In this post I link to and excerpt from IBCC chapter – Disseminated Intravascular Coagulation (DIC). January 18, 2021 by Josh Farkas:
Disseminated Intravascular Coagulation (DIC) is like the ARDS of the hematological system. It’s not really one disorder, but rather a collection of different disorders with some shared features. The diagnosis and optimal treatment remain elusive.
Dr. Farkas gives us direct links to each part of his DIC chapter:
CONTENTS
Here are excerpts:
pathophysiology
general concept of DIC
The fundamental pathophysiologic signature of DIC is widespread, uncontrolled clot formation. Clotting may be initiated by a variety of factors (e.g., bacterial lipopolysaccharides, tissue factor released by monocytes or the placenta, or damage to the endothelial glycocalyx).
different flavors of DIC
Different causes of DIC tend to cause different problems. Infection and septic shock tend to cause a more prothrombotic form of DIC, which predominantly causes excess clot formation. Alternatively, DIC due to obstetric catastrophes or leukemia tends to cause depletion of fibrinogen leading to hemorrhage. Consequently, DIC is not a monolithic construct – and appropriate treatment often depends on understanding the context.
symptoms
DIC may be asymptomatic, or it may cause bleeding and clotting. Symptoms may be dominated by either bleeding or clotting.
microvascular thrombosis causes organ failure
- Microvascular thromboses may be an occult cause of organ failures. With the exception of the skin, it is often difficult or impossible to tell that microvascular thrombosis is occurring. The most common manifestations are:
- (1) Renal failure is probably the most common.
- (2) Acute respiratory distress syndrome (ARDS).
- (3) Brain involvement may cause delirium, coma, seizure.
- (4) Adrenal failure (Waterhouse-Friedrichsen syndrome) may result from microvascular thromboses, followed by hemorrhagic transformation. The net result is adrenal failure which can precipitate adrenal crisis.
- (5) Skin: purpura fulminans (occlusion of microvasculature in the skin potentially causing gangrene). This signifies an extreme form of DIC which requires aggressive management (more on this here)
macrovascular thrombosis
- Primarily causes venous thrombosis (e.g., deep vein thrombosis, pulmonary embolism).
bleeding
- Petechiae and purpura may occur on the skin.
- Oozing may originate from intravenous catheters or mucus membranes.
- Life-threatening hemorrhage can result from gastrointestinal or intracranial hemorrhage. However, in most forms of DIC, severe bleeding is uncommon.
laboratory studies
DIC screening lab panel
- Complete blood count (CBC)
- INR or PT, PTT
- Fibrinogen
- D-dimer
D-dimer
- D-dimer is invariably elevated in DIC, often dramatically so (e.g., >4,000 ng/mL).
- A normal D-dimer essentially excludes DIC.
- Advanced sepsis-induced DIC may impair fibrinolysis (“fibrinolytic shutdown”), which limits further increases in D-dimer.
thrombocytopenia
- Moderate thrombocytopenia is usually seen in DIC. Platelet counts of <30,000/uL are uncommon (25413378, 22735856, 26308340, 23159146, 19581801)
- Thrombocytopenia may be the most sensitive laboratory test for detection of DIC.(30828800) A down-trending platelet count is often the first laboratory abnormality which to be noted.(30634199) Of course, thrombocytopenia is not specific to DIC: thrombocytopenia is very common in the ICU due to a variety of causes (more on causes of thrombocytopenia here).
INR and PTT abnormalities
- Elevations of INR and PTT support a diagnosis of DIC. However, they can be normal in over half of patients with DIC.(30634199)
- These abnormalities may not correspond to clinical hypocoagulability (since they measure only clotting factors, but do not reflect deficiencies which may be occurring in anticoagulant factors, such as protein C and protein S).
- In sepsis-induced DIC, PTT prolongation may lag behind PT prolongation as DIC develops. This occurs because the PTT is reduced by elevated factor VIII levels. (27578502)
fibrinogen
- 🔑 The fibrinogen is low only in about a quarter of patients with DIC.(30634199) Low fibrinogen supports a diagnosis of DIC, but this usually isn’t seen.
- Low fibrinogen is more common in predominantly fibrinolytic types of DIC (e.g., patients with acute promyelocytic leukemia or obstetric-related DIC).
- Sepsis increases fibrinogen levels, so sepsis-induced DIC may have elevated or normal levels of fibrinogen. Falling fibrinogen may suggest ongoing DIC with consumption of fibrinogen.
- 🔑 Changes in fibrinogen over time may be more illuminative than a single value.
microangiopathic hemolytic anemia (MAHA) can occur
- Microangiopathic hemolytic anemia can occur due to microthrombi in the capillaries. This causes intravascular hemolysis with the generation of schistocytes (fragmented RBCs).
causes of DIC
Certain forms of DIC tend to cause predominantly thrombosis or hemorrhage – these are indicated below.
[For details on each of these set of causes below click on causes of DIC]
sepsis due to a wide variety of pathogens, most notably:
direct tissue damage
malignancy, especially:
- Acute promyelocytic leukemia (AML-M3) and monocytic leukemia (AML-M5) – Tend to cause a hyperfibrinolytic form of DIC, with prominent bleeding. More on the management of acute promyelocytic leukemia here.
- Adenocarcinoma (especially pancreas, prostate, lung, gastric, ovary). This is usually a chronic, compensated form of DIC with a tendency towards thrombosis.
- Chemotherapy.
obstetric catastrophe (hemorrhage often predominates)
- Placental abruption
- Amniotic fluid embolism
- Preeclampsia, HELLP syndrome
- Fetal demise
- Maternal septic shock (e.g., chorioamnionitis, group A streptococcus infection)
- Acute fatty liver of pregnancy
other
- Post-Cardiopulmonary Resuscitation (thrombosis predominates)(29255070)
- Severe collagen vascular disease, vasculitis
- Drug-induced
- Heparin-Induced Thrombocytopenia (HIT) can cause DIC in ~10% of cases (although the two processes are usually separate)
- Severe hepatitis
- Vascular malformations (e.g., giant hemangiomas or large aortic aneurysms) – hemorrhage predominates.
- Catastrophic antiphospholipid antibody syndrome
- Hemophagocytic lymphohistiocytosis (HLH)
differential diagnosis
Isolated sepsis-induced thrombocytopenia vs. DIC
- Sepsis may cause thrombocytopenia via a variety of mechanisms:(29255070)
- Impaired platelet production
- Increased platelet consumption, (sometimes due to hemophagocytosis)
- Splenic sequestration of platelets
- Septic patients often have thrombocytopenia without other abnormalities in their coagulation factors (e.g., normal INR and PTT). Such patients do not have DIC.
cirrhosis vs. DIC
- Cirrhosis is a risk factor for DIC, because patients with cirrhosis often have reduced levels of coagulation factors and also anti-coagulant proteins (e.g., protein C and protein S). With lower levels of these proteins at baseline, patients with cirrhosis have a delicate balance of procoagulants vs. anticoagulants, which may be more easily disrupted by an acute insult. Thus, cirrhosis may often coexist with DIC. However, the two processes are usually separate and can be sorted out.
- Abnormalities which can occur due to cirrhosis:
- Cirrhosis commonly causes thrombocytopenia and reductions in clotting factor levels (e.g., elevation of INR).
- Rarely, patients with cirrhosis may fail to clear endogenous tPA, leading to ongoing fibrinolysis (accelerated intravascular coagulation and fibrinolysis AICF) – a condition which can closely mimic DIC.
- Diagnostic clues to sort out DIC vs. cirrhosis
- (1) Comparison to baseline coagulation factors: if coagulation tests (e.g., thrombocytopenia) are at their baseline level, then acute DIC is unlikely.(29255070)
- (2) Most patients with cirrhosis have only mildly elevated D-dimer. Thus, profoundly elevated D-dimer may favor DIC. However, markedly elevated D-dimer also does occur in accelerated intravascular coagulation and fibrinolysis (AICF), as a complication of cirrhosis (more on this here)(REF).
- (3) Factor VIII levels: Factor VIII is produced by endothelial cells. In cirrhosis, factor VIII levels will remain normal or elevated. Alternatively, in DIC, factor VIII will be reduced along with other coagulation factors.
HIT (heparin-induced thrombocytopenia) vs. DIC
- A useful feature to sort out HIT vs. DIC is chronicity. DIC usually occurs gradually, beginning around the time of admission. Alternatively, HIT tends to begin abruptly, more than five days after exposure to heparin. The 4-T score may be used to more precisely quantify the risk of HIT (below).
- Most patients with HIT do not have DIC. However, severe HIT may trigger DIC in ~10% of cases.
- HIT alone typically causes the following pattern of findings:
- Platelet counts aren’t profoundly reduced (e.g., platelets >20,000/uL)
- INR and fibrinogen levels are normal.
- HIT complicated by DIC may cause the following pattern:
- Platelets may be profoundly reduced (e.g., platelets <20,000/uL)
- INR prolongation and hypofibrinogenemia may occur
- More on HIT here.
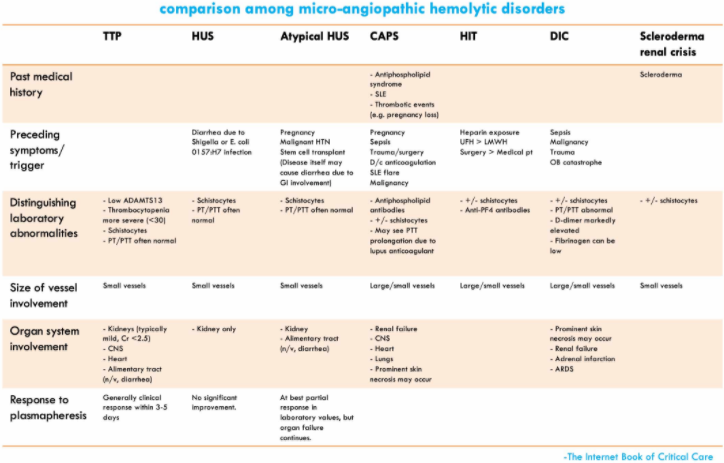
primary thrombotic microangiopathies (e.g., TTP, HUS) vs. DIC
- Both DIC and thrombotic microangiopathies may lead to anemia, schistocyte formation, and thrombocytopenia. However, schistocyte formation is considerably more exuberant in primary thrombotic microangiopathy, compared to DIC.
- Patients with TTP, HUS, or drug-induced thrombotic microangiopathy should have normal coagulation factors (e.g., INR, PTT, fibrinogen), because clots are composed of platelets and don’t involve activation of the coagulation system. Similarly, the D-dimer should be normal or only slightly elevated.(29255070)
catastrophic antiphospholipid antibody syndrome (CAPS) vs. DIC
- This can be challenging, because CAPS causes DIC in ~25% of cases.
- Features which may suggest CAPS include:
- A history of connective tissue disease (especially antiphospholipid antibody syndrome).
- A history of pregnancy loss, or illness onset during pregnancy.
- Recent withdrawal of anticoagulation.
- Skin manifestations (e.g., livedo reticularis or cutaneous necrosis).
- More on the diagnosis of CAPS here.
diagnostic criteria of DIC
DIC is a clinical diagnosis
- No single lab test is diagnostic of DIC. Rather, the diagnosis of DIC rests roughly on three components:
- (1) Constellation of laboratory abnormalities which is consistent with DIC.
- (2) Presence of an underlying disorder known to cause DIC (e.g., trauma, sepsis).
- (3) Exclusion of an alternative explanation for coagulation abnormalities (e.g., see differential diagnosis section above).
DIC scoring systems can help determine if coagulation abnormalities are consistent with DIC
- Several DIC scoring systems exist. The International Society of Thrombosis and Hemostasis (ISTH) score (shown below) is the most widely utilized and accepted.
- Scoring systems can help provide an objective metric to evaluate the patient’s laboratory abnormalities, but they are neither the end-all nor be-all of diagnosis.
- Early in the process of DIC, the patient may not quite reach a positive DIC score (sometimes called “non-overt DIC”). If there is ongoing concern regarding DIC, coagulation labs may be repeated in 12-24 hours to determine if the patient is developing DIC. Often changes in serial labs may be more illuminating than a single set of labs (e.g., a fibrinogen which is actively falling from 300 mg/dL to 100 mg/dL is more worrisome than a fibrinogen which is stably low at 100 mg/dL).
sepsis induced coagulopathy (SIC)
[Click on the link above for details from this part of the Chapter]
management
some specific types of DIC require different management:
- Patients with DIC due to acute promyelocytic leukemia (APML) require specific treatment described here.
- Patients with purpura fulminans require a specific management strategy described here.
- Patients with pregnancy-associated DIC and clinical bleeding often need aggressive factor replacement, with an emphasis on maintaining adequate fibrinogen levels.
treat the underlying disorder
- The most important principle of DIC management is to treat the underlying cause.
- DIC due to septic shock is perhaps the most notable example of this – treatment should overall focus on the basic tenets of sepsis care.
platelet transfusion
- Platelet transfusion should generally be avoided, unless:
- Platelets <10,000/uL.
- There is a planned procedure with a substantial risk of bleeding.
- There is active bleeding.
- For patients with active bleeding or a pending procedure, transfusion to >30,000-50,000/uL may be reasonable.(29255070)
- Due to ongoing platelet consumption, results from platelet transfusion may be disappointing.
fibrinogen supplementation
- Profoundly low fibrinogen levels may increase the risk of bleeding, including intracranial hemorrhage.
- It may be reasonable to attempt to maintain fibrinogen levels above >~50-80 mg/dL (a target that may be personalized to some extent, depending on the risk of bleeding vs. thrombosis).
- For patients with active bleeding or a planned procedure, targeting a fibrinogen level >~150 mg/dL may be reasonable. Various sources recommend targets ranging from >100 mg/dL to >200 mg/dL, with little supporting evidence. The target may be individualized for a specific patient, when considering the overall risks of hemorrhage and the state of coagulation (e.g., for patients with marked thrombocytopenia, a higher fibrinogen target may compensate for the thrombocytopenia to a certain degree).
- Fibrinogen levels may be supported using cryoprecipitate or fibrinogen concentrates.
Coagulation factor replacement (e.g., fresh frozen plasma)
- This is confusing, because traditional labs (e.g., INR) don’t reflect the true coagulation tendency of the blood. INR measures only clotting factors (while ignoring endogenous anticoagulants such as protein C and protein S).
- Thromboelastography is likely a better tool to determine the true balance of coagulation. The use of thromboelastography for patients with DIC has not been specifically studied. However, thromboelastography has a proven track record for management of patients with enormously complex coagulopathies in the operating room (e.g., hepatic transplantation, multiple trauma patients, cardiopulmonary bypass operations) – many of whom doubtless had DIC. Thus, it is a logical extension that thromboelastography could be used to tailor blood product selection in the intensive care unit.
- When possible, it may be better not to treat INR and PTT values. However, in patients with active bleeding or planned procedures, factor replacement is indicated if thromboelastography reveals enzymatic coagulopathy (e.g., with prolonged R-time). If thromboelastography isn’t available, INR may need to be used as a blunt indicator of whether coagulation factors might potentially be needed.
- Fresh frozen plasma (FFP) has traditionally been used for coagulation factor repletion. However, prothrombin complex concentrates may be another option (especially in patients with volume overload).
vitamin K administration
- It may be difficult to determine whether vitamin K deficiency exists (since INR prolongation results from DIC).
- If vitamin K deficiency is suspected, then vitamin K should be administered empirically (e.g., 10 mg intravenously infused over 30-60 minutes).
DVT prophylaxis
- DVT prophylaxis should be provided to all critically ill patients, unless contraindicated. Contraindications may include:
- Active bleeding.
- Profound thrombocytopenia (e.g., platelet count <30,000/uL).(30828800)
- Profound hypofibrinogenemia (e.g., fibrinogen <80 mg/dL).
- Planned procedure with high concern regarding bleeding (e.g., neurosurgery, lumbar puncture).
- Subgroup analysis of the SCARLET and KYBERSEPT trials investigating antithrombin III and thrombomodulin anticoagulation suggest that these anticoagulants were potentially beneficial – but only among patients not taking concomitant heparin. This indirectly suggests that heparin could provide benefits analogous to more expensive, designer anticoagulants.(31988789)
therapeutic heparin anticoagulation
- Rationale:
- Theoretically, anticoagulation with heparin could impede ongoing thrombosis and thereby stop the primary abnormality of DIC (disseminated activation of coagulation).
- Unfortunately, research into therapeutic heparin anticoagulation has generally been unimpressive. Thus, heparin anticoagulation isn’t indicated in most cases of DIC.
- Indications for heparin anticoagulation:
- (1) Clinical thrombosis (e.g., DVT or pulmonary embolism).
- (2) Purpura fulminans (this subset of DIC requires an entirely different treatment strategy; more on this here).
- When possible, heparin infusions should be monitored using an anti-Xa level. Patients will often have an elevated PTT at baseline, which may confuse the use of PTT to monitor heparin dosing.
- Patients with DIC may often have heparin resistance, due to low levels of antithrombin III. Higher doses of heparin may be required than usual. More on heparin resistance here.