An outstanding brief review of the article below is The Cribsiders #16: Kawasaki Disease with Recrudescent Guest Dr. Tremoulet. JANUARY 6, 2021 By DR JUSTIN BERK. Dr. Tremulent’s podcast and show notes are a brilliant summary of the resource just below from the American Heart Association.
In this post, I link to and excerpt from 2017 Diagnosis, Treatment, and Long-Term Management of Kawasaki Disease: A Scientific Statement for Health Professionals From the American Heart Association [PubMed Abstract] [Full-Text HTML] [Full-Text PDF]. Circulation. 2017 Apr 25;135(17):e927-e999.
All that follows is from the above outstanding resource.
Abstract
Background:
Kawasaki disease is an acute vasculitis of childhood that leads to coronary artery aneurysms in ≈25% of untreated cases. It has been reported worldwide and is the leading cause of acquired heart disease in children in developed countries.
Methods and Results:
To revise the previous American Heart Association guidelines, a multidisciplinary writing group of experts was convened to review and appraise available evidence and practice-based opinion, as well as to provide updated recommendations for diagnosis, treatment of the acute illness, and long-term management. Although the cause remains unknown, discussion sections highlight new insights into the epidemiology, genetics, pathogenesis, pathology, natural history, and long-term outcomes. Prompt diagnosis is essential, and an updated algorithm defines supplemental information to be used to assist the diagnosis when classic clinical criteria are incomplete. Although intravenous immune globulin is the mainstay of initial treatment, the role for additional primary therapy in selected patients is discussed. Approximately 10% to 20% of patients do not respond to initial intravenous immune globulin, and recommendations for additional therapies are provided. Careful initial management of evolving coronary artery abnormalities is essential, necessitating an increased frequency of assessments and escalation of thromboprophylaxis. Risk stratification for long-term management is based primarily on maximal coronary artery luminal dimensions, normalized as Z scores, and is calibrated to both past and current involvement. Patients with aneurysms require life-long and uninterrupted cardiology follow-up.
Conclusions:
These recommendations provide updated and best evidence-based guidance to healthcare providers who diagnose and manage Kawasaki disease, but clinical decision making should be individualized to specific patient circumstances.
____________________________________________
Kawasaki disease (KD) is an acute, self-limited febrile illness of unknown cause that predominantly affects children <5 years of age. When initially described, the potential for coronary artery complications was not appreciated. KD is now the most common cause of acquired heart disease in children in developed countries. In the absence of pathognomonic tests, the diagnosis continues to rest on the identification of principal clinical findings and the exclusion of other clinically similar entities with known causes. Timely initiation of treatment with intravenous immunoglobulin (IVIG) has reduced the incidence of coronary artery aneurysms defined from absolute luminal dimensions from 25% to ≈4%. Ongoing studies with additional therapies have not substantially reduced this residual risk. The long-term prognosis is determined by the initial and current level of coronary artery involvement. Certain subsets of patients are at risk for myocardial ischemia from coronary artery thrombosis and stenoses. Medical management of such patients hinges on judicious use of thromboprophylaxis and vigilance to identify evolving stenoses. Invasive revascularization procedures might be required for selected patients.
Key Points: Epidemiology
The cause is unknown.
The estimated incidence in North America is ≈25 cases per 100 000 children <5 years of age per year.
The highest relative risk is in Asian children, especially of Japanese ancestry.
The ratio of males to females is ≈1.5:1.
KD affects predominantly, but not exclusively, young children.
It is most common in winter and early spring in North America.
Predisposing factors have been reported inconsistently.
Nonspecific symptoms are common in the 10 days before diagnosis.
In Japan, the recurrence rate is ≈3%, and the relative risk in siblings is 10-fold higher.
The case fatality rate is <0.1% in Japan.
Coronary artery aneurysms from KD account for 5% of acute coronary syndromes (ACS) in adults <40 years of age.
Pathological outcomes of coronary artery damage depend on the severity of the lesions. Very mildly dilated and inflamed arteries may be able to return to normal. Large saccular aneurysms have lost their intima, media, and elastica, which cannot be regenerated. The rim of remaining adventitia can rupture or undergo sequential thrombosis that can organize, recanalize, and calcify. Fusiform aneurysms with partially preserved media can thrombose or develop progressive stenosis from LMP. Large aneurysms can appear to “resolve” when the lumen size decreases because of layered mural thrombi or LMP. The largest aneurysms (“giant aneurysms”) have generally lost virtually all of the media, with only a rim of adventitia remaining. These aneurysms develop successive layers of thrombi, with organization and calcification of the oldest thrombi closest to the remaining adventitia. Giant aneurysms can rupture in the first 2 to 3 weeks after fever onset but rarely do so thereafter. MI can occur from acute or progressive thrombosis or from stenosis caused by LMP.73 A recent study of pediatric vasculitis fatalities over the past 50 years from Japan indicated that the vast majority of such deaths were the result of KD and that fatality rates markedly decreased around the time IVIG therapy was introduced, in the mid to late 1980s.78
Key Points: Pathology
KD vasculopathy primarily involves muscular arteries and is characterized by 3 linked processes: (1) necrotizing arteritis; (2) subacute/chronic vasculitis; and (3) LMP.
Large or giant coronary artery aneurysms ≥8 mm in diameter or with a Z score ≥10 do not “resolve,” “regress,” or “remodel.” They rarely rupture and virtually always contain thrombi (the oldest of which may calcify) that can become occlusive.
Aneurysms with markedly damaged but partially preserved media may develop decreases in lumen diameter over time as the result of LMP or thrombi and can become progressively stenotic.
Atherosclerotic features are not characteristic of KD vasculopathy even in late deaths or transplants.
Pericarditis and myocarditis result from subacute/chronic inflammation, which is usually concentrated around coronary arteries.
Diagnosis
Clinical criteria are used to diagnose KD.1,79Table 3 describes the clinical features that constitute the epidemiological case definition, as well as other clinical and laboratory findings. Patients who meet the case definition based on principal clinical findings are said to have complete KD (also sometimes referred to as typical or classic KD). Patients who do not have sufficient principal clinical findings may be diagnosed with incomplete KD (also sometimes referred to as atypical KD). In the absence of a specific diagnostic test, other clinical, laboratory, and echocardiographic findings can support the diagnosis of incomplete KD in a patient whose clinical presentation suggests KD but whose clinical features do not meet the epidemiological case definition.
Principal Clinical Findings
The diagnosis of classic KD is based on the presence of ≥5 days of fever (first calendar day of fever is illness day 1) and the presence of ≥4 of the 5 principal clinical features (Table 3, Figure 2).1 In the presence of ≥4 principal clinical criteria, particularly when redness and swelling of the hands and feet are present, the diagnosis may be made with only 4 days of fever. Similarly, experienced clinicians who have treated many KD patients may make the diagnosis in rare instances with only 3 days of fever in the presence of a classic clinical presentation. Typically the clinical features are not all present at a single point in time, and it is generally not possible to establish the diagnosis very early in the course. Similarly, some clinical features may have abated in patients who present after 1 to 2 weeks of fever, and a careful review of prior signs and symptoms can help establish the diagnosis.
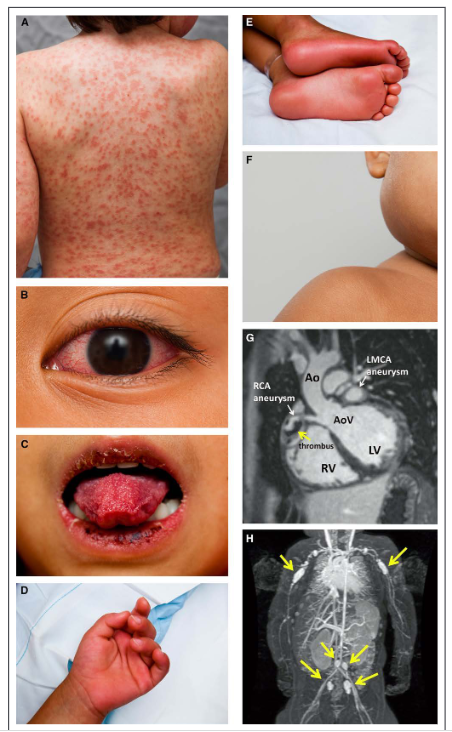
Figure 2. Clinical features of classic Kawasaki disease.A, Rash: Maculopapular, diffuse erythroderma, or erythema multiforme-like. B, Conjunctivitis: Bulbar conjunctival injection without exudate; bilateral. C, Oral changes: Erythema and cracking of lips (cheilitis); strawberry tongue; erythema of oral and pharyngeal mucosa. D and E, Palmar and plantar erythema: Usually accompanied by swelling; resolves with subsequent periungual desquamation in the subacute phase. F, Cervical adenopathy: Usually unilateral, node ≥1.5 cm in diameter. G, Coronary artery aneurysms: Magnetic resonance image of the left ventricular outflow tract showing a giant right coronary artery (RCA) aneurysm with nonocclusive thrombus (yellow arrow) and a giant left main coronary artery (LMCA) aneurysm. Ao indicates aorta; AoV, aortic valve; LV, left ventricle; and RV, right ventricle. H, Peripheral artery aneurysms: Magnetic resonance image showing aneurysms in the axillary and subclavian arteries and the iliac and femoral arteries (yellow arrows). Patient photographs used with permission from the Kawasaki Disease Foundation, Inc.
Incomplete (Atypical) KD
Although the presence of fever for ≥4 days with 4 of the 5 other principal clinical findings establishes the diagnosis of complete KD, these criteria unfortunately do not identify all children with the illness. KD should be considered in the differential diagnosis of prolonged unexplained fever in childhood associated with any of the principal clinical features of the disease, and the diagnosis can be considered confirmed when coronary artery aneurysms are identified in such patients by echocardiography. However, coronary artery dilatation is generally not detected by echocardiography until after the first week of illness, and a normal echocardiogram in the first week of illness does not rule out the diagnosis of KD. Patients with incomplete KD, particularly those <6 months of age and those lacking eye or oral mucosal changes, may experience significant delays in diagnosis.92 Studies evaluating the incomplete KD diagnostic algorithm first proposed in the 2004 guidelines1 suggest its usefulness in identifying patients who require treatment and in preventing coronary artery aneurysms.93,94 Incomplete KD occurs most commonly in infants, who are at substantial risk of developing coronary artery abnormalities and who may have prolonged fever as the sole clinical finding or have subtle or fleeting clinical signs in addition to fever. Laboratory findings and cardiovascular sequelae in incomplete and complete cases appear the same. Although there are no pathognomonic laboratory findings, the presence of certain laboratory features may raise the clinical suspicion of KD. The finding of coronary artery Z scores (based on body surface area [BSA]) of ≥2.5 for the left anterior descending (LAD) or right coronary artery (RCA) branches lacks sensitivity but has a very high specificity for the diagnosis.95,96
Diagnosis of Incomplete KD
The diagnosis of incomplete (sometimes referred to as atypical) KD should be considered in any infant or child with prolonged unexplained fever, fewer than 4 of the principal clinical findings, and compatible laboratory or echocardiographic findings (Figure 3).
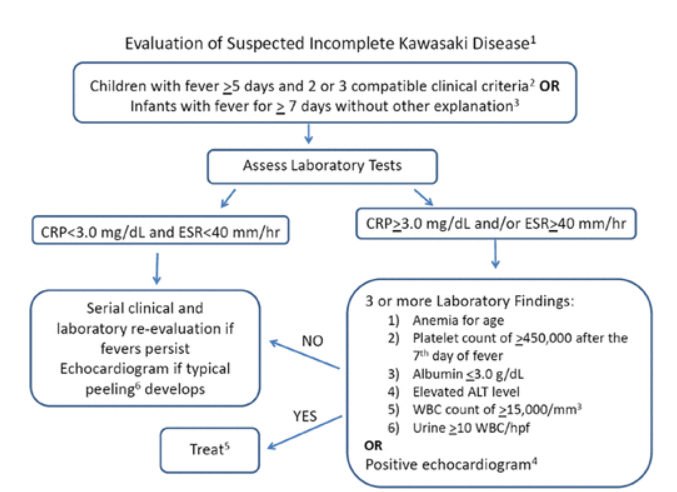
Figure 3. Evaluation of suspected incomplete Kawasaki disease. (1) In the absence of a “gold standard” for diagnosis, this algorithm cannot be evidence based but rather represents the informed opinion of the expert committee. Consultation with an expert should be sought any time assistance is needed. (2) Clinical findings of Kawasaki disease are listed in Table 3. Characteristics suggesting that another diagnosis should be considered include exudative conjunctivitis, exudative pharyngitis, ulcerative intraoral lesions, bullous or vesicular rash, generalized adenopathy, or splenomegaly. (3) Infants ≤6 months of age are the most likely to develop prolonged fever without other clinical criteria for Kawasaki disease; these infants are at particularly high risk of developing coronary artery abnormalities. (4) Echocardiography is considered positive for purposes of this algorithm if any of 3 conditions are met: Z score of left anterior descending coronary artery or right coronary artery ≥2.5; coronary artery aneurysm is observed; or ≥3 other suggestive features exist, including decreased left ventricular function, mitral regurgitation, pericardial effusion, or Z scores in left anterior descending coronary artery or right coronary artery of 2 to 2.5. (5) If the echocardiogram is positive, treatment should be given within 10 days of fever onset or after the tenth day of fever in the presence of clinical and laboratory signs (C-reactive protein [CRP], erythrocyte sedimentation rate [ESR]) of ongoing inflammation. (6) Typical peeling begins under the nail beds of fingers and toes. ALT indicates alanine transaminase; and WBC, white blood cells.
Common Pitfalls in Diagnosis
A high index of suspicion for the diagnosis is particularly important in certain clinical situations. In the infant <6 months of age, prolonged fever and irritability may be the only clinical manifestations of KD, and these children are at high risk of developing coronary artery abnormalities. Delayed diagnosis is common in older children and adolescents with KD, and they appear to have a high prevalence of coronary artery abnormalities.97 The presence of fever and pyuria in an infant or young child can be mistakenly attributed to a urinary tract infection, and subsequent development of rash, red eyes, and red lips to an antibiotic reaction. Likewise, irritability and a culture-negative pleocytosis of the cerebrospinal fluid in an infant with prolonged fever suggestive of aseptic meningitis (or if antibiotics have been given, partially treated meningitis) may cause a diagnosis of KD to be overlooked. Patients with cervical lymphadenitis as the primary clinical manifestation can be misdiagnosed as having bacterial adenitis, and many such patients will have concurrent retropharyngeal phlegmon that is attributed to bacterial infection.84 Patients with prominent gastrointestinal symptoms are sometimes admitted to a surgical service, and other physical findings of KD can be overlooked. Patients who present with shock may be misdiagnosed as having bacterial sepsis or staphylococcal or streptococcal toxic shock syndrome. In these clinical scenarios, consultation with an expert in the diagnosis of KD can be useful.
Key Points: Consider KD in the Differential Diagnosis of Certain Infants or Children
Infants <6 months old with prolonged fever and irritability
Infants with prolonged fever and unexplained aseptic meningitis
Infants or children with prolonged fever and unexplained or culture-negative shock
Infants or children with prolonged fever and cervical lymphadenitis unresponsive to antibiotic therapy
Infants or children with prolonged fever and retropharyngeal or parapharyngeal phlegmon unresponsive to antibiotic therapy
Evaluation for Cardiovascular Abnormalities
Echocardiography
Echocardiography is the primary imaging modality for cardiac assessment because it is noninvasive and has a high sensitivity and specificity for the detection of abnormalities of the proximal coronary artery segments.134 The initial echocardiogram should be performed as soon as the diagnosis is suspected, but initiation of treatment should not be delayed by the timing of the study. Because detailed echocardiographic imaging is compromised if a child is uncooperative, sedation is frequently needed for those <3 years of age and may also be required in older, irritable children.135 If a poor-quality initial echocardiogram is obtained because sedation was not administered, a sedated study should be repeated as soon as possible within the 48 hours after diagnosis and initial treatment. This initial study establishes a baseline for longitudinal follow-up monitoring of coronary artery morphology, LV wall motion, valvular regurgitation, and pericardial effusion. An initial echocardiogram in the first week of illness is typically normal and does not rule out the diagnosis.
Imaging Standards
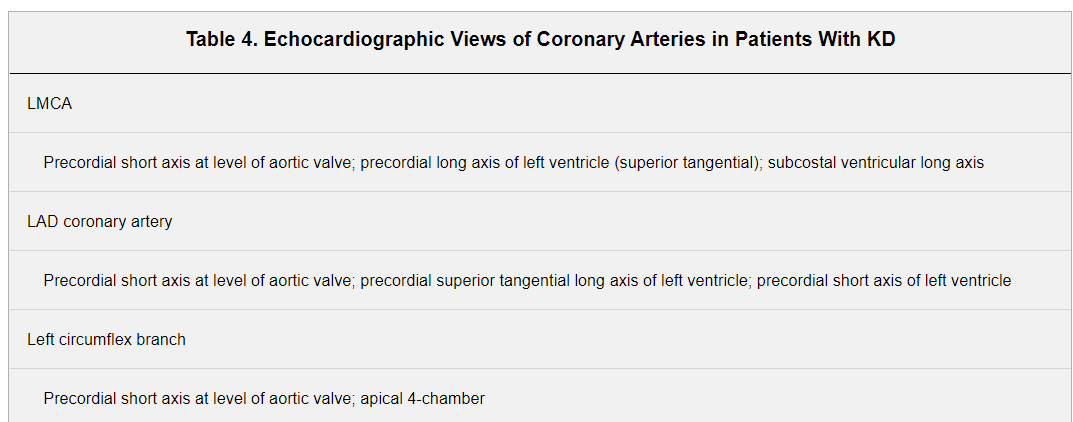
Echocardiography should be performed with equipment with appropriate transducers and should be supervised by an experienced pediatric echocardiographer. The 2-dimensional (2D) imaging should be performed with the highest-frequency transducer possible, even for older children, because these probes allow for high-resolution detailed evaluation of the coronary arteries. Studies should be recorded in a dynamic video or digital cine format that enables future review and comparison with subsequent studies. In addition to standard anatomic and physiological imaging from parasternal, apical, subcostal, and suprasternal notch windows, 2D echocardiographic evaluation of patients with suspected KD should focus on imaging the left main coronary artery (LMCA), LAD, left circumflex, RCA (proximal, middle, and distal segments), and posterior descending coronary arteries. Multiple imaging planes and transducer positions are required for the optimal visualization of all major coronary segments (Table 4).136 Maximal efforts should be made to visualize all major coronary artery segments. In order of highest to lowest frequency of occurrence, typical sites of coronary artery aneurysms include the proximal LAD and proximal RCA, followed by the LMCA, left circumflex, distal RCA and, least often, the junction between the RCA and posterior descending coronary artery.
Recommendations for Cardiovascular Assessment for Diagnosis and Monitoring During the Acute Illness
Echocardiography should be performed when the diagnosis of KD is considered, but unavailability or technical limitations should not delay treatment (Class I; Level of Evidence B).
Coronary arteries should be imaged, and quantitative assessment of luminal dimensions, normalized as Z scores adjusted for body surface, should be performed (Class I; Level of Evidence B).
For uncomplicated patients, echocardiography should be repeated both within 1 to 2 weeks and 4 to 6 weeks after treatment (Class I; Level of Evidence B).
For patients with important and evolving coronary artery abnormalities (Z score >2.5) detected during the acute illness, more frequent echocardiography (at least twice per week) should be performed until luminal dimensions have stopped progressing to determine the risk for and presence of thrombosis (Class I; Level of Evidence B).
To detect coronary artery thrombosis, it may be reasonable to perform echocardiography for patients with expanding large or giant aneurysms twice per week while dimensions are expanding rapidly and at least once weekly in the first 45 days of illness, and then monthly until the third month after illness onset, because the failure to escalate thromboprophylaxis in time with the rapid expansion of aneurysms is a primary cause of morbidity and mortality (Class IIa; Level of Evidence C).
Treatment of the Acute Illness
Initial Treatment of KD
The goal of therapy in the acute phase is to reduce inflammation and arterial damage and to prevent thrombosis in those with coronary artery abnormalities. The original guidelines for diagnosis of KD were created by a committee appointed by the Japanese Ministry of Health in 1970, at which time the coronary artery complications of KD were not yet appreciated and there was neither effective treatment nor a noninvasive method of assessing coronary artery abnormalities. The case definition was created, therefore, for epidemiological surveillance and has evolved over time. The mainstay of initial treatment for both complete and incomplete KD is a single high dose of IVIG together with acetylsalicylic acid (ASA), which is supported by clinical trial evidence.127,157
This section covers treatment from onset of the acute illness until resolution of acute systemic inflammation and when coronary artery luminal dimensions have stabilized and are no longer expanding.
Patient Selection for Treatment
All patients meeting the AHA diagnostic criteria for KD (Table 3) should be treated as soon in the course of illness as the diagnosis can be established.
Although the current case definition provides a specific tool for epidemiological surveillance, it might not be optimal for aiding clinicians in the recognition of children with a systemic vasculitis that requires prompt treatment. An algorithm to aid the clinician in deciding which patients with fever and fewer than 4 classic criteria, that is, suspected incomplete KD, should be treated is summarized in Figure 3. Use of the algorithm appears to be largely successful in ensuring that children at highest risk are treated with IVIG.93,94 In view of the low risks associated with IVIG administration and the high risks of coronary artery aneurysms among children who do not receive timely treatment, the current algorithm should be applied to the child with suspected incomplete KD until an evidence-based algorithm or a specific diagnostic test for KD becomes available.
IVIG should be instituted as early as possible within the first 10 days of illness onset of fever, that is, as soon as the diagnosis can be established. Patients with a delayed diagnosis of KD (ie, later than day 10 of fever) may still be candidates for treatment. IVIG should also be administered to children presenting after the tenth day of illness (ie, in whom the diagnosis was missed earlier) if they have ongoing systemic inflammation as manifested by elevation of ESR or CRP (CRP >3.0 mg/dL) together with either persistent fever without other explanation or coronary artery aneurysms (luminal dimension Z score >2.5). Those in whom fever has resolved and laboratory values have normalized and whose echocardiograms are normal do not require IVIG treatment. Patients with recurrent KD, defined as a repeat episode of complete or incomplete KD after complete resolution of the previous episode, should receive standard therapy with IVIG and ASA.