In this post, I link to and excerpt from An EAPCI Expert Consensus Document on Ischaemia with Non-Obstructive Coronary Arteries in Collaboration with European Society of Cardiology Working Group on Coronary Pathophysiology & Microcirculation Endorsed by Coronary Vasomotor Disorders International Study Group [PubMed Abstract] [Full-Text HTML] [Full-Text PDF]. EuroIntervention. 2021 Jan 20;16(13):1049-1069. doi: 10.4244/EIJY20M07_01.
The above article has been cited by 47 articles in PubMed.
All that follows is from the above resource.
Abbreviations
ACEis Angiotensin-converting enzyme inhibitors
ACH Acetylcholine
ARB Angiotensin receptor blockade
ATP Adenosine-50-triphosphate
BP blood pressure
CABG Coronary artery bypass surgery
CAD Coronary artery disease
CCS Chronic coronary syndrome
CCTA Coronary computed tomographic angiography
CFR Coronary flow reserve
CFVR Coronary flow velocity reserve
CMD Coronary microvascular dysfunction
CVD cardiovascular disease
COVADIS Coronary Vasomotor Disorders International Study
EAPCI European Association of Percutaneous Cardiovascular
Interventions
EECP Enhanced external counterpulsation
ESC European Society of Cardiology
FCA Invasive functional coronary angiography
FFR Fractional flow reserve
GTN Glyceryl trinitrate
HMR Hyperaemic myocardial velocity resistance
hsCRP high-sensitive C reactive protein
IHD Ischaemic heart disease
IMR Index of microcirculatory resistance
INOCA Ischaemia with non-obstructive coronary arteries
MI Myocardial infarction
MVA Microvascular angina
PET Positron emission tomography
SDAIC Scientific Documents and Initiatives Committee
WISE Women’s Ischaemia Syndrome EvaluationAbstract
This consensus document, a summary of the views of an expert panel organized by the European Association of Percutaneous Cardiovascular Interventions (EAPCI), appraises the importance of ischaemia with non-obstructive coronary arteries (INOCA). Angina pectoris affects approximately 112 million people globally. Up to 70% of patients undergoing invasive angiography do not have obstructive coronary artery disease, more common in women than in men, and a large proportion have INOCA as a cause of their symptoms. INOCA patients present with a wide spectrum of symptoms and signs that are often misdiagnosed as non-cardiac leading to under-diagnosis/investigation and under-treatment. INOCA can result from heterogeneous mechanism including coronary vasospasm and microvascular dysfunction and is not a benign condition. Compared to asymptomatic individuals, INOCA is associated with increased incidence of cardiovascular events, repeated hospital admissions, as well as impaired quality of life and associated increased health care costs. This consensus document provides a definition of INOCA and guidance to the community on the diagnostic approach and management of INOCA based on existing evidence from research and best available clinical practice; noting gaps in knowledge and potential areas for further investigation.
Introduction
Angina pectoris, the most common symptom of ischaemic heart disease (IHD), affects approximately 112 million people globally.1 The 2019 ESC guidelines provides guidance on the diagnosis and management of patients with chronic coronary syndromes (CCS).2 A large proportion of patients (up to 70%) undergoing coronary angiography because of angina and evidence of myocardial ischaemia do not have obstructive coronary arteries but have demonstrable ischaemia.2,3 Studies carried out in the past two decades have highlighted that coronary microvascular dysfunction (CMD) and epicardial vascular dysfunction are additional pathophysiologic mechanisms of IHD.4 Coronary microvascular dysfunction and epicardial vasospasm, alone or in combination with coronary artery disease (CAD), are adjunctive mechanisms of myocardial ischaemia. However, these conditions are rarely correctly diagnosed and, therefore, no tailored therapy is prescribed for these patients. As a consequence, these patients continue to experience recurrent angina with impaired quality of life, leading to repeated hospitalizations, unnecessary coronary angiography and adverse cardiovascular outcomes in the short-and long term.5,6
This consensus document provides a definition of ischaemia with non-obstructive coronary arteries (INOCA) and guidance to the clinical community on the diagnostic approach and management of INOCA based on existing evidence and best current practices.
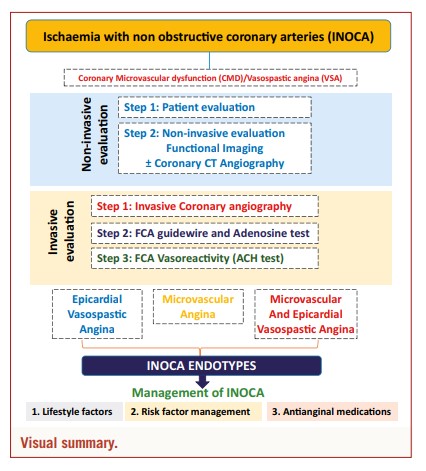
A failure to diagnose epicardial CAD in a patient with documented angina/ischaemia should promote a subsequent search pathway to elucidate INOCA endotypes before a search for non-cardiac causes of chest discomfort is explored.
INOCA endotypes
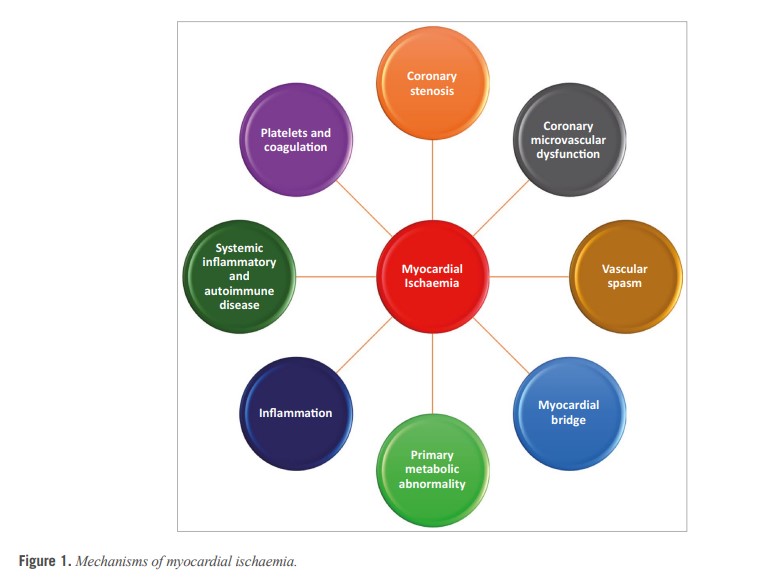
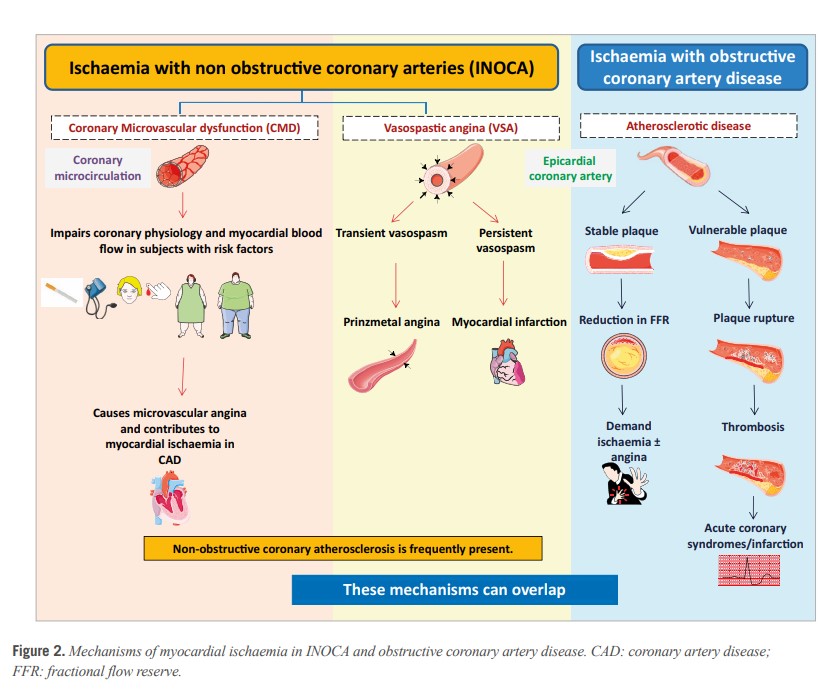
In the setting of CCS, a mismatch of demand-supply of coronary artery blood flow may lead to transient or recurrent cardiac chest pain related to myocardial ischaemia due to inadequate cellular availability of adenosine-50-triphosphate.8 Although obstructive CAD is a frequent and well-acknowledged cause of myocardial ischaemia, many stenoses judged as severe on visual assessment, are not flow-limiting. Functional misclassification of obstructive lesions frequently occurs in the range of 40-80% stenosis severity, being particularly high in case of patients with multiple coronary lesions.9,10,11 The most recent ESC guidelines recommend the use of myocardial fractional flow reserve (FFR) or instantaneous wave-free ratio to identify patients at high event risk who will benefit from revascularisation.2 Cardiac ischaemia may also be caused by vascular dysfunction without obstructive CAD, a condition recently termed INOCA. In INOCA, the mismatch between blood supply and myocardial oxygen demands may be caused by CMD and/or epicardial coronary artery spasm, typically in the setting of non-obstructive coronary atherosclerosis.12 Figure 213,14 shows the mechanisms of INOCA. Of note, these mechanisms may also cause ischaemia in patients with concomitant obstructive CAD and atherosclerosis with outward remodelling but these cases are not included in INOCA by definition.
MICROVASCULAR ANGINA
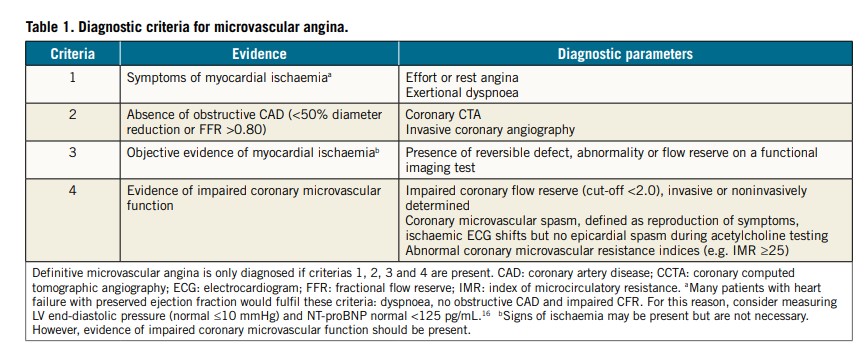
Microvascular angina (MVA) is the clinical manifestation of myocardial ischaemia caused by CMD. In this clinical entity, myocardial ischaemia may result from structural remodelling of the microvasculature (leading to fixed reduced microcirculatory conductance) or vasomotor disorders affecting the coronary arterioles (causing dynamic arteriolar obstruction).15,16 Both vascular dysfunction mechanisms may co-exist and contribute to MVA. An updated standardization of criteria for MVA in patients presenting with angina pectoris or ischaemia-like symptoms in the absence of flow-limiting CAD has been proposed by the COVADIS group15 (Table 1).
EPICARDIAL VASOSPASTIC ANGINA
Vasospastic angina (VSA) is the clinical manifestation of myocardial ischaemia caused by dynamic epicardial coronary obstruction caused by a vasomotor disorder. In 1959, Prinzmetal described the clinical and electrocardiographic manifestations (transient ST-segment elevation) of a disorder thought to be due to epicardial coronary artery spasm.17 Subsequently, other forms of vasomotor disorders causing chest pain with transient ST-segment depression or T-wave inversion were described. Overall, these clinical entities caused by epicardial vessel spasm were grouped under the term VSA. A standardization of diagnostic criteria for VSA has been previously described by the COVADIS group (Supplementary material online, Table S1).18 Microvascular angina and epicardial VSA can co-exist which is associated with worse prognosis.19
Epidemiology
PREVALENCE IN THE GENERAL POPULATION AND ACCORDING TO SEX AND AGE
The majority of patients referred for assessment for angina do not have obstructive coronary arteries. In unselected populations referred for assessment less than 10% have obstructive CAD.3,20 In all studies, there is a strong female preponderance for the condition. A large US multicentre study showed that nearly 39% of the patients selected for coronary angiography because of suspected angina and/or positive stress test have non-obstructive CAD.21 This frequency is higher among women (approximately 50-70%), compared to men (30-50%).
PREVALENCE OF CORONARY MICROVASCULAR DYSFUNCTION
The prevalence of CMD in patients with angina and no obstructive CAD undergoing invasive angiography depends on the methods and cut-off applied. In the iPower study, 26% of 963 symptomatic women with no obstructive CAD had coronary flow velocity reserve (CFVR) below two when assessed by transthoracic Doppler echo.23 However, these studies should be interpreted in the context that non-invasive estimation of CFVR has several limitations.24,25
Other studies assessing CMD invasively or by positron emission tomography with different cut-offs have found 39-54% have CMD.21,26 In a large study with invasive assessment of CMD in 1439 men and women with chest pain and no obstructive CAD included over a period of 19 years, 30% had abnormal CFVR in response to adenosine.27
The association between traditional cardiovascular risk factors and INOCA is not well established. Smoking has been associated with CMD.28 Age, diabetes, hypertension, and dyslipidaemia were associated with impaired CMD both in the iPower study and WISE study.21,23 Other studies have shown that diabetes was uncommon among patients presenting with angina and non-obstructive CAD, while hypertension and dyslipidaemia were relatively more prevalent.27,29
Coronary microvascular dysfunction is associated with proinflammatory markers in women with INOCA.30,31 In the WISE cohort, novel risk variables like those associated with inflammation seemed to play a role in CMD.32 For instance, systematic lupus erythematosus and rheumatoid arthritis are associated with CMD and are frequently encountered in patients with angina and CMD.33,34
PREVALENCE OF CORONARY ARTERY SPASM
The Japanese population has a higher prevalence of angina related to coronary vasomotor disorders38 compared with western populations. In addition, the frequencies of multiple coronary spasm (≥2 spastic arteries) by provocative testing in Japanese (24.3%)39 and Taiwanese populations (19.3%)40 are markedly higher than those in Caucasians (7.5%).41 Interestingly, VSA is more prevalent among men than women.40 Most patients with VSA are between 40 and 70 years of age, and the prevalence tends to decrease after the age of 70 years.40 Previous Asian studies of patients with non-obstructive CAD have shown that the prevalence of coronary vasomotor disorders is around 50% in patients with angina.42,43 European studies have also shown a high prevalence of epicardial vasospasm when systematically tested.44,45 However, due to differences in stress protocols and definitions applied, the studies are not directly comparable. Female patients were more sensitive to acetylcholine with vasomotor dysfunction occurring at lower acetylcholine doses compared with male patients. Smoking is a risk factor for VSA, unlike diabetes and hypertension, and the relationship with dyslipidaemia is unclear.46,47
Pathophysiology and endotypes
MICROVASCULAR ANGINA AND EPICARDIAL CORONARY ARTERY SPASM
In the absence of flow-limiting coronary artery disease, myocardial ischaemia can result from specific pathways of microcirculatory dysfunction.16 Two microcirculatory dysfunction endotypes account for most cases of MVA: structural microcirculatory remodelling and functional arteriolar dysregulation. In other words, microvascular dyfsunction may be structural, functional or both.16,48
(i) Structural remodelling of the coronary microvasculature is associated with a decrease in microcirculatory conductance and impaired oxygen delivery capacity.49 This is typically caused by inward remodelling of coronary arterioles, with an increase in wall to lumen ratio, loss of myocardial capillary density (capillary rarefaction) or both.50 Remodelling may occur as a result of cardiovascular risk factors, atherosclerosis, left ventricular hypertrophy, or cardiomyopathies.50 A direct consequence of these pathological changes is a reduction of the vasodilatory range of the coronary microcirculation, limiting maximal blood and oxygen supply to the myocardium. Furthermore, remodelled arterioles are hypersensitive to vasoconstricting stimuli.51 The haemodynamic correlates of structural microcirculatory remodelling in response to a non-endothelium-dependent vasodilator, like adenosine, are (i) a reduced coronary flow reserve (CFR) and (ii) an increase in minimal (hyperaemic) microcirculatory resistance.
(ii) Functional arteriolar dysregulation typically takes place in medium and large size arterioles, in which flow-mediated vasodilation is predominant.16 Under physiological conditions, an increase in myocardial oxygen consumption generates an upstream vasodilatory cascade in coronary resistance vessels. This is initiated by metabolically triggered vasodilation of distal arterioles, that are particularly sensitive to certain metabolites, and it is followed by flow-mediated (endothelium-dependent) vasodilation of larger arterioles located up-stream, as well as epicardial vessels.52 In the presence of endothelial dysfunction, dysregulation of the described upstream vasodilatory cascade occurs. Thus, endothelial dysfunction is associated with impaired vasodilation and even paradoxical vasoconstriction of upstream arteries and arterioles when myocardial oxygen demands increase which may be the result of hypersensitivity to vasoconstrictor stimuli.53 Some of the haemodynamic correlates of arteriolar dysregulation, observed during intracoronary acetylcholine challenge, are (i) a limited vasodilatory response to the drug (less than 1.5 times resting flow), (ii) a marked reduction in blood flow, equivalent to the no-reflow phenomenon, without epicardial vessel spasm –denoting arteriolar spasm– and (iii) the development of diffuse narrowing of distal epicardial vessels without focal, tight coronary spasm. The above-mentioned changes frequently run along the development of anginal symptoms and ischaemic electrocardiogram changes, which confirm the ischaemia-generating potential of this endotype of micro-circulatory dysfunction. Effects of fluctuating oestrogen levels on epicardial vessel and arteriolar vasomotion have been postulated as explanations for a higher frequency of symptoms in premenopausal women without obstructive CAD.54