The following are excerpts from Reference (1) in Resources:
Introduction
A drug-drug interaction (DDI) is a pharmacokinetic or pharmacological influence of 1 medication on another that differs from the known or anticipated effects of each agent alone.1 A DDI may result in a change in either drug efficacy or drug toxicity for 1 or both of the interacting medications.2 Pharmacokinetic DDIs result in altered absorption, distribution, metabolism, or excretion of a medication. A pharmacodynamic DDI occurs when 1 medication modifies the pharmacological effect of another in an additive, a synergistic, or an antagonistic fashion.
It is estimated that ≈2.8% of hospital admissions occur as a direct result of DDIs.3 However, the actual incidence of hospitalization secondary to clinically significant DDIs is likely to be highly underestimated because medication-related issues are more commonly reported as adverse drug reactions. Complex underlying disease states also may make recognizing a DDI more challenging, further contributing to a lower reported incidence. The overall clinical impact of a DDI can range from mild to life-threatening. Therefore, not all DDIs require a modification in therapy. The variability in the clinical significance of a DDI depends on both medication-specific and patient-specific factors. Medication-specific factors include the individual pharmacokinetic characteristics of each medication implicated in the DDI (eg, binding affinity, half-life [t1/2]), dose of the medications, serum concentrations, timing and sequence of administration, and duration of therapy. Patient-specific factors include age, sex, lifestyle, genetic polymorphisms causing differences in enzyme expression or activity, and disease impairment affecting drug metabolism (eg, hepatic or renal impairment, cardiac failure) or predisposition to differences in efficacy or safety (eg, statin intolerance in patients with a history of myopathy).
Clinically significant DDIs are usually preventable. To optimize patient safety, healthcare providers must have an understanding of the mechanisms, magnitude, and potential consequences of any given DDI. Interpreting this information will assist clinicians in the safe prescribing of medications and permits careful consideration of the benefits and risks of concomitant medications.
Statins reduce morbidity and mortality in patients with known atherosclerotic cardiovascular disease (ASCVD) and in many primary prevention patients.4–9 Current guidelines recommend high-intensity statin therapy in all patients with ASCVD age ≤75 years and moderate- to high-intensity statin therapy in patients with ASCVD and age >75 years, diabetes mellitus, and familial hypercholesterolemia and in primary prevention patients with 10-year ASCVD risk ≥7.5%.10 Given the important role of statins in patients with ASCVD and those at high ASCVD risk, combination therapy with statins and other cardiovascular medications is highly likely, and potentially significant DDIs must be considered in patients treated with statins.
Another important aspect of prescribing medications in combination is evaluating the risks versus benefits. Given the continuing increase in healthcare costs, trying to minimize costs to the health system through minimization of adverse effects and optimizing efficacy is of paramount importance. Prescription drug coverage and affordability may often dictate which medications may be prescribed. Patients who have exhausted prescription drug coverage or who are uninsured may require less-than-ideal medication combinations to provide the most cost-effective strategy possible. Therefore, a clear understanding of the magnitude and clinical significance of DDIs will enable clinicians to make well-informed decisions to provide evidence-based and cost-effective health care as safely as possible.
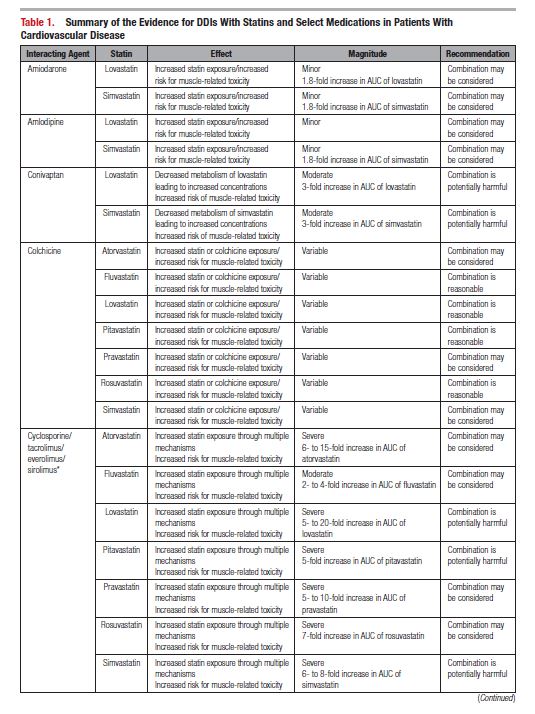
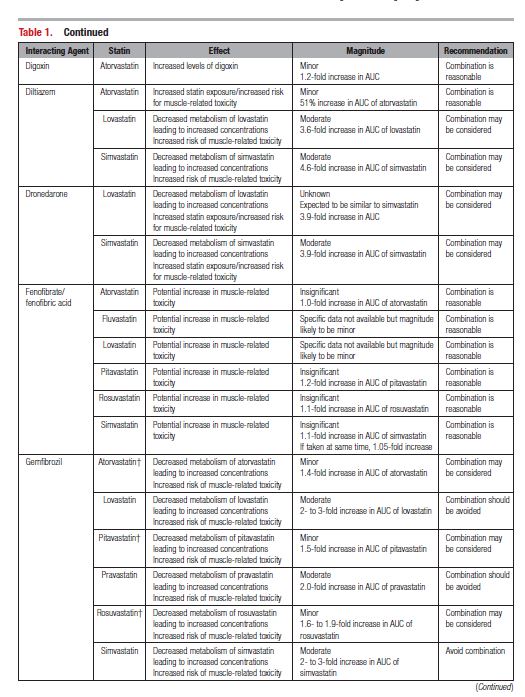
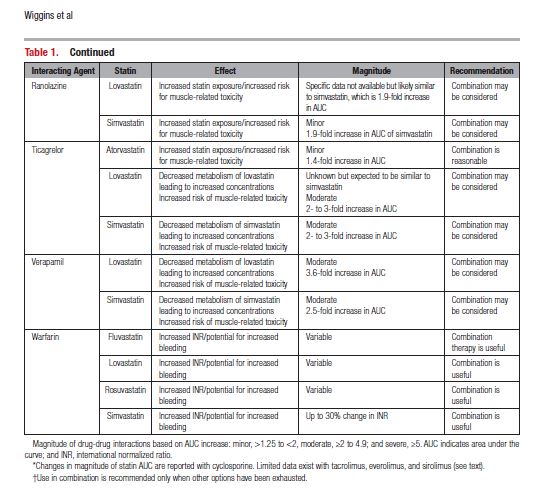
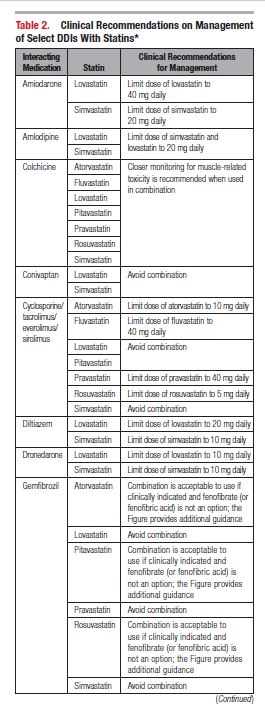
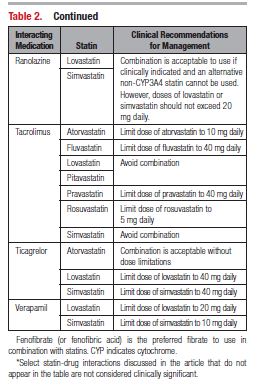
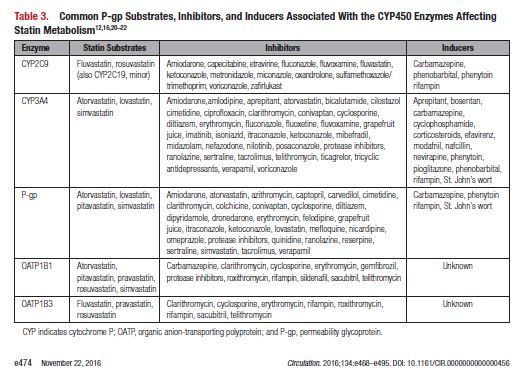
This document reviews the basics of DDIs, the pharmacological differences in the various statins, and the significance of statin DDIs with select medications used to treat patients with cardiovascular disease. Recommendations on the clinical management of these DDIs are provided to enable clinicians to optimize management while minimizing untoward effects. The writing committee considered data from clinical trials, case reports, prescribing information, and pharmacokinetic studies to provide guidance on how statin DDIs with select medications used in cardiovascular patients should be managed. A summary of the evidence and the specific recommendations for the clinical management of the DDIs discussed are given in Tables 1 and 2.
Resources:
(1) Statins and Select Agents Used in Patients With Cardiovascular Disease: A Scientific
Recommendations for Management of Clinically Significant Drug-Drug Interactions With Statement From the American Heart Association [PubMed Abstract] [Full Text HTML]. Circulation. 2016 Nov 22;134(21):e468-e495. Epub 2016 Oct 17.