In this post I link to and excerpt from the Internet Book Of Critical Care [Link is to Table of Contents] chapter on Torsades de Pointes, November 22, 2016 by Dr. Josh Farkas.
First I copy Dr. Farkas’ Cardiac Magnesium Protocols*:
*Please note that the dosage that Dr. Farkas recommends in his protocol is different from the dose recommended in the 2020 Handbook of Emergency Cardiovascular Care For Healthcare Providers. In his chapter, Dr. Farkas explains his dosing recommendation.
Next, I excerpt the Magnesium Sulfate section from The 2020 Of Emergency Cardiac Care For Healthcare Providers:
| Drug/Therapy | Indications/Precautions | Adult Dosages |
| Magnesium Sulfate | Indications
– Recommended for use in cardiac arrest only if torsades de pointes or suspected hypomagnesemia is present – Life-threatening ventricular arrythmias due to digitals toxicity – Routine administration in hospitalized patients with AMI is not recomended. |
Cardiac Arrest (Due to Hypomagnesiemia or Torsades de Pointes
1-2 g (2-4 ml of a 50% solution diluted in 10 mL [eg, D5W, normal saline] given IV/IO |
| Precautions
– Occasional fall in blood pressure with rapid administration – Use with caution if renal failure present |
Torsades de Pointes With a Pulse or AMI With Hypomagnesemia
– Loading dose of 1 – 2 g mixed in 50 – 100 mL of diluent (eg, D5W, normal saline) over 5 – 60 minutes IV – Follow with 0.5 – 1 g per hour IV (titrate to control tosades)
|
Here are direct links that Dr. Farkas has provided us to all the sections of his chapter:
CONTENTS
And here are excerpts:
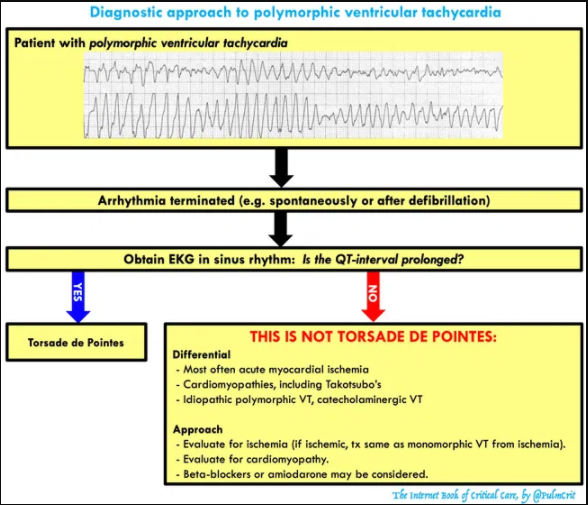
Diagnosis of torsades
getting some definitions straight
- Polymorphic ventricular tachycardia
- Defined as ventricular tachycardia with varying QRS amplitude.
- This is commonly referred to as torsades de pointes, but it’s actually not the same thing. Polymorphic ventricular tachycardia may be caused by several etiologies (e.g. congenital QT prolongation, acquired QT prolongation, ischemia, Takotsubo’s cardiomyopathy).
- Torsades de Pointes
- Torsades is defined as the combination of polymorphic ventricular tachycardia plus a prolonged QT-interval.
- Torsades can be caused by either congenital long-QT syndrome or acquired long-QT syndrome (due to electrolyte abnormalities and/or medications). The vast majority of torsades results from acquired long-QT syndrome, which is the focus of this chapter.
how to measure QT & QTc interval
- The EKG computer will generally get this right, but it cannot be relied upon entirely.
- QT may vary between leads. The lead with the longest easily measurable QT interval should be used.
- If there is a U-wave, the QT interval should be determined as the intersection of the T-wave maximum slope with the isoelectric line (figure above).
- The QTc may be determined using a calculator (e.g. MDCalc).
- QTc > 475 ms is statistically elevated (above the 99th percentile).1
- Torsades is unusual unless QTc is >500 ms.2
- Bundle branch blocks will prolong the QT without increasing the risk of torsades.3 Interpretation of QT intervals here is murky; consider comparing to prior EKGs.
clinical presentations
- Depending on the length of the episode and whether torsades degenerates into ventricular fibrillation, clinical presentations include:
- Syncope or presyncope
- “Seizure” episode(s)
- Cardiac arrest
- Telemetry monitoring during an episode makes the diagnosis more straightforward.
EKG differential diagnosis: things that masquerade as torsades
- Severe hyperkalemia with “sine wave” pattern
- Coarse ventricular fibrillation
- Atrial fibrillation with Wolf Parkinson White (varying morphology may create polymorphic appearance)
- Polymorphic ventricular tachycardia with normal QT interval (may be caused by acute myocardial ischemia, Takotsubo cardiomyopathy, or such genetic conditions as catecholaminergic VT).
Tx #1: breaking active polymorphic VT
unstable: immediate defibrillation
- Patients with unstable polymorphic VT need immediate defibrillation.
- The defibrillator will often be unable to lock onto any QRS complexes, so you generally need to perform an unsynchronized defibrillation.
- Technically the appropriate amount of energy is 200J biphasic. However, it might also be reasonable to use more energy particularly in a larger patient (with the rationale that unsuccessful defibrillation could be very dangerous, potentially pushing the patient into ventricular fibrillation).
“stable polymorphic VT”
- Polymorphic VT is never truly stable. This is a short-lived transitional state which usually flips back into sinus rhythm, or less commonly degenerates into ventricular fibrillation. Even if the patient looks OK, they’re not really stable: attach pads and don’t leave the room.
- Given the risk of degeneration into VF, urgent defibrillation is reasonable if the patient shows any hint of instability.
- Intravenous magnesium is a reasonable treatment, but a practitioner should be in the patient’s room with a defibrillator ready to shock the patient.
- Give 2 grams (8 mM) IV magnesium sulfate over 10-15 minutes, repeat if no response. If the patient remains stable and the rhythm persists, repeat an EKG and consider the possibility of an alternative diagnosis.
- Note that most treatments associated with torsades are used for the prevention of VT, not breaking an episode of VT. Thus treatments such as isoproterenol or pacing have no role here.
Tx #2: Basic tx to prevent recurrent torsades
If the EKG shows a prolonged QT-interval, the patient is diagnosed with torsades. If you simply break torsades but do nothing else, it is likely to recur. The following therapies will prevent recurrence of ventricular tachycardia in this situation:
magnesium = 1st line therapy
- Patients with torsades should receive magnesium, even if they have a normal magnesium level.
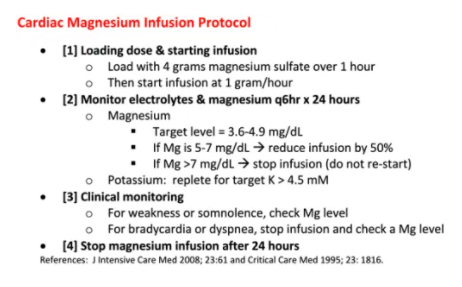
- Four grams magnesium sulfate IV (16 mM) is a reasonable place to start. Unfortunately, if you stop after four grams then the magnesium level will fall over several hours and torsades may recur.4
- Additional magnesium should be provided based on the patient’s renal function:
- (a) If GFR > 30 ml/min, a magnesium infusion is useful (see protocol below).
- (b) If GFR < 30 ml/min, cycle magnesium levels and bolus intermittently to target a magnesium level of 3.5-5 mg/dL (1.5-2 mM).
- Magnesium has a very wide safety margin. A protocoled magnesium infusion may seem aggressive, but overall this is far safer than the risk of recurrent cardiac arrest.
- Below is a magnesium protocol (one version in American units, one in SI units).5,6 This will work best if pasted directly into the patient chart so that everyone is literally on the same page (physicians, nurses, and pharmacists).
- The rationale for a magnesium infusion is explored further here.
potassium
- Aggressively treat any hypokalemia, targeting a high-normal potassium level (>4.5 mEq/L).7
cessation of all QT-prolonging medications
- The medication list should be carefully reviewed for any medications which may prolong QT interval. Patients are often on several QT-prolonging medications, all of which should be stopped if possible.
- Evidence regarding which drugs cause torsades is
- extremely murky. In some cases it seems that drugs have been incorrectly maligned (e.g. azithromycin, olanzapine). One complicating factor is that there isn’t a simple relationship between QT prolongation and torsades (some drugs such as amiodarone increase the QT without causing much torsades).
- A comprehensive list of medications implicated in causing torsades is here. The most common offenders are listed below.
more common medications linked to torsades2,3,7
- Antiarrhythmics
- Class IA: Quinidine, disopyramide, procainamide
- Class IC: Flecainide
- Class III: Dofetilide, ibutilide, sotalol, dronadarone
- Psychotropic
- Haloperidol, droperidol, chlorpromazine, pimozide
- Citalopram, escitalopram
- Tricyclic antidepressants
- Antibiotics
- Clarithromycin, erythromycin
- Fluoroquinolones
- Fluconazole, itraconazole, voreconazole, posaconazole
- Pentamidine
- Other
- Methadone
- Cocaine, loperamide (when abused in massive doses)
- Ondansetron
- Propofol
- Arsenic trioxide, sunitinib, vandetanib
Tx #3: Refractory torsades storm
Occasional patients will have recurrent episodes of torsades (“Torsades storm”). Each individual episode may be treated with magnesium or defibrillation, if needed (Treatment step #1 above). However, additional therapies are required to stop recurrence and end the storm.
re-load magnesium PRN
- Recurrent torsades may reflects inadequate magnesium dosing (e.g. patient is bolused with 2-4 grams, without an infusion). The first step when managing recurrent torsades is therefore to ensure that the patient has truly received an adequate dose of magnesium.
- If the patient was bolused with magnesium a few hours ago without an infusion, re-load with 2-4 grams IV immediately (8-16 mM).
- If the patient is a candidate for magnesium infusion (GFR >30 ml/hr), this should be ordered.
- If the patient has renal failure and has already received 4-6 grams of magnesium (16-24 mM), then check magnesium levels and ensure that a high level is achieved. Note that a therapeutic level for torsades is roughly 3.5-5 mg/dL (1.5-2 mM) – not a “normal” level.
- More on magnesium above (Treatment step #2).
optimize the potassium
- The target potassium level here is probably >4.5-4.7 mEq/L.
- Giving potassium alone is unlikely to work, but it might help a bit.
speed up the heart
- Speeding up the heart rate will decrease the QT interval and reduce the risk of acquired torsades (but it may be contraindicated in some forms of congenital torsades).
- The usefulness of chronotropy depends on the patient’s baseline heart rate.
- Chronotropy is most beneficial for patients starting out with bradycardia.
- If the patient is already significantly tachycardic, chronotropy is unlikely to provide benefit. The usual target heart rate is 100-110 b/m, but occasionally heart rates up to 140 b/m may be needed.2 There’s no high-quality data on this.
- Medical chronotropy is generally the easiest & fastest way to stabilize the patient. The ideal chronotrope depends on the patient’s hemodynamics and baseline blood pressure:
- Baseline severe hypotension: epinephrine infusion.
- Baseline normotension or mild hypotension: dobutamine or isoproterenol infusion.
- Electrical chronotropy may be used if medical chronotropy fails:
- Transcutaneous pacing may work, but this is painful for conscious patients.
- Transvenous pacing is more comfortable, but this is more invasive and takes a bit longer to achieve.
- Patients with a pacemaker may have the device rate increased.
lidocaine
- Lidocaine is the preferred anti-arrhythmic drug for torsades, although there isn’t a ton of evidence supporting its use.
- Do not use amiodarone, procainamide, beta-blockers, or most other antiarrhythmics. Most of these will stretch out the QT interval even further! Beta-blockers will slow down the heart rate, increasing the risk of torsades.
- Start with a loading dose of 1-1.5 mg/kg followed by a 1 mg/min infusion. For recurrent arrhythmias, re-load with another 1 mg/kg bolus and increase the maintenance infusion.
consider an alternative diagnosis
- Torsades is generally fairly easy to control with a combination of high-dose magnesium, heart rate augmentation, and occasionally some lidocaine. Failure to respond to these interventions suggests an alternative diagnosis (e.g. polymorphic VT due to ischemia or catecholaminergic ventricular tachycardia).
Pitfalls
- Under-dosing magnesium (e.g. giving two-four grams magnesium and walking away). This will often cause recurrence of ventricular tachycardia in a few hours after the serum magnesium levels fall.
- Leaving patients on QT-prolonging meds (make sure to scour the medication list for any problematic drugs).
- Don’t give amiodarone or other QT-prolonging antiarrhythmics (e.g. procainamide). These may actually aggravate the situation.
- Recognize that polymorphic VT with a normal QT interval isn’t torsades, this requires an entirely different treatment strategy.
Going further:
- New approach to Torsades (PulmCrit) & Prior post on Mg infusions
- QT-interval and Torsades & polymorphic VT (Ed Burns, LITFL)
- Podcast on torsades by Rob Orman and Joe Bellezzo (ERCast). This is a pretty epic episode and well worth listening to. I wholeheartedly agree with Joe Bellezzo regarding the need to do something pro-active to prevent recurrent torsades, but would suggest that a magnesium infusion can be used instead of temporary transvenous pacing.
- Polymorphic VT (Steve Smith’s ECG blog)
- Torsades de Pointes (Justin Morganstern, First 10 in EM)
- Evaluation of QTc drug interactions (Pharmacy Joe)
- EKG pointers: Is it Torsades de Pointes? (emDocs)