In this post, I link to and excerpt from Long-term cardiovascular outcomes of COVID-19 [PubMed Abstract] [Full-Text HTML] [Full-Text PDF] from Nature Medicine. Published: 07 February 2022.
All that follows is from the above resource.
Abstract
The cardiovascular complications of acute coronavirus disease 2019 (COVID-19) are well described, but the post-acute cardiovascular manifestations of COVID-19 have not yet been comprehensively characterized. Here we used national healthcare databases from the US Department of Veterans Affairs to build a cohort of 153,760 individuals with COVID-19, as well as two sets of control cohorts with 5,637,647 (contemporary controls) and 5,859,411 (historical controls) individuals, to estimate risks and 1-year burdens of a set of pre-specified incident cardiovascular outcomes. We show that, beyond the first 30 d after infection, individuals with COVID-19 are at increased risk of incident cardiovascular disease spanning several categories, including cerebrovascular disorders, dysrhythmias, ischemic and non-ischemic heart disease, pericarditis, myocarditis, heart failure and thromboembolic disease. These risks and burdens were evident even among individuals who were not hospitalized during the acute phase of the infection and increased in a graded fashion according to the care setting during the acute phase (non-hospitalized, hospitalized and admitted to intensive care). Our results provide evidence that the risk and 1-year burden of cardiovascular disease in survivors of acute COVID-19 are substantial. Care pathways of those surviving the acute episode of COVID-19 should include attention to cardiovascular health and disease.
Main
Post-acute sequelae of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)—the virus that causes coronavirus disease 2019 (COVID-19)—can involve the pulmonary and several extrapulmonary organs, including the cardiovascular system1.
In this study, we used the US Department of Veterans Affairs national healthcare databases to build a cohort of 153,760 US veterans who survived the first 30 d of COVID-19 and two control groups: a contemporary cohort consisting of 5,637,647 users of the US Veterans Health Administration (VHA) system with no evidence of SARS-CoV-2 infection and a historical cohort (pre-dating the COVID-19 pandemic) consisting of 5,859,411 non-COVID-19-infected VHA users during 2017. These cohorts were followed longitudinally to estimate the risks and 12-month burdens of pre-specified incident cardiovascular outcomes in the overall cohort and according to care setting of the acute infection (non-hospitalized, hospitalized and admitted to intensive care).
Results
Cerebrovascular disorders
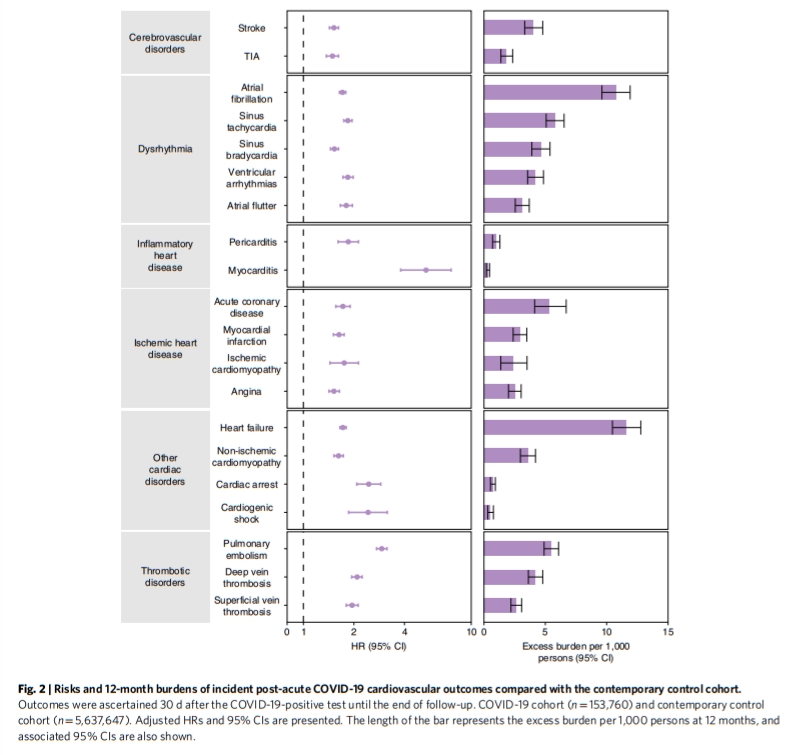
People who survived the first 30 d of COVID-19 exhibited increased risk of stroke (hazard ratio (HR) = 1.52 (1.43, 1.62); burden 4.03 (3.32, 4.79) per 10,00 persons at 12 months; for all HRs and burdens, parenthetical ranges refer to 95% confidence intervals (CIs)) and transient ischemic attacks (TIA) (HR = 1.49 (1.37, 1.62); burden 1.84 (1.38, 2.34)). The risks and burdens of a composite of these cerebrovascular outcomes were 1.53 (1.45, 1.61) and 5.48 (4.65, 6.35).
Dysrhythmias
There were increased risks of atrial fibrillation (HR = 1.71 (1.64, 1.79); burden 10.74 (9.61, 11.91)), sinus tachycardia (HR = 1.84 (1.74, 1.95); burden 5.78 (5.07, 6.53)), sinus bradycardia (HR = 1.53 (1.45, 1.62); burden 4.62 (3.90, 5.38)), ventricular arrhythmias (HR = 1.84 (1.72, 1.98); burden 4.18 (3.56, 4.85)); and atrial flutter (HR = 1.80 (1.66, 1.96); burden 3.10 (2.55, 3.69)). The risks and burdens of a composite of these dysrhythmia outcomes were 1.69 (1.64, 1.75), and 19.86 (18.31, 21.46).
Inflammatory disease of the heart or pericardium
Inflammatory disease of the heart or pericardium included pericarditis (HR = 1.85 (1.61, 2.13)); burden 0.98 (0.70, 1.30) and myocarditis (HR = 5.38 (3.80, 7.59); burden 0.31 (0.20, 0.46)). The risks and burdens of a composite of these inflammatory diseases of the heart or pericardium were 2.02 (1.77, 2.30) and 1.23 (0.93, 1.57).
Ischemic heart disease
Ischemic heart disease included acute coronary disease (HR = 1.72 (1.56, 1.90); burden 5.35 (4.13, 6.70)), myocardial infarction (HR = 1.63 (1.51, 1.75); burden 2.91 (2.38, 3.49)), ischemic cardiomyopathy (HR = 1.75 (1.44, 2.13); burden 2.34 (1.37, 3.51)) and angina (HR = 1.52 (1.42, 1.64); burden 2.50 (2.00, 3.03)). The risks and burdens of a composite of these ischemic heart disease outcomes were 1.66 (1.52, 1.80) and 7.28 (5.80, 8.88).
Other cardiovascular disorders
Other cardiovascular disorders included heart failure (HR = 1.72 (1.65, 1.80); burden 11.61 (10.47, 12.78)), non-ischemic cardiomyopathy (HR = 1.62 (1.52, 1.73); burden 3.56 (2.97, 4.20)), cardiac arrest (HR = 2.45 (2.08, 2.89); burden 0.71 (0.53, 0.93)) and cardiogenic shock (HR = 2.43 (1.86, 3.16); burden 0.51 (0.31, 0.77)). The risks and burdens of a composite of these other cardiovascular disorders were 1.72 (1.65, 1.79) and 12.72 (11.54, 13.96).
Thromboembolic disorders
Thromboembolic disorders included pulmonary embolism (HR = 2.93 (2.73, 3.15); burden 5.47 (4.90, 6.08)); deep vein thrombosis (HR = 2.09 (1.94, 2.24); burden 4.18 (3.62, 4.79)) and superficial vein thrombosis (HR = 1.95 (1.80, 2.12); burden 2.61 (2.20, 3.07)). The risks and burdens of a composite of these thromboembolic disorders were 2.39 (2.27, 2.51) and 9.88 (9.05, 10.74).
Additional composite endpoints
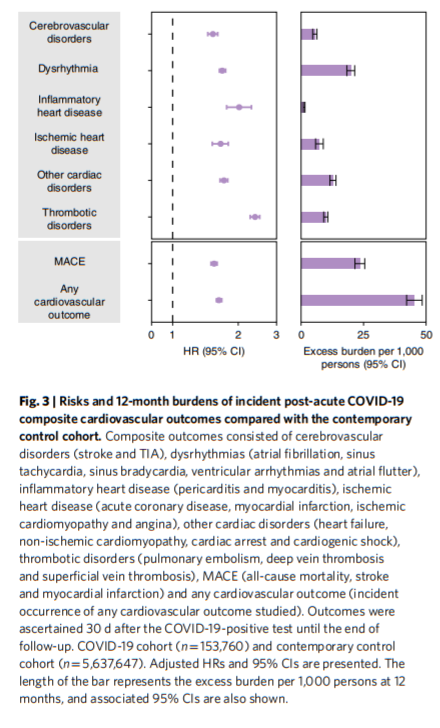
We then examined the risks and burdens of two composite endpoints, including major adverse cardiovascular event (MACE)—a composite of myocardial infarction, stroke and all-cause mortality—and any cardiovascular outcome (defined as the occurrence of any incident pre-specified cardiovascular outcome included in this study). Compared to the contemporary control group, there were increased risks and burdens of MACE (HR = 1.55 (1.50, 1.60); burden 23.48 (21.54, 25.48)) and any cardiovascular outcome (HR = 1.63 (1.59, 1.68); burden 45.29 (42.22, 48.45)).
Subgroup analyses
We examined the risks of incident composite cardiovascular outcomes in subgroups based on age, race, sex, obesity, smoking, hypertension, diabetes, chronic kidney disease, hyperlipidemia and cardiovascular disease. The risks of incident composite cardiovascular outcomes were evident in all subgroups (Fig. 4 and Supplementary Table 4),
Discussion
. . . we provide evidence that, beyond the first 30 d of infection, people with COVID-19 exhibited increased risks and 12-month burdens of incident cardiovascular diseases, including cerebrovascular disorders, dysrhythmias, inflammatory heart disease, ischemic heart disease, heart failure, thromboembolic disease and other cardiac disorders.
The risks were evident regardless of age, race, sex and other cardiovascular risk factors, including obesity, hypertension, diabetes, chronic kidney disease and hyperlipidemia; they were also evident in people without any cardiovascular disease before exposure to COVID-19, providing evidence that these risks might manifest even in people at low risk of cardiovascular disease.
Our analyses of the risks and burdens of cardiovascular outcomes across care settings of the acute infection reveal two key findings: (1) that the risks and associated burdens were evident among those who were not hospitalized during the acute phase of the disease—this group represents the majority of people with COVID-19; and (2) that the risks and associated burdens exhibited a graded increase across the severity spectrum of the acute phase of COVID-19 (from non-hospitalized to hospitalized individuals to those admitted to intensive care). The risks and associated burdens were consistent in analyses considering the contemporary control group and, separately, the historical control group as the referent category.
The difference-in-differences analyses, which are designed to further investigate the causality of study findings, show that the increased risks of post-acute COVID-19 cardiovascular outcomes are attributable sequelae to COVID-19 itself. The results were robust to challenge in multiple sensitivity analyses.
Taken together, our results show that 1-year risks and burdens of cardiovascular diseases among those who survive the acute phase of COVID-19 are substantial and span several cardiovascular disorders. Care strategies of people who survived the acute episode of COVID-19 should include attention to cardiovascular health and disease.
The mechanism or mechanisms that underlie the association between COVID-19 and development of cardiovascular diseases in the post-acute phase of the disease are not entirely clear11,12. Putative mechanisms include lingering damage from direct viral invasion of cardiomyocytes and subsequent cell death, endothelial cell infection and endotheliitis, transcriptional alteration of multiple cell types in heart tissue, complement activation and complement-mediated coagulopathy and microangiopathy, downregulation of ACE2 and dysregulation of the renin–angiotensin–aldosterone system, autonomic dysfunction, elevated levels of pro-inflammatory cytokines and activation of TGF-β signaling through the Smad pathway to induce subsequent fibrosis and scarring of cardiac tissue11,13,14,15,16,17. An aberrant persistent hyperactivated immune response, autoimmunity or persistence of the virus in immune-privileged sites has also been cited as putative explanations of extrapulmonary (including cardiovascular) post-acute sequelae of COVID-19 (refs. 11,13,14,18). Integration of the SARS-CoV-2 genome into DNA of infected human cells, which might then be expressed as chimeric transcripts fusing viral with cellular sequences, has also been hypothesized as a putative mechanism for continued activation of the immune-inflammatory-procoagulant cascade19,20. These mechanistic pathways might explain the range of post-acute COVID-19 cardiovascular sequelae investigated in this report. A deeper understanding of the biologic mechanisms will be needed to inform development of prevention and treatment strategies of the cardiovascular manifestations among people with COVID-19.