Note to myself: I originally posted this on December 20, 2019. I’ve recently reviewed this excellent podcast and chapter and so I moved it up to today. I’ve also made a few changes. And be sure and review all of the Dr. Farkas outstanding related posts which he links to in his Community-Onset Pneumonia chapter of December 9, 2016. These are:
- Using the Procalcitonin strategy
- Myth-busting: Azithromycin does not cause torsade de pointes or increase mortality
July 27, 2015 by Josh Farkas 9 Comments - Episode 216: Should the dose of ceftriaxone in critically ill patients be 1 gram or 2 grams?
AUGUST 14, 2017 BY PHARMACY JOE - PulmCrit- The siren’s call: Double-coverage for ventilator associated PNA August 29, 2016 by Josh Farkas 6 Comments
- Mythbusting: (Empty IVC + hyperkinetic heart) does not equal volume depletion
December 14, 2014 by Josh Farkas 4 Comments - Understanding lactate in sepsis & Using it to our advantage
July 5, 2015 by Josh Farkas 30 Comments - Evidence-based treatment for severe community-acquired pneumonia
August 16, 2015 by Josh Farkas 2 Comments - Pneumonia, BiPAP, secretions, and HFNC: New lessons from FLORALI
May 25, 2015 by Josh Farkas - Large volume thoracentesis: How much can safely be removed?
September 24, 2014 by Josh Farkas 12 Comments
In this post I link to and excerpt from the chapter, Pneumonia, September 17, 2019, of Dr. Farkas’ incredible Internet Book Of Critical Care [Link is to TOC]. And after reviewing the chapter, listen to the summary podcast of the chapter.
Note to myself and to my readers: I recommend reviewing the awesome Pneumonia Chapter on the IBBC. I only make the excerpts on many posts because the exercise of excerpting helps me fix the subject in my mind
All that follows are excerpts from the Pneumonia Chapter of The Internet Book Of Critical Care [except where noted – material in brackets]:
CONTENTS
- definitions
- diagnosis
- triage: who needs ICU?
- treatment
- treatment failure
- duration of treatment
- checklists & algorithms
- podcast
- questions & discussion
- pitfalls
- PDF of this chapter (or create customized PDF)
checklists & algorithms
main checklist:
Pitfalls
- Failure to cover for atypical (e.g. treating with piperacillin-tazobactam monotherapy).
- Unnecessary MRSA coverage in patients at low risk for MRSA. In particular, after 2-3 days if there is no evidence that the patient has MRSA (e.g. negative nares PCR & negative sputum), then MRSA coverage should be stopped.
- Triaging patients based on their oxygen requirement, while ignoring tachypnea and work of breathing.
- Under-utilization of high-flow nasal cannula, over-utilization of BiPAP.
- Under-utilization of steroid (especially in patients who may benefit substantially, e.g. underlying asthma/COPD).
- Missing a pleural effusion which develops insidiously after admission.
- Egregiously weird antibiotic regimens for patients with dubious penicillin allergy (ceftriaxone is fine here; more on this to come).
- Using fluoroquinolones (it’s a trap).
- Giving clindamycin for anaerobic coverage.
- Double-coverage of pseudomonas.
- Dumping 30 cc/kg fluid into a sick pneumonia patient on the verge of intubation because the lactate is elevated. Please, please, please, stop this madness, I beg of you.
definitions
Until we have better definitions, this chapter is entitled “community-onset pneumonia” which is quite simply defined as anyone presenting to the hospital with pneumonia. This is a combination of the CAP plus HCAP. As explored below, we will use a personalized strategy to determine which patients need broad-spectrum antibiotic coverage (rather than the CAP-vs-HCAP algorithm).
[See definitions for details on the above terms]
does this patient have pneumonia?
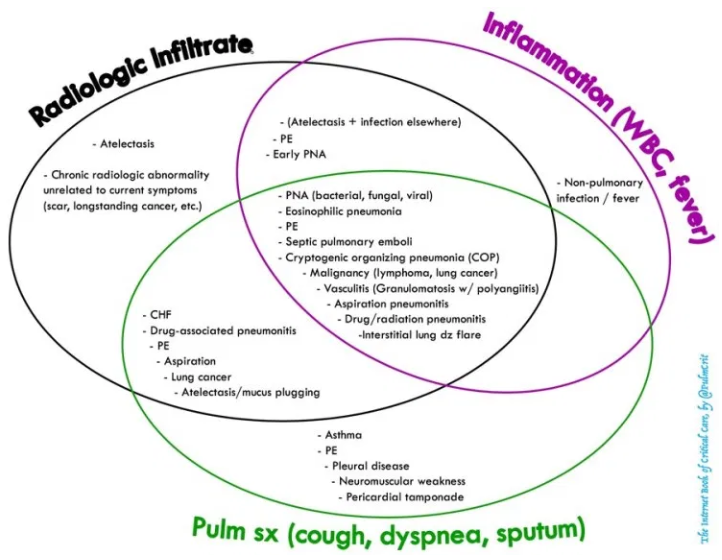
diagnosing pneumonia can be tricky!
- Diagnosis is generally based on three lines of evidence:
- Imaging evidence of a chest infiltrate (e.g., CXR, CT, ultrasound)
- Inflammation (e.g., fever/hypothermia, rigors, night sweats, leukocytosis, left-shift, procalcitonin)
- Pulmonary symptoms (e.g., dyspnea, cough, sputum production, pleuritic chest pain) and signs (tachypnea, hypoxemia).
- Elderly patients may present with non-pulmonary complaints (e.g. falling, delirium, sepsis).
- When in doubt, it is reasonable to get cultures and start antibiotics for pneumonia. Within the next 24-48 hours, the diagnosis may be re-considered and antibiotics discontinued as appropriate.
- More common pneumonia mimics are listed below. These can be devilishly hard to find, because you’re searching for a needle in a haystack.
avoid pneumonia over-diagnosis
- Patients often present to the hospital with septic shock plus pulmonary infiltrates on chest X-ray. Two possibilities are:
- 1) Pneumonia
- 2) Chest infiltrates due to atelectasis/aspiration plus occult focus of sepsis elsewhere (e.g. abdominal sepsis)
- A common error is to assume that the septic shock must be due to pneumonia, when in fact the chest infiltrates are a red herring. When in doubt, err on the side of investigating further to exclude an alternative source of sepsis.
CT scan to assist the diagnosis of pneumonia
- Some patients with pneumonia will have a negative chest X-ray with a positive CT scan (due to subtle infiltrates). However, among patients who are critically ill due to pneumonia, there really ought to be some abnormality seen on chest X-ray.
- The main use for CT scan is differentiation from pneumonia mimics. As shown in the table above, a CT scan is probably the single most versatile test to differentiate pneumonia from a mimic.
- CT scan can be helpful to detect pneumonia in patients with chronic lung disease and chronically abnormal chest X-ray.
post-diagnosis testing
tests to obtain after diagnosing pneumonia
- Blood cultures
- Recommended for severe pneumonia, although yield is low (~10%).3
- If patient already had blood cultures at another hospital, don’t repeat them (follow up on results from the outside hospital lab).
- Sputum for gram stain & culture
- Intubated patient: tracheal aspirate is very useful.
- Non-intubated patient: expectorated sputum (low yield, but very helpful when high-quality sputum reveals single type of organism).3
- Urine legionella antigen
- Sensitivity 80% and specificity of ~95%.3
- Negative result doesn’t exclude legionella, but positive result may allow focusing antibiotic therapy on legionella.
- Urine pneumococcal antigen
- Sensitivity 70% and specificity 95% (may have false-positive due to pneumonia within past several weeks).3 4
- Nares PCR for MRSA
- Winter: PCR for influenza & respiratory viruses
- If nasopharyngeal influenza PCR is negative and high suspicion remains, a lower respiratory tract PCR may be positive.5
- Be careful: patients may be co-infected with viral and bacterial pathogens. Just because the viral PCR is positive doesn’t mean that you should stop antibacterial therapy.3
- Procalcitonin
- Procalcitonin <0.5 ng/mL argues against typical bacterial pneumonia (but this may still be seen with atypical infections).
- Procalcitonin is unreliable in immunocompromised patients (e.g. neutropenia).
- Daily procalcitonin may help determine timing of antibiotic discontinuation (more on this below).
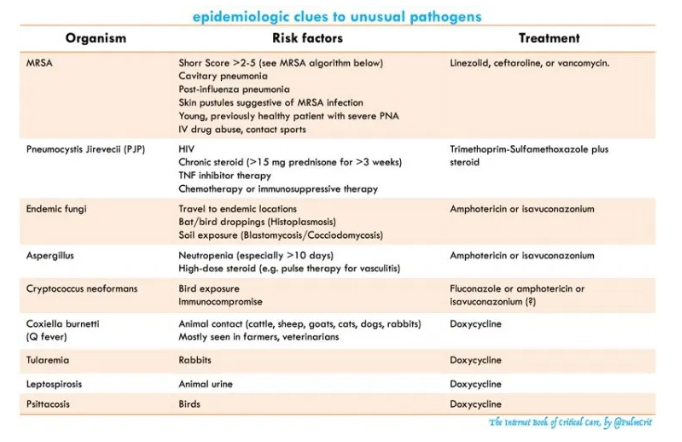
- Epidemiological history (table below).
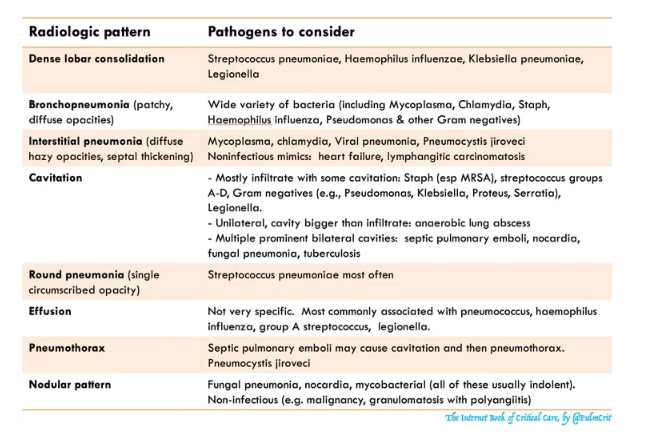
- Review of radiograph for diagnostic clues (table below)
epidemiological history
radiographic patterns
- Cannot be entirely relied upon. However, they can provide useful clues, so they shouldn’t be ignored either.
- In general, radiographic patterns should be used primarily to broaden the differential diagnosis (not to narrow it).
ultrasonography for effusion
- If there is any doubt regarding possible effusion (e.g. basilar opacities), bedside ultrasonography should be performed to clarify this.
- Ultrasonography should be repeated daily to watch for the development of an effusion or empyema over time.
CT scan & bronchoscopy
- Main indications for more advanced evaluation:
- Immunocompromise
- Unusual chest imaging (e.g. chest X-ray suggestive of nodular/cavitating pneumonia).
- CT scan may increase the index of suspicion for unusual pathogens, for example:
- Diffuse infiltrates with a ground-glass pattern may suggest pneumocystis jiroveci pneumonia.
- Multi-focal dense nodular infiltrates may suggest a fungal pneumonia
- Bronchoscopy may be needed to exclude unusual organisms.
Additional Links
The American College of Radiology [Link is to ACR Home Page] Appropriateness Criteria [Link is to the ACR Appropriateness Criteria Home Page] have excellent imaging guidelines on this topic:
- Acute Respiratory Illness in Immunocompetent Patients Revised 2018
- Acute Respiratory Illness in Immunocompromised Patients Revised 2019
triage: who needs ICU?
better approaches to triage
- Patients with significant work of breathing or tachypnea (e.g. respiratory rate >30) should be considered for ICU admission and high-flow nasal cannula.4 6
- The IDSA/ATS criteria have been validated for use in ICU triage.7 8 Patients with three or more criteria may benefit from ICU admission:9
- Respiratory rate >29 breaths/min10
- Hypotension requiring volume resuscitation
- PaO2/FiO2 < 250 (patients requiring >3 liters oxygen)11 12
- Temperature < 36C
- Confusion
- Multilobar infiltrates
- BUN >20 mg/dL
- WBC <4,000/mm3
- Platelets <100,000/mm3
- These criteria are guidelines which won’t apply perfectly to every patient. When in doubt, observe the patient carefully over a few hours and use your judgement.
antibiotic selection
don’t forget atypical coverage!
- Should always be included in the empiric antibiotic regimen for severe pneumonia.
- Remember: Legionella causes ~10-15% of severe pneumonia. This won’t be covered by the broadest beta-lactams in the world (e.g. cefepime, piperacillin-tazobactam, meropenem)
- Azithromycin is an excellent choice here:
- Solid track record in pneumonia.
- Retrospective studies suggest mortality benefit, even in pneumococcal pneumonia sensitive to beta-lactams (possibly due to anti-inflammatory activity, or coinfection with atypical pathogens).
- If the patient is diagnosed with pneumococcus, azithromycin should still be continued for 3-5 days.13 14
- Well-tolerated, very safe. Don’t worry about the QT interval, the concept that azithromycin causes torsade de pointes is mythological.15
- Doxycycline is also an excellent choice for atypical coverage, with the following advantages:
- Covers weird organisms acquired from animal contact (coxiella, tularemia, psittacosis, leptospirosis).
- Usually active against MRSA in vitro, but it’s unclear whether this is effective for clinical MRSA pneumonia.16
- Fluoroquinolones are a poor choice for atypical coverage in the ICU for several reasons.
beta-lactam backbone
- The beta-lactam backbone will cover gram-positives (especially pneumococcus) and gram negatives.
- Ceftriaxone is an excellent choice for most patients.
- It’s controversial whether to use 1 or 2 grams IV daily. Increasing drug resistance over time may be an argument to use 2 grams. This should also be considered in obese patients.
- Pseudomonal beta-lactam (piperacillin-tazobactam or cefepime) may be used in patients with risk factors for pseudomonas, for example:
- Septic shock due to pneumonia.
- Structural lung disease (e.g. bronchiectasis or advanced COPD with frequent exacerbations).
- Broad-spectrum antibiotics for >7 days within past month.
- Hospitalization for >1 day within past three months.
- Immunocompromise (e.g. chemotherapy, chronic use of >10 mg prednisone daily).
- Nursing home resident with poor functional status.
- Patients with penicillin allergy:
- Non-anaphylactic reaction to penicillin: may use ceftriaxone or cefepime.
- Anaphylaxis or angioedema from penicillin: may use meropenem.
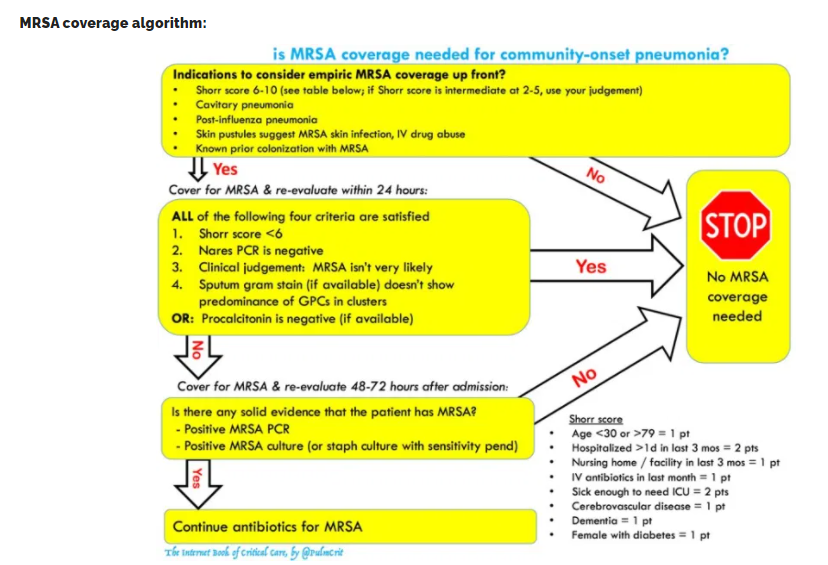
MRSA coverage is occasionally needed as 3rd drug
- An evidence-based algorithm to guide the use of MRSA coverage is shown above (more detail here).
- The key is ongoing, thoughtful evaluation of data.
- Staph generates lots of purulence and is generally not difficult to isolate.
- MRSA coverage should be stopped within 48-72 hours unless there is some objective data that the patient has MRSA.
- Choice of agent
- Linezolid is arguably first-line therapy for MRSA pneumonia (compared to vancomycin, linezolid has superior lung penetration, causes no nephrotoxicity, and suppresses bacterial toxin synthesis).17 18
- Vancomycin is the traditional option if linezolid is contraindicated. Unfortunately, resistance to vancomycin is increasing over time. If susceptibility testing shows borderline sensitivity to vancomycin (MIC 1.5-2 mcg/mL) this may increase the risk of treatment failure and an alternative agent might be better. If the MIC is >2 mcg/mL then a different antibiotic should definitely be used.
- Ceftaroline is a fifth-generation cephalosporin active against MRSA. It might be superior to vancomycin (particularly for strains with MIC>1 mcg/mL), but there is no high-quality evidence available.19 20
- Daptomycin isn’t an option here because it is degraded by surfactant and thus cannot treat pneumonia.
double-coverage for pseudomonas is not needed
- Unless you’re living in a post-apocalyptic hellscape where pseudomonas are insanely resistant to beta-lactams, this shouldn’t be necessary. Double-coverage doesn’t even appear to benefit patients with ventilator-associated pneumonia (which involves a much greater risk of resistant pseudomonas). More on this here.
anaerobic coverage is not needed for pneumonia
- Sometimes there is concern that the patient may have aspirated, so they should be covered for anaerobes.
- The lung is the best oxygenated organ in the body, so it is not very susceptible to anaerobic infection. The only way anaerobic infection can occur is if there is an anatomic disruption that creates a poorly oxygenated compartment (abscess or fluid collection).
- Anaerobic coverage is indicated only for empyema or lung abscess.
resuscitation
avoid large-volume fluid resuscitation
- Large volume fluid resuscitation may worsen hypoxemic respiratory failure and thereby precipitate the need for intubation.
- Most patients with PNA can be stabilized adequately with small-moderate volumes of fluid combined with vasopressors if needed.
- Consider early institution of vasopressors. In many cases, a low-dose vasopressor (e.g. norepinephrine 5-10 mcg/min) may substantially reduce the amount of fluid which is needed to stabilize the patient.
- Fluid should be used only if the following conditions are met:
- Organ hypoperfusion (e.g. poor urine output) or refractory hypotension PLUS
- History and evaluation indicates true volume depletion (as opposed to hypotension which is merely due to vasodilation). Please note that a reduced central venous pressure or collapsed inferior vena cava doesn’t necessarily indicate volume depletion, these findings can also be caused by systemic vasodilation.
- Lactate elevation is not a sign of organ malperfusion, nor is it an indication for fluid.
respiratory support
high-flow nasal cannula (HFNC)
- The FLORALI trial suggested improved mortality among patients with severe hypoxemia treated with HFNC.
- HFNC should be considered in patients with significant work of breathing and/or tachypnea. The goal of HFNC is to reduce the work of breathing, and thereby prevent patients from tiring out. In order for this to work, HFNC must be started before the patient is exhausted and in extremis.
- Advantages of HFNC:
- Oxygenation support
- Ventilation support due to dead-space washout
- Humidification may promote secretion clearance
- Doesn’t interfere with sputum clearance, coughing, or eating
- Patients may remain on HFNC for several days if needed (often the case for severe lobar pneumonia).
generally avoid BiPAP
- BiPAP doesn’t allow patients to clear their secretions. Patients treated on BiPAP often do well initially, but eventually may fail due to retained secretions and mucus plugging.
- BiPAP may be used for limited periods of time to stabilize patients (e.g. for transportation).
- Occasional patients with COPD plus pneumonia may benefit from a rotating schedule of BiPAP and HFNC. Pulmonary toilet and secretion clearance may be performed while the patient is on HFNC.
endotracheal intubation
- Generally used as a second-line therapy after trying HFNC.
- Indications for intubation in pneumonia are usually:
- Refractory hypoxemia
- Progressively worsening work of breathing, respiratory exhaustion
adjuvant therapies
steroid
- Several RCTs show that steroid may reduce the length of stay and risk of intubation among pneumonia patients. The SCCM/ESICM guidelines currently recommend steroid for patients with severe community-acquired pneumonia.21
- Steroid should be given to patients with severe PNA in the absence of contraindications.
- Patients in whom steroid may be contraindicated:
- Paralytic infusion (risk of myopathy)
- Suspicion of pneumonia due to fungus, tuberculosis, or possibly influenza.
- Immunocompromise (HIV, chemotherapy, neutropenia)
- There is no specific regimen of steroid. The following are all reasonable options:
- Prednisone burst (e.g. 50 mg PO daily for 5 days) or equivalent dose of methylprednisolone (e.g. 40 mg IV daily for five days).
- Traditional stress dose steroid (50 mg hydrocortisone IV q6hr) – this may be preferred for patients in shock.
ascorbic acid
- One before-after single-center trial suggested a mortality benefit from metabolic resuscitation.22 The regimen used was hydrocortisone 50 mg IV q6hr, thiamine 200 mg IV q12hours, and ascorbic acid 1.5 grams IV q6h5.
- High-dose IV vitamin C is currently under investigation within a multicenter RCT for treatment of acute lung injury (CITRIS-ALI trial).
- At this point in time, metabolic resuscitation for severe PNA is reasonable but not proven. Watch this space.
effusion management
[See link above for details on effusion management. POCUS will be an important aspect in decision making.]
treatment failure
defining treatment failure
- No clear definition, but clinical improvement should generally be seen within ~3 days.
- Persistent or rising procalcitonin may be an early sign of treatment failure.
- Ongoing deterioration in oxygenation and infiltrates >24 hours after antibiotics is the most concerning feature.
- Radiographic improvement takes weeks, so failure for chest x-ray to improve over a few days means nothing.
- Indeed, if the chest x-ray clears up within 24-48 hours that might be suggestive of aspiration pneumonitis, rather than true bacterial pneumonia.
differential diagnosis
- Wrong initial diagnosis (e.g. heart failure, pulmonary embolism, alveolar hemorrhage, cryptogenic organizing pneumonia, eosinophilic pneumonia, – see differential diagnosis figure above).
- Noninfectious complication of hospitalization (iatrogenic volume overload, pulmonary embolism, drug fever, aspiration).
- Wrong antibiotic (e.g. multi-drug resistant organism, fungal pneumonia, Q-fever, psittacosis).
- Inadequate antibiotic dose or penetration into lung tissue.
- Intra-thoracic complication of infection (abscess, empyema, pleural effusion, ARDS).
- Metastatic infection (endocarditis, meningitis, arthritis).
- Weak host.
evaluation
- Review all data carefully (especially microbiology).
- CT chest is generally performed to secure the diagnosis of pneumonia and exclude anatomic complication (e.g. abscess or empyema) or pulmonary embolism.
- Repeat cultures (blood and sputum).
- Bronchoscopy may be considered.
- If a significant pleural effusion is present, it may be drained and sampled.
- Procalcitonin is helpful occasionally to sort out infectious vs. non-infectious illness.
- Negative procalcitonin (<0.25 ng/ml) after three days suggests the presence of a non-infectious complication, whereas persistently elevated procalcitonin suggests active infection.
- Among patients with renal insufficiency, C-reactive protein might be used in an analogous fashion (with CRP levels <30 mg/L roughly analogous to a negative procalcitonin).23
duration of treatment
Either time or procalcitonin may be used to guide the length of treatment. When in doubt, both factors may be considered [See this section of the post [Link above].
checklists & algorithms