dL or non–high-density lipoprotein cholesterol remains ≥100 mg/dL, consideration can be given to adding a PCSK9 inhibitor. ESC/EAS guidelines offer a similar recommendation for very high-risk patients except that the threshold for adding nonstatins is an LDL-C level ≥55 mg/dL (1.4 mmol/L).
Marginal gains in ASCVD risk reduction diminish as LDL-C levels fall to very low levels. The reason is the curvilinear (log-linear) relation between LDL-C and ASCVD risk. Moreover, a high percentage of patients with ASCVD will have LDL-C ≥55 mg/dL even on maximal statin plus ezetimibe; thus, efficacy and cost considerations may make greatly expanded use of PCSK9 inhibitors problematic. Even in clinical trials of high-intensity statin therapy, ≈75% of patients with acute coronary syndrome continue to have LDL-C levels ≥50 mg/dL, with ≈55% of the patients with LDL-C ≥60 mg/dL.3 Whether such a large number of patients should be prescribed ezetimibe or PCSK9 inhibitors to lower LDL-C levels to <55 mg/dL, at which point gains exist but in terms of net benefit are marginal, remains worthy of further study.
In summary, the AHA/ACC guideline focuses on strictly defined criteria in the clinical trials of statin and nonstatin therapy. That guideline uses the concept of LDL-C and non–high-density lipoprotein cholesterol thresholds. By defining a very high-risk patient with ASCVD subset using clinical criteria, this guideline attempts to identify patients with ASCVD who will derive a much larger absolute reduction in recurrent ASCVD risk compared with an average patient with ASCVD.4 The ESC/EAS guidelines recommend an alternative goal-based approach in all patients with either clinical or imaging-based ASCVD (moderate subclinical atherosclerosis). This approach can probably be justified on theoretical grounds and the general concept that lower is better for LDL-C. However, it lacks rigorous testing of efficacy by randomized controlled trials and cost-effectiveness analysis. We hope that further dialog and evidence will guide future iterations of both guidelines in this ever-growing field. Meanwhile, it is crucial to improve implementation of both guidelines to ensure that the maximum ASCVD events are prevented among patients with clinical ASCVD.
Published online April 6, 2020.The opinions expressed in this article are not necessarily those of the editors or of the American Heart Association.